Advanced Electrochemical Strategy for Scalable Production of Aryl-Substituted Quinoxaline Intermediates
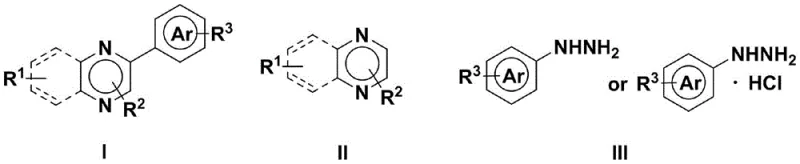
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for heterocyclic scaffolds, particularly quinoxaline derivatives, which serve as critical cores in antitumor, antibacterial, and phosphatase inhibitor drug candidates. Patent CN113774411B introduces a groundbreaking electrochemical methodology for synthesizing aryl-substituted quinoxaline (ketone) derivatives, addressing the longstanding challenges of cost and environmental impact in fine chemical manufacturing. This innovation leverages direct electrocatalytic coupling between quinoxaline precursors and arylhydrazine compounds, bypassing the need for transition metal catalysts. By utilizing electricity as a traceless reagent, this technology offers a sustainable alternative to traditional thermal condensation or cross-coupling reactions, ensuring high purity profiles essential for regulatory compliance in active pharmaceutical ingredient (API) production.
As a reliable pharmaceutical intermediates supplier, understanding the shift towards green chemistry is paramount for maintaining competitive advantage. The disclosed method operates under mild conditions, typically between 10°C and 40°C, using common electrolytes such as tetrabutylammonium tetrafluoroborate and solvents like acetonitrile or DMSO. This approach not only simplifies the operational complexity but also aligns with global sustainability goals by minimizing hazardous waste generation. For procurement managers and supply chain heads, adopting such technologies translates to reduced dependency on volatile precious metal markets and streamlined waste disposal protocols, ultimately securing a more resilient supply chain for complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl-substituted quinoxaline derivatives has relied heavily on transition metal-catalyzed cross-coupling reactions, such as Suzuki or Heck couplings, which necessitate the use of expensive palladium catalysts. These traditional routes often suffer from significant drawbacks, including the difficulty in removing trace metal residues to meet stringent pharmaceutical purity specifications, which can drastically increase downstream processing costs. Furthermore, alternative C-H functionalization methods reported in literature frequently require stoichiometric amounts of chemical oxidants, leading to substantial atom inefficiency and the generation of large volumes of chemical waste that complicate environmental compliance. The harsh reaction conditions often associated with these legacy methods, such as high temperatures and strong bases, can also limit substrate scope and lead to decomposition of sensitive functional groups, thereby reducing overall yield and process reliability.
The Novel Approach
In stark contrast, the electrochemical synthesis method described in patent CN113774411B utilizes electric current to drive the oxidative coupling reaction, effectively replacing both the metal catalyst and the chemical oxidant. This novel approach enables the direct C-H arylation of quinoxaline rings with arylhydrazines under exceptionally mild conditions, typically stirring at room temperature (25°C) for 2 to 10 hours with current intensities ranging from 5 to 20 mA per mmol. The elimination of heavy metals removes the burden of extensive purification steps required for metal scavenging, while the absence of stoichiometric oxidants significantly improves the atom economy of the process. This paradigm shift allows for the efficient production of diverse derivatives, including those with sensitive substituents like halogens or trifluoromethyl groups, demonstrating superior regioselectivity and operational simplicity suitable for industrial scale-up.
Mechanistic Insights into Electrochemical C-H Arylation
The core mechanism of this transformation involves the anodic oxidation of the arylhydrazine species to generate reactive nitrogen-centered radicals or cationic intermediates, which subsequently attack the electron-deficient quinoxaline ring system. In the electrolytic cell, the anode, often composed of graphite carbon or platinum, facilitates the single-electron transfer process that activates the hydrazine coupling partner without the need for external chemical oxidants. The cathode, typically platinum or nickel foam, balances the circuit by reducing protons to hydrogen gas or facilitating other reduction half-reactions, maintaining charge neutrality within the reaction medium. This electrocatalytic cycle ensures a continuous regeneration of active species, driving the reaction forward with high efficiency while minimizing side reactions that typically plague thermal radical processes.
