Advanced Iridium-Catalyzed Synthesis of Chiral 1-Oxabenzoheterocycles for Commercial Pharmaceutical Intermediates
Advanced Iridium-Catalyzed Synthesis of Chiral 1-Oxabenzoheterocycles for Commercial Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds that serve as the backbone for numerous bioactive molecules. Patent CN101921252A introduces a groundbreaking methodology for the synthesis of optically active 1-oxabenzoheterocyclic compounds, utilizing a sophisticated iridium complex catalytic system. This technology represents a significant leap forward in asymmetric catalysis, enabling the construction of chiral chromene and benzoxepine derivatives with exceptional enantioselectivity. The core innovation lies in the tandem combination of allylic etherification and ring-closing metathesis, which streamlines the production of these valuable intermediates. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial, as it offers a robust alternative to traditional multi-step syntheses that often suffer from poor stereocontrol and harsh reaction conditions. The ability to generate these structures with high optical purity directly impacts the efficacy and safety profile of downstream drug candidates, making this intellectual property a vital asset for any organization focused on high-value pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzopyran and related 1-oxabenzoheterocyclic frameworks has relied on a variety of classical organic transformations that frequently present significant operational challenges for large-scale manufacturing. Traditional routes often involve multiple discrete steps, requiring the isolation and purification of unstable intermediates, which drastically reduces overall process efficiency and increases waste generation. Many conventional methods utilize stoichiometric amounts of chiral auxiliaries or resolving agents, leading to a theoretical maximum yield of only 50% for the desired enantiomer unless dynamic kinetic resolution is employed, which adds further complexity. Furthermore, older methodologies often necessitate the use of strong acids, high temperatures, or toxic heavy metal reagents that pose severe safety hazards and environmental compliance issues for modern chemical plants. The lack of regioselectivity in traditional allylation reactions can also result in complex mixtures of isomers, necessitating costly and time-consuming chromatographic separations that are impractical for commercial production. These inherent limitations create bottlenecks in the supply chain, extending lead times and inflating the cost of goods sold for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101921252A employs a highly efficient iridium-catalyzed asymmetric allylic etherification followed by a ruthenium-catalyzed ring-closing metathesis. This novel approach allows for the direct construction of the heterocyclic core from readily available phenolic compounds and allyl carbonates in a streamlined fashion. The use of chiral iridium complexes ensures that the stereogenic center is established with high fidelity during the initial bond-forming event, thereby obviating the need for downstream resolution steps. The reaction conditions are notably mild, typically operating between 40°C and 80°C, which significantly reduces energy consumption and minimizes the thermal degradation of sensitive functional groups. By integrating the etherification and cyclization steps, this methodology achieves superior atom economy and step economy, directly translating to reduced solvent usage and lower waste disposal costs. The versatility of this system is further evidenced by its tolerance to a wide range of substituents on both the phenolic and allylic components, allowing for the rapid generation of diverse compound libraries for drug discovery programs without the need for extensive process re-optimization.
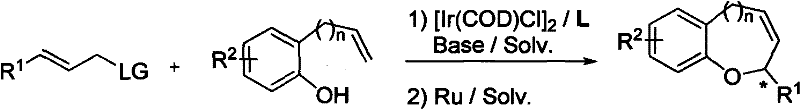
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylic Etherification and RCM
The success of this synthetic route hinges on the precise orchestration of the catalytic cycle involving the iridium complex and the chiral ligand. The mechanism initiates with the oxidative addition of the allyl carbonate to the low-valent iridium center, forming a pi-allyl iridium intermediate that is stabilized by the coordination of the chiral phosphine-amine ligand. This ligand environment creates a specific chiral pocket that dictates the facial selectivity of the nucleophilic attack by the phenolic oxygen, ensuring the formation of the desired enantiomer with high optical purity. The electronic properties of the ligand, such as the steric bulk of the binaphthyl backbone, play a critical role in suppressing competing side reactions and enhancing the turnover number of the catalyst. Following the etherification, the resulting intermediate undergoes a ring-closing metathesis (RCM) mediated by a ruthenium carbene catalyst, such as Zhan-1B, to close the ring and form the final heterocyclic structure. This tandem sequence is particularly elegant because it avoids the isolation of the linear ether intermediate, thereby reducing material handling and potential yield losses. Understanding this mechanistic pathway is essential for process chemists aiming to troubleshoot potential issues during scale-up, such as catalyst deactivation or incomplete conversion, ensuring that the high enantioselectivity observed on a laboratory scale is maintained in commercial reactors.
