Advanced Organocatalytic Synthesis of Optically Active Trifluoromethyl Acrylates for Pharmaceutical Applications
Advanced Organocatalytic Synthesis of Optically Active Trifluoromethyl Acrylates for Pharmaceutical Applications
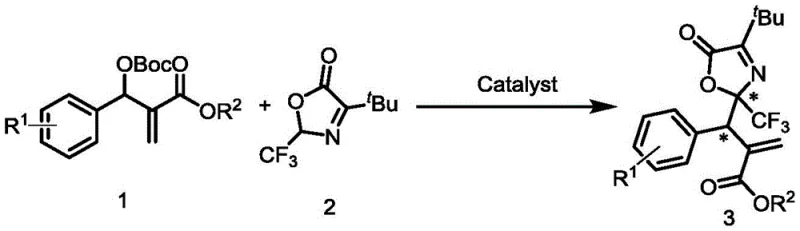
The development of efficient methodologies for constructing chiral trifluoromethyl-containing scaffolds remains a paramount challenge in modern organic synthesis, particularly for the pharmaceutical industry where such motifs often dictate metabolic stability and bioactivity. Patent CN114835655A introduces a groundbreaking approach to synthesizing optically active trifluoromethyl acrylate compounds through an asymmetric allylic alkylation reaction. This innovation utilizes Morita-Baylis-Hillman (MBH) carbonates and oxazolones as primary building blocks, mediated by a novel class of L-tertiary leucine-derived chiral bifunctional tertiary amine urea-phosphine catalysts. The significance of this technology lies in its ability to generate complex chiral architectures with exceptional stereocontrol without relying on scarce transition metals. By leveraging a metal-free organocatalytic system, this process addresses critical pain points in API manufacturing, including heavy metal contamination risks and harsh reaction conditions, positioning it as a superior alternative for producing high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of quaternary stereocenters bearing trifluoromethyl groups has heavily relied on transition metal-catalyzed reactions, such as those employing palladium, rhodium, or copper complexes. While effective in laboratory settings, these conventional methods present substantial hurdles for industrial scale-up. The primary drawback is the inevitable presence of residual heavy metals in the final product, which necessitates rigorous and expensive purification protocols to meet stringent regulatory limits for pharmaceutical ingredients. Furthermore, many of these metal-catalyzed processes require sensitive ligands that are costly to synthesize and unstable under ambient conditions, often demanding cryogenic temperatures or strictly inert atmospheres. These factors collectively inflate the cost of goods sold (COGS) and complicate the supply chain logistics, making the commercial production of such chiral intermediates economically challenging for procurement teams seeking cost-effective solutions.
The Novel Approach
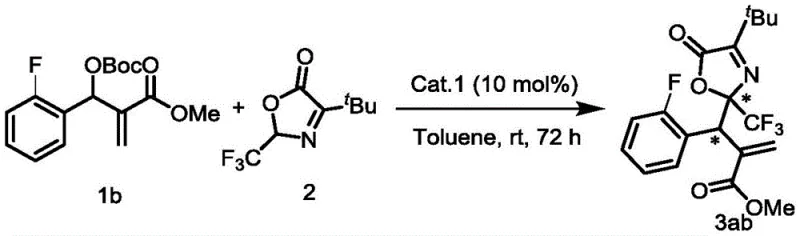
In stark contrast, the methodology disclosed in the patent utilizes a robust organocatalytic system that operates efficiently under mild conditions, typically at room temperature (25°C) and even in the presence of air. This represents a paradigm shift towards greener and more sustainable chemical manufacturing. The use of MBH carbonates as electrophiles and oxazolones as nucleophiles allows for the direct formation of carbon-carbon bonds with high atom economy. The reaction tolerates a wide range of functional groups, including halogens, alkyls, and heterocycles like thiophene, as demonstrated by the successful synthesis of various derivatives such as compound 3ab shown below. This versatility ensures that the process can be adapted for diverse molecular targets without extensive re-optimization, providing R&D directors with a flexible platform for developing new drug candidates while simultaneously offering procurement managers a more predictable and stable supply chain.
Mechanistic Insights into Chiral Bifunctional Organocatalysis
The core of this technological advancement lies in the unique design of the chiral catalysts, specifically the L-tertiary leucine-derived bifunctional tertiary amine urea-phosphine/ester series (labeled C1 through C8). These catalysts operate via a dual-activation mechanism that precisely controls the stereochemical outcome of the reaction. The phosphine moiety acts as a Lewis base to activate the MBH carbonate by forming a zwitterionic intermediate, facilitating the departure of the carbonate leaving group. Simultaneously, the urea or thiourea component engages in hydrogen bonding with the oxazolone nucleophile, enhancing its acidity and orienting it within the chiral pocket of the catalyst. This synergistic activation ensures that the nucleophilic attack occurs from a specific face, resulting in the high enantiomeric excesses observed, often exceeding 90% ee and reaching up to 99% ee with optimized catalysts like C1.