From an impurity control perspective, the electrochemical pathway offers distinct advantages by avoiding the formation of metal-ligand complexes that can persist as difficult-to-remove contaminants. The reaction environment, moderated by additives such as trifluoroacetic acid or hydrobromic acid, helps stabilize the intermediates and directs the regioselectivity towards the desired C3-position of the quinoxaline ring. The use of supporting electrolytes like tetrabutylammonium hexafluorophosphate ensures sufficient conductivity in organic solvents, allowing for uniform current distribution and consistent reaction rates across the batch. This precise control over the reaction parameters results in a cleaner crude product profile, reducing the load on subsequent chromatographic purification steps and enhancing the overall throughput of the manufacturing process.
How to Synthesize Aryl-Substituted Quinoxaline Efficiently
To implement this electrochemical synthesis effectively, operators must carefully prepare the reaction mixture by dissolving the quinoxaline substrate and arylhydrazine reagent in a mixed solvent system, often comprising DMSO or acetonitrile with a small percentage of water to enhance solubility and conductivity. The addition of a suitable acid additive and electrolyte is critical to initiate the electrocatalytic cycle, followed by the insertion of inert electrodes and the application of a constant current. The detailed standardized synthesis steps, including specific molar ratios, electrode configurations, and workup procedures involving aqueous quenching and organic extraction, are outlined in the guide below to ensure reproducible high-yield results.
- Dissolve quinoxaline compounds, arylhydrazine compounds, additives (such as trifluoroacetic acid), and electrolyte (such as tetrabutylammonium tetrafluoroborate) in a solvent system like DMSO/water or acetonitrile/water.
- Place the reaction solution into an electrolytic cell equipped with a graphite carbon rod anode and a platinum sheet cathode, then connect the power supply.
- Apply a current intensity of 5-20 mA per mmol of substrate, stir at 10-40°C for 2-10 hours, and perform post-treatment involving quenching, extraction, and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology presents a compelling value proposition centered on cost stability and operational efficiency. By eliminating the requirement for palladium or copper catalysts, manufacturers can insulate their production costs from the volatility of precious metal markets, leading to significant long-term savings in raw material expenditures. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the simplified workup procedure minimizes the usage of auxiliary chemicals and solvents, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and stoichiometric oxidants directly lowers the bill of materials for every batch produced. Without the need for specialized metal scavengers or extensive purification trains to meet residual metal limits, the downstream processing costs are drastically simplified. This streamlined workflow allows for faster batch turnover and reduced labor hours, translating into substantial overall cost savings for the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary reagent decouples the synthesis from the supply constraints of specialized chemical oxidants or rare earth catalysts. The raw materials required, such as simple arylhydrazines and quinoxalines, are commodity chemicals with robust global supply chains, ensuring consistent availability. This resilience against supply chain disruptions guarantees uninterrupted production schedules, which is critical for meeting the just-in-time delivery demands of downstream API manufacturers.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for a seamless transition from laboratory optimization to commercial-scale production without significant re-engineering of the process. The green nature of this technology, characterized by minimal waste generation and the absence of toxic heavy metals, simplifies environmental permitting and waste treatment compliance. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of quinoxaline derivatives, based on the specific embodiments and data provided in the patent documentation. These insights are designed to clarify the feasibility and advantages of implementing this technology in a commercial setting.
Q: What are the primary advantages of this electrochemical method over traditional palladium-catalyzed routes?
A: The electrochemical method eliminates the need for expensive and toxic palladium catalysts, significantly reducing raw material costs and simplifying the removal of heavy metal impurities from the final pharmaceutical intermediate.
Q: Does this process require stoichiometric chemical oxidants?
A: No, unlike traditional C-H functionalization methods that rely on stoichiometric oxidants which generate substantial waste, this process uses electricity as the clean oxidant, making it more environmentally friendly and cost-effective.
Q: What is the typical reaction temperature range for this synthesis?
A: The reaction proceeds under mild conditions, typically between 10°C and 40°C, which enhances safety profiles and reduces energy consumption compared to high-temperature thermal condensation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl-Substituted Quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced electrochemical reactors capable of maintaining stringent purity specifications, guaranteeing that every batch meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with our technical team to explore how this metal-free synthesis route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your development timeline and reduce your overall manufacturing costs.