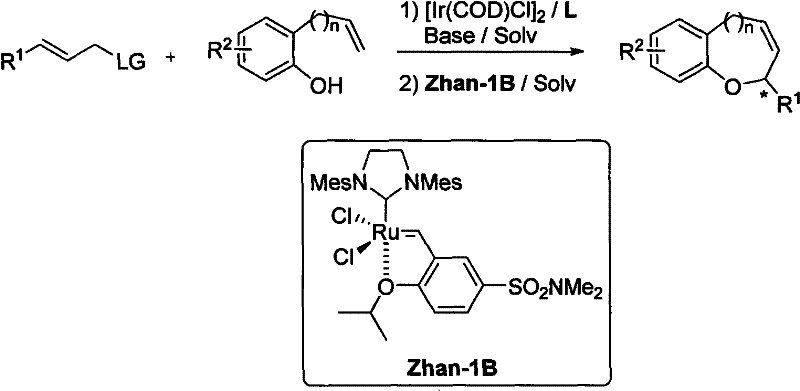
Impurity control is another critical aspect of this mechanism that offers substantial benefits for pharmaceutical manufacturing. The high regioselectivity of the iridium-catalyzed allylation ensures that the branched product is favored over the linear isomer, which is a common impurity in non-catalyzed allylation reactions. This selectivity is driven by the specific geometry of the pi-allyl iridium complex and the steric constraints imposed by the chiral ligand. Furthermore, the mild reaction conditions minimize the formation of decomposition products or polymerization byproducts that often plague high-temperature processes. The subsequent RCM step is also highly selective, driven by the release of ethylene gas, which pushes the equilibrium towards the cyclic product. This inherent cleanliness of the reaction profile simplifies the downstream purification process, often allowing for crystallization or simple distillation rather than complex chromatography. For quality control teams, this means a more consistent impurity profile and a reduced risk of genotoxic impurities carrying through to the final active pharmaceutical ingredient, thereby facilitating regulatory approval and ensuring patient safety.
How to Synthesize 1-Oxabenzoheterocyclic Compound Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and enantioselectivity. The process begins with the preparation of the active iridium catalyst species by mixing the iridium precursor with the chiral ligand in a suitable solvent, followed by the addition of the phenolic substrate and allyl carbonate. It is imperative to maintain an inert atmosphere to prevent catalyst oxidation, which can lead to reduced activity. The reaction temperature should be carefully controlled within the recommended range to balance reaction rate and selectivity. After the completion of the etherification step, the ruthenium catalyst is introduced to effect the ring closure. Detailed standard operating procedures for this synthesis, including specific molar ratios, solvent choices, and workup protocols, are essential for reproducible manufacturing. For a comprehensive guide on the exact experimental conditions and optimization strategies, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining phenolic compounds and allyl carbonate compounds with an iridium catalyst complex and chiral ligand in an organic solvent.
- Conduct the allylic etherification reaction under mild heating conditions (40°C to 80°C) for 4 to 24 hours to form the intermediate ether.
- Perform the subsequent ring-closing metathesis (RCM) using a Ruthenium catalyst to cyclize the intermediate into the final 1-oxabenzoheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed technology offers profound advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies for pharmaceutical intermediates. The primary benefit lies in the significant cost reduction in fine chemical manufacturing achieved through process intensification. By combining multiple synthetic steps into a streamlined sequence, the overall processing time is drastically reduced, leading to lower labor costs and increased throughput in existing production facilities. The elimination of resolution steps further enhances the effective yield, meaning less raw material is required to produce the same amount of final product, which directly impacts the cost of goods. Additionally, the use of readily available starting materials such as substituted phenols and allyl carbonates ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that can disrupt production schedules. The mild reaction conditions also translate to lower energy consumption and reduced wear and tear on reactor equipment, contributing to long-term operational savings and sustainability goals.