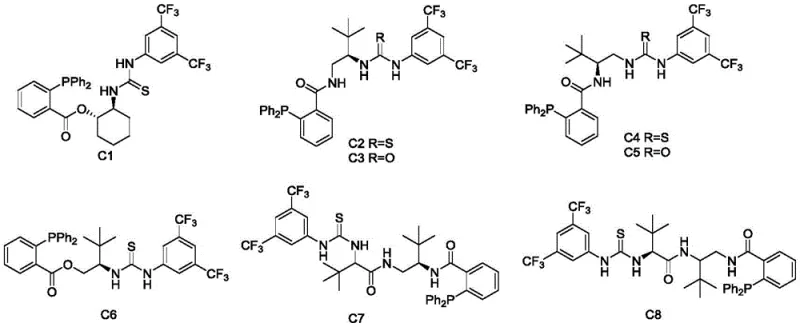
From an impurity control perspective, this mechanism offers distinct advantages over radical or metal-mediated pathways. The absence of single-electron transfer processes minimizes the formation of radical byproducts and homocoupling impurities that are common in transition metal catalysis. Additionally, the catalyst structure itself, as seen in the synthesis of catalyst C1 from 2-aminocyclohexanol, is robust and chemically stable. The reaction proceeds through a well-defined ionic pathway, which simplifies the impurity profile of the crude reaction mixture. This clarity in the impurity spectrum significantly reduces the burden on analytical teams during method development and validation, allowing for faster regulatory filings. The ability to achieve high diastereoselectivity (dr values up to 12:1) further streamlines downstream purification, ensuring that the final API intermediate meets the rigorous purity specifications required by global health authorities.
How to Synthesize Optically Active Trifluoromethyl Acrylates Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The standard protocol involves dissolving the MBH carbonate substrate and the chiral catalyst in anhydrous toluene, followed by the addition of the oxazolone derivative. The mixture is then stirred at ambient temperature for a period typically ranging from 72 hours, depending on the specific substrate reactivity. Reaction progress is easily monitored using thin-layer chromatography (TLC), and upon completion, the workup procedure is remarkably straightforward. The solvent is removed under reduced pressure, and the crude product is purified directly via flash column chromatography using a gradient of petroleum ether and ethyl acetate. This elimination of complex aqueous workups or specialized extraction techniques underscores the process efficiency.
- Dissolve Morita-Baylis-Hillman (MBH) carbonate and the chiral bifunctional tertiary amine urea-phosphine catalyst in ultra-dry toluene under inert or air atmosphere.
- Add 4-tert-butyl-2-trifluoromethyloxazol-5(2H)-one to the reaction mixture and stir at room temperature (25°C) for approximately 72 hours.
- Monitor reaction completion via TLC, remove solvent under reduced pressure, and purify the crude product using flash silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical yield. The most significant advantage is the drastic simplification of the manufacturing process, which directly impacts the bottom line. By eliminating the need for transition metals, manufacturers can bypass the expensive and time-consuming steps associated with metal scavenging and residue testing. This not only reduces the consumption of specialized reagents but also shortens the overall production cycle time. Furthermore, the catalysts themselves are derived from abundant chiral pool materials like amino acids, ensuring a stable and cost-effective supply of the catalytic system, unlike proprietary metal-ligand complexes that may be subject to supply volatility or exorbitant pricing from sole-source vendors.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. Traditional methods often incur hidden costs related to waste disposal of heavy metal contaminants and the energy-intensive processes required for their removal. In this new approach, the reaction conditions are mild (room temperature), which significantly lowers energy consumption compared to processes requiring heating or cooling. The simplicity of the workup, involving direct evaporation and chromatography, reduces solvent usage and labor hours. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate isomers, leading to higher overall recovery rates of the desired product and reducing the raw material input required per kilogram of final output.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials. MBH carbonates and oxazolones are commodity chemicals that can be sourced from multiple suppliers globally, reducing the risk of single-point failures. The robustness of the catalyst, which can be synthesized in high yields from simple precursors like 2-aminocyclohexanol and 3,5-bis(trifluoromethyl)phenyl isothiocyanate, ensures that the key reagent is not a bottleneck. Moreover, the reaction's tolerance to air and moisture variations (within reason) means that it does not require specialized glovebox infrastructure, allowing for production in standard multipurpose reactors, thereby increasing manufacturing flexibility and capacity utilization.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by its inherent safety and environmental profile. The absence of pyrophoric reagents or toxic metal salts aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. Waste streams are primarily organic solvents which are easier to treat and recycle compared to heavy metal sludge. The high atom economy of the allylic alkylation ensures that the majority of the reactant mass is incorporated into the final product, minimizing waste generation. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity, a key metric for modern ESG-conscious investors and partners.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing pipelines, we have compiled answers to common inquiries regarding the process parameters and performance metrics. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and reliability for your process assessment. Understanding these details is crucial for projecting timelines and resource allocation for potential scale-up projects.
Q: What are the advantages of this organocatalytic method over transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metals like palladium or rhodium, thereby removing the costly and complex heavy metal removal steps required in API manufacturing. It also operates under mild conditions (room temperature, air atmosphere), significantly simplifying process control.
Q: What level of enantioselectivity can be achieved with this process?
A: The patented process demonstrates excellent enantioselectivity, achieving up to 99% ee (enantiomeric excess) with specific catalysts like C1. This high optical purity is critical for pharmaceutical intermediates where chirality dictates biological activity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is highly scalable due to the use of readily available starting materials (MBH carbonates and oxazolones), simple catalyst structures, and mild reaction conditions that do not require cryogenic temperatures or strict anhydrous environments beyond standard dry solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Acrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic synthesis route described in patent CN114835655A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale supply. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess required for your pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your budget. We encourage you to contact us to request specific COA data for similar trifluoromethyl-containing scaffolds and to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable supply of high-purity pharmaceutical intermediates while driving down your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
