Scalable Synthesis of Spatially Distinct Phosphocorrole Compounds for Advanced Photodynamic Therapy Applications
Scalable Synthesis of Spatially Distinct Phosphocorrole Compounds for Advanced Photodynamic Therapy Applications
The landscape of photodynamic therapy (PDT) is undergoing a significant transformation driven by the development of next-generation photosensitizers that offer superior selectivity and reduced systemic toxicity. Patent CN114621290A introduces a groundbreaking class of phosphocorrole compounds characterized by distinct spatial structures, representing a major leap forward in the design of non-metal coordinated corrole derivatives. Unlike traditional porphyrin-based agents which often suffer from slow metabolic clearance and prolonged skin photosensitivity, these novel phosphocorroles leverage the unique electronic properties of phosphorus coordination to enhance singlet oxygen generation while maintaining excellent biocompatibility. The invention details a robust synthetic methodology that allows for the precise installation of functional groups such as hydroxyl, bromo, and methoxy moieties at specific meso-positions, thereby fine-tuning the photophysical properties for optimal therapeutic outcomes. For pharmaceutical developers seeking a reliable pharmaceutical intermediates supplier, this technology offers a pathway to produce high-efficacy PDT agents that address the critical limitations of current clinical candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of corrole-based photosensitizers has been hindered by inherent structural instability and the challenges associated with introducing heavy metal centers for coordination. Traditional free-base corroles possess three inner-ring protons which can lead to rapid degradation under physiological conditions, limiting their utility in long-term therapeutic applications. Furthermore, many existing metallo-corroles rely on expensive and potentially toxic transition metals like gallium or indium, which raise significant safety concerns regarding long-term accumulation in patient tissues. The synthesis of these conventional derivatives often requires harsh reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate purification and increase the environmental footprint of manufacturing. Additionally, achieving high singlet quantum yields with conventional structures often necessitates complex multi-step functionalization processes that result in low overall yields and inconsistent batch-to-batch quality, posing a severe bottleneck for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by utilizing a phosphorus coordination strategy that fundamentally alters the electronic landscape of the corrole macrocycle. By replacing the inner protons with a phosphorus atom coordinated via axial hydroxyl groups, the resulting complexes exhibit markedly improved light stability and enhanced fluorescence intensity in the Q-band region. This approach not only eliminates the need for toxic heavy metals but also leverages the biological compatibility of phosphorus, ensuring that decomposition products are essentially non-toxic to the human body. The synthetic route is elegantly designed to accommodate diverse aldehyde precursors, enabling the creation of a library of compounds with varying spatial arrangements of substituents such as bromine and methoxy groups. This structural diversity allows researchers to systematically optimize the balance between hydrophilicity and cellular uptake, directly addressing the commercial scale-up of complex polymer additives and fine chemicals by providing a versatile platform for derivative synthesis. The ability to tune the position of heavy atoms like bromine relative to hydroxyl groups provides a powerful tool for maximizing the heavy atom effect, thereby boosting intersystem crossing efficiency and singlet oxygen production without compromising safety profiles.
Mechanistic Insights into Phosphorus-Coordinated Corrole Formation
The core of this innovation lies in a sophisticated two-step mechanistic pathway that ensures high fidelity in the formation of the phosphocorrole core. The process initiates with an acid-catalyzed condensation reaction between 5-(pentafluorophenyl)dipyrromethane and various substituted benzaldehydes in a dichloromethane solvent system. Trifluoroacetic acid acts as a potent catalyst to facilitate the formation of the corrolin intermediate, which is subsequently oxidized by 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) to establish the fully conjugated 18-pi electron aromatic system characteristic of corroles. This oxidative cyclization is critical for generating the rigid planar structure required for effective light absorption, and the use of DDQ ensures a clean conversion with minimal side reactions that could otherwise lead to chlorin or porphyrin impurities. The subsequent coordination step involves refluxing the purified corrole intermediate with phosphorus trichloride in pyridine under an inert nitrogen atmosphere, where the phosphorus atom inserts into the corrole cavity to form stable P-N bonds. This coordination is accompanied by the attachment of axial ligands, typically hydroxyl groups derived from subsequent hydrolysis, which further modulate the solubility and electronic properties of the final complex.
