Advanced Manufacturing of Planar Chiral Ferrocene Compounds for Asymmetric Catalysis
The landscape of asymmetric catalysis is undergoing a significant transformation with the advent of direct C-H functionalization strategies, as detailed in patent CN102964390A. This groundbreaking technology introduces a highly efficient method for synthesizing planar chiral ferrocene compounds, which serve as critical precursors for advanced chiral ligands. Unlike traditional approaches that rely on complex multi-step sequences, this innovation leverages palladium-catalyzed asymmetric C-H activation using commercially available chiral amino acids as ligands. For R&D directors and procurement specialists seeking a reliable planar chiral ferrocene supplier, this methodology represents a paradigm shift towards more sustainable and cost-effective manufacturing processes. The ability to directly install planar chirality onto the ferrocene scaffold without pre-functionalization offers substantial advantages in terms of atom economy and step reduction, addressing key pain points in the supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of planar chiral ferrocenes has been fraught with significant challenges that hinder large-scale adoption in the fine chemical industry. The most prevalent strategy involves diastereoselective ortho-metalation induced by chiral auxiliaries, which necessitates the pre-installation of central chirality on the ferrocene ring. This approach is inherently inefficient as it requires additional synthetic steps to introduce and subsequently remove the directing group, leading to increased waste generation and lower overall yields. Furthermore, alternative methods developed by research groups such as Snieckus utilize external chiral bases like (-)-sparteine in conjunction with stoichiometric amounts of hazardous lithium reagents. These reactions often demand苛刻 cryogenic conditions and strictly anhydrous environments, posing severe safety risks and escalating operational costs for manufacturing facilities. The reliance on expensive, difficult-to-source chiral inducers and the generation of stoichiometric metal waste make these conventional routes economically unviable for the commercial scale-up of complex polymer additives or electronic materials.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a catalytic system driven by palladium and chiral amino acid derivatives to achieve direct asymmetric C-H functionalization. This method bypasses the need for pre-installed chirality or stoichiometric chiral reagents, utilizing simple aminomethyl-substituted ferrocene derivatives and various boronic acids as starting materials. The reaction proceeds under mild conditions, typically between 60°C and 80°C, using air or oxygen as a benign oxidant, which drastically simplifies the engineering requirements for reactor design. By employing commercially abundant chiral amino acids such as Boc-L-Val-OH as ligands, the process ensures high enantioselectivity while maintaining a low catalyst loading. This transition from stoichiometric to catalytic chirality induction not only enhances the sustainability profile of the synthesis but also significantly reduces the cost of goods sold (COGS), making it an attractive solution for cost reduction in pharmaceutical intermediate manufacturing.
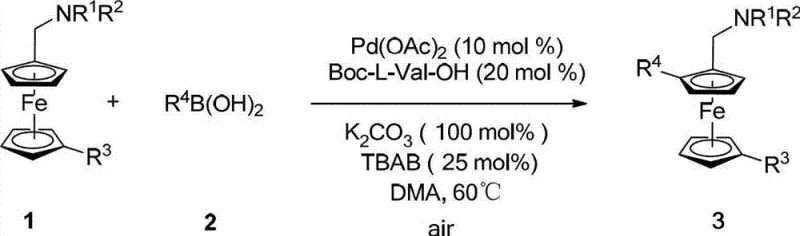
Mechanistic Insights into Pd-Catalyzed Asymmetric C-H Functionalization
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst and the chiral amino acid ligand, which creates a highly organized transition state for stereoselective C-H bond cleavage. The mechanism likely initiates with the coordination of the palladium species to the nitrogen atom of the aminomethyl group on the ferrocene, directing the metal center to the proximal C-H bond. The chiral environment provided by the amino acid ligand, particularly its bulky side chains, exerts precise steric control during the C-H activation step, ensuring that the metalation occurs exclusively on one face of the planar ferrocene ring. This kinetic resolution or dynamic kinetic asymmetric transformation is crucial for achieving the high enantiomeric excess values reported, often exceeding 95% ee. Following C-H activation, transmetallation with the boronic acid species occurs, facilitated by the base and phase transfer catalyst, followed by reductive elimination to release the chiral product and regenerate the active palladium catalyst. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters for specific substrate classes.