- Cost Reduction in Manufacturing: The streamlined nature of this catalytic process eliminates the need for expensive chiral resolving agents and reduces the number of isolation steps, which significantly lowers solvent consumption and waste disposal costs. The high catalytic efficiency means that precious metal loading can be optimized, reducing the overall cost of the catalyst system while maintaining high performance. Furthermore, the simplified purification requirements reduce the reliance on costly chromatographic resins and silica gel, allowing for more economical crystallization or distillation techniques to be employed. These factors combine to create a highly cost-competitive manufacturing route that can withstand market pressure and pricing fluctuations in the pharmaceutical intermediate sector.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent product quality and supply continuity, which is critical for meeting the demanding timelines of drug development projects. The wide substrate scope allows for the flexible production of various analogues using the same core process infrastructure, enabling rapid response to changing market demands or clinical trial requirements. The use of standard organic solvents and common reagents means that supply chains are not dependent on exotic or hard-to-source chemicals, reducing the risk of logistical bottlenecks. This reliability fosters stronger partnerships between chemical suppliers and pharmaceutical companies, as it guarantees the on-time delivery of high-purity intermediates necessary for maintaining clinical and commercial production schedules.
- Scalability and Environmental Compliance: This process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory glassware to industrial-scale reactors without significant re-engineering. The mild temperatures and pressures involved reduce the safety risks associated with high-energy processes, facilitating easier regulatory approval for new manufacturing sites. Moreover, the improved atom economy and reduced waste generation align with green chemistry principles, helping companies meet increasingly stringent environmental regulations and corporate sustainability targets. The ability to produce high-value intermediates with a lower environmental footprint enhances the brand reputation of manufacturers and provides a competitive edge in markets where eco-friendly sourcing is a priority for downstream pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The responses cover aspects of catalytic performance, substrate compatibility, and downstream processing, ensuring a comprehensive overview of the technology's capabilities.
Q: What are the primary advantages of this iridium-catalyzed method over traditional synthesis?
A: This method offers high enantioselectivity (86-96% ee) and operates under mild reaction conditions, eliminating the need for harsh reagents often required in conventional chromene synthesis.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available raw materials and standard organic solvents, making it highly suitable for commercial scale-up from kilogram to metric ton quantities.
Q: What is the substrate scope for this 1-oxabenzoheterocycle synthesis?
A: The method demonstrates a wide substrate application range, accommodating various phenolic compounds and allyl carbonates with different electronic and steric properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Oxabenzoheterocyclic Compound Supplier
The technological potential of this iridium-catalyzed synthesis route is immense, offering a pathway to high-value chiral intermediates that are essential for the development of next-generation therapeutics. NINGBO INNO PHARMCHEM, as a premier CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry from the lab to the market. Our state-of-the-art facilities are equipped to handle sensitive catalytic reactions under strict inert conditions, ensuring that the stringent purity specifications and rigorous QC labs standards are met for every batch. We understand the critical nature of chiral purity in pharmaceutical applications and have the analytical capabilities to verify enantiomeric excess and impurity profiles with the highest degree of accuracy. Partnering with us means gaining access to a team of dedicated process chemists who can optimize this route for your specific needs, ensuring a seamless transition from process development to commercial manufacturing.
We invite you to initiate a dialogue with our technical procurement team to explore how this technology can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this catalytic method for your specific intermediates. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our commitment to transparency and technical excellence ensures that you receive the data needed to make confident sourcing decisions. Let us help you secure a reliable supply of high-quality 1-oxabenzoheterocyclic compounds that will accelerate your drug development timelines and reduce your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