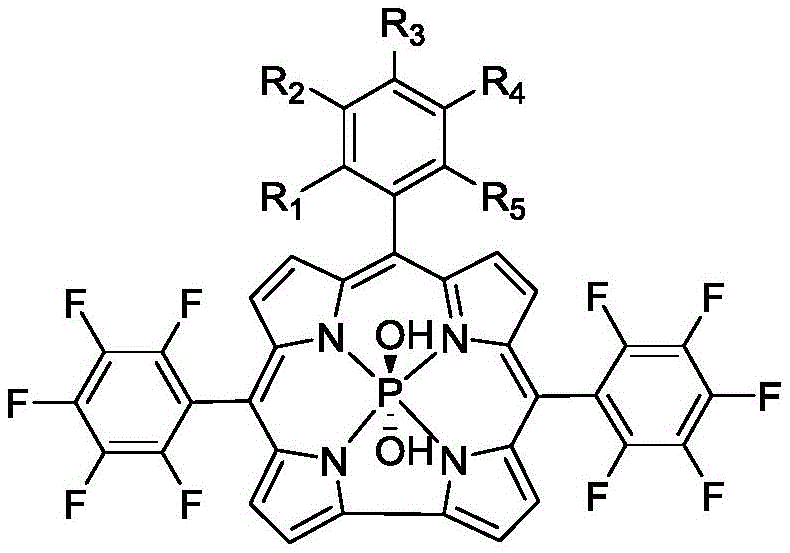
The introduction of specific substituents at the meso-phenyl rings plays a pivotal role in defining the biological activity and physicochemical properties of the final product. As illustrated in the structural diagrams, variations such as the placement of bromine atoms at ortho- or meta-positions relative to hydroxyl groups create distinct electronic environments that influence the rate of intersystem crossing. The heavy atom effect induced by bromine significantly enhances the spin-orbit coupling, facilitating the transition from the excited singlet state to the triplet state, which is the precursor for singlet oxygen generation. Meanwhile, the presence of methoxy groups or hydroxyl groups modulates the hydrophilicity of the molecule, affecting its ability to penetrate cell membranes and localize within specific organelles like mitochondria or lysosomes. Understanding these mechanistic nuances is essential for reducing lead time for high-purity pharmaceutical intermediates, as it allows process chemists to predict and control the outcome of the synthesis with greater precision, minimizing the need for extensive trial-and-error optimization during scale-up.
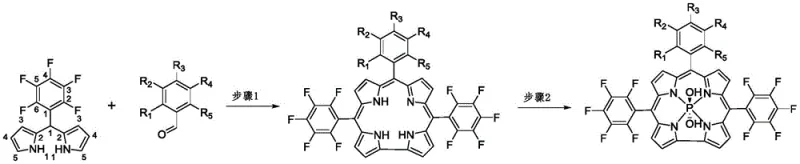
How to Synthesize Phosphocorrole Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for producing these advanced photosensitizers with high purity and consistency. The process is designed to be operationally simple, utilizing standard laboratory equipment and readily available reagents, which makes it highly attractive for industrial adoption. The initial cyclization step is performed at ambient temperatures, reducing energy consumption and simplifying thermal management requirements, while the subsequent phosphorus coordination utilizes a straightforward reflux setup in pyridine. Detailed standard operating procedures for each reaction stage, including precise molar ratios, solvent volumes, and workup techniques, are critical for ensuring that the final product meets the stringent quality standards required for biomedical applications. For a comprehensive guide on executing this synthesis, please refer to the standardized steps below.