Furthermore, the impurity profile of the resulting ferrocene compounds is tightly controlled by the specificity of the catalytic cycle. Traditional lithiation methods often suffer from over-lithiation or non-selective metalation at multiple positions on the cyclopentadienyl rings, leading to complex mixtures that are difficult to separate. In this palladium-catalyzed system, the directing effect of the amine group combined with the steric bulk of the ligand minimizes side reactions such as homocoupling of the boronic acid or non-selective arylation. The use of air as the terminal oxidant also prevents the accumulation of reduced palladium black, which can catalyze unwanted decomposition pathways. This high level of chemoselectivity ensures that the crude reaction mixture contains predominantly the desired planar chiral isomer, simplifying downstream purification processes and enhancing the overall purity specifications required for sensitive applications in asymmetric catalysis and materials science.
How to Synthesize Planar Chiral Ferrocene Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to a standardized protocol that maximizes catalyst turnover and enantioselectivity. The process begins with the careful selection of the chiral amino acid ligand, where N-protected variants like Boc-L-Val-OH have shown superior performance in screening studies. The reaction is typically conducted in polar aprotic solvents such as N,N-dimethylacetamide (DMA), which solubilizes both the organic substrates and the inorganic base. Maintaining an aerobic environment is critical, as molecular oxygen serves as the green oxidant to close the catalytic cycle, eliminating the need for hazardous chemical oxidants like silver salts or benzoquinone. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining aminomethyl-substituted ferrocene derivatives, aryl boronic acids, palladium acetate, and a chiral amino acid ligand such as Boc-L-Val-OH in a suitable solvent like DMA.
- Add inorganic bases like potassium carbonate and phase transfer catalysts such as tetrabutylammonium bromide to facilitate the reaction under aerobic conditions.
- Heat the mixture to moderate temperatures between 60°C and 80°C for 6 to 24 hours, then isolate the high-enantiopurity product via standard extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers transformative benefits that extend beyond mere technical feasibility. The shift towards a catalytic process using commodity chemicals fundamentally alters the cost structure and risk profile of producing high-value chiral intermediates. By replacing expensive, specialized chiral reagents with bulk-available amino acids, the raw material costs are drastically simplified, leading to substantial cost savings in the final product pricing. Moreover, the operational simplicity of running reactions at moderate temperatures under air atmosphere reduces the energy footprint and eliminates the need for specialized cryogenic equipment, thereby enhancing the overall supply chain reliability and reducing capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and hazardous lithium reagents results in a significantly leaner bill of materials. Since the chiral information is imparted by a catalytic amount of inexpensive amino acid derivatives rather than a stoichiometric quantity of complex chiral bases, the direct material costs are minimized. Additionally, the use of air as the oxidant removes the expense associated with purchasing and disposing of chemical oxidants, further driving down the operational expenditures. This economic efficiency allows for more competitive pricing strategies when supplying high-purity OLED material or pharmaceutical building blocks to global markets.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as aryl boronic acids and protected amino acids ensures a robust and resilient supply chain. Unlike specialized chiral ligands that may have long lead times or single-source dependencies, the key components of this reaction are produced by multiple vendors worldwide, mitigating the risk of supply disruptions. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites without requiring extensive re-validation of harsh safety protocols, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The absence of heavy metal waste streams associated with stoichiometric lithiation and the use of benign oxidants simplify waste treatment procedures, reducing the environmental compliance burden. The process is inherently scalable, as demonstrated by the successful synthesis of diverse substrates including sterically hindered biaryls and heterocycles, proving its versatility for commercial scale-up of complex polymer additives. This scalability ensures that production volumes can be ramped up quickly to meet market demand without compromising on the stringent purity specifications required by end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this planar chiral ferrocene synthesis technology. These insights are derived directly from the experimental data and scope of the patent, providing clarity on the method's capabilities and limitations for potential partners and licensees.
Q: What are the advantages of using chiral amino acids over traditional chiral auxiliaries?
A: Using commercially available chiral amino acids eliminates the need for stoichiometric amounts of expensive chiral bases or pre-installed central chirality, significantly reducing raw material costs and simplifying the synthetic route.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes air as a green oxidant and operates at mild temperatures without cryogenic conditions, making it highly suitable for commercial scale-up and reducing energy consumption.
Q: What level of enantioselectivity can be achieved with this catalytic system?
A: The patented process demonstrates excellent stereocontrol, consistently achieving enantiomeric excess (ee) values exceeding 90%, with many substrates reaching up to 99% ee.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Planar Chiral Ferrocene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced chiral technologies in driving innovation across the pharmaceutical and fine chemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing, guaranteeing that every batch of planar chiral ferrocene meets the exacting standards required for downstream asymmetric catalysis applications. Our infrastructure is designed to handle complex organometallic chemistry safely and effectively, positioning us as a trusted partner for your most challenging synthesis projects.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. Whether you require custom synthesis of novel ferrocene ligands or large-scale production of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project goals. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