- Cyclization and Oxidation: React 5-(pentafluorophenyl)dipyrromethane with substituted benzaldehydes in dichloromethane using trifluoroacetic acid catalysis, followed by oxidation with DDQ.
- Phosphorus Coordination: Reflux the resulting corrole intermediate with phosphorus trichloride in pyridine under nitrogen atmosphere to form the final phosphocorrole complex.
- Purification: Quench the reaction with water, extract with ethyl acetate, and purify via column chromatography to isolate the target spatial isomers.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this phosphocorrole synthesis technology offers substantial strategic benefits that extend beyond mere technical performance. The reliance on phosphorus trichloride as the coordinating agent represents a significant cost advantage over traditional methods that utilize scarce and expensive noble metals or rare earth elements. Phosphorus is an abundant and inexpensive commodity chemical, which translates directly into lower raw material costs and reduced exposure to volatile metal markets. Furthermore, the synthetic route avoids the use of cryogenic conditions or ultra-high pressure reactors, allowing for the utilization of standard glass-lined or stainless steel reaction vessels that are common in most fine chemical manufacturing facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for contract manufacturing organizations and reduces the capital expenditure required for technology transfer.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of cost-effective oxidants like DDQ contribute to a leaner bill of materials. The process operates under mild conditions that minimize energy consumption and reduce the wear and tear on processing equipment, leading to lower maintenance costs and extended asset life. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the burden on downstream purification processes such as chromatography and crystallization. These factors collectively drive down the cost of goods sold, making the final photosensitizer more competitive in the global market while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted benzaldehydes and dipyrromethanes, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent yields across different batches and production sites, providing supply chain managers with the confidence to forecast demand accurately. The absence of sensitive reagents that require special handling or storage conditions further simplifies logistics and inventory management, reducing the likelihood of disruptions due to transportation delays or storage failures. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been demonstrated to work effectively from milligram to gram scales without significant loss of efficiency. The use of common organic solvents like dichloromethane and pyridine allows for established recovery and recycling protocols, aligning with modern green chemistry principles and regulatory expectations for waste minimization. The non-toxic nature of the phosphorus byproducts simplifies effluent treatment and reduces the environmental impact of the manufacturing process. This alignment with sustainability goals not only enhances the corporate social responsibility profile of the manufacturer but also facilitates smoother regulatory approvals in jurisdictions with strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel phosphocorrole compounds. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing product pipelines and assess the potential return on investment.
Q: Why is phosphorus coordination preferred over metal coordination for corrole photosensitizers?
A: Phosphorus is an essential element in the human body, meaning the decomposition products of phosphocorroles exhibit significantly lower toxicity compared to heavy metal complexes. Additionally, P-coordination enhances the fluorescence quantum yield and light stability of the Q-band absorption.
Q: How does the spatial structure of substituents affect photodynamic activity?
A: The position of hydroxyl and bromine groups critically influences singlet oxygen generation. Meta-positioned bromine atoms combined with para-hydroxyl groups have been shown to drastically improve phototoxicity against cancer cells while minimizing dark toxicity.
Q: What are the key scalability advantages of this synthesis route?
A: The process utilizes commercially available raw materials like substituted benzaldehydes and avoids cryogenic conditions. The reaction proceeds at room temperature for cyclization and moderate reflux for coordination, facilitating easy scale-up from laboratory to commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphocorrole Supplier
As the demand for advanced photodynamic therapy agents continues to grow, partnering with an experienced chemical manufacturer is essential for translating laboratory innovations into commercial realities. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of phosphocorrole intermediate meets the highest industry standards for biomedical applications. We understand the critical importance of timeline and quality in the pharmaceutical sector and are committed to delivering solutions that accelerate your drug development programs.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized manufacturing processes can reduce your overall production expenses. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to bring these next-generation photosensitizers from the bench to the bedside, improving patient outcomes through superior chemical engineering.
