Advanced Synthesis of Biotin-Conjugated Photosensitizers for Effective Hypoxic Tumor Therapy
Introduction to Next-Generation Photodynamic Therapy Intermediates
The landscape of oncological therapeutics is undergoing a significant transformation with the advent of advanced photodynamic therapy (PDT) agents capable of overcoming the physiological barriers of solid tumors. Patent CN114306624B introduces a groundbreaking methodology for the synthesis of novel biotin-conjugated photosensitizers that effectively address the critical challenge of tumor hypoxia. Unlike conventional photosensitizers that rely heavily on ambient oxygen to generate cytotoxic singlet oxygen, this innovative approach leverages the unique electron-rich properties of D-biotin to induce a Type I photodynamic mechanism. This strategic molecular modification ensures potent therapeutic efficacy even in the oxygen-depleted microenvironments characteristic of aggressive malignancies. For pharmaceutical developers and procurement specialists, this technology represents a pivotal shift towards more reliable and effective cancer treatment modalities, offering a robust platform for the development of high-purity therapeutic intermediates.
The technical significance of this patent extends beyond mere efficacy; it provides a chemically feasible and scalable route for modifying existing photosensitizer scaffolds. By covalently linking a tumor-targeting biotin unit to the molecular structure of terminal carboxyl-bearing photosensitizers, the invention achieves a dual function of enhanced cellular uptake and improved reactive oxygen species (ROS) generation profiles. This dual-mechanism action not only solves the problem of high oxygen dependence but also maintains the favorable photophysical properties of the parent compounds. As a reliable photosensitizer supplier, understanding the nuances of this synthetic pathway is essential for evaluating the commercial viability and supply chain stability of these next-generation medical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photodynamic therapy has long been constrained by its reliance on Type II mechanisms, which necessitate the presence of molecular oxygen to generate singlet oxygen (1O2) for tumor ablation. This dependency creates a fundamental paradox in cancer treatment, as rapidly growing tumors often outpace their blood supply, resulting in hypoxic regions that are inherently resistant to oxygen-dependent therapies. Consequently, conventional photosensitizers frequently exhibit diminished therapeutic outcomes in these critical hypoxic zones, leading to incomplete tumor eradication and higher rates of recurrence. Furthermore, the lack of intrinsic tumor targeting in many standard photosensitizers results in non-specific distribution, increasing the risk of off-target toxicity and prolonged photosensitivity in healthy tissues. These limitations underscore the urgent need for chemical innovations that can decouple therapeutic efficacy from oxygen availability while enhancing tumor specificity.
The Novel Approach
The methodology disclosed in CN114306624B circumvents these physiological barriers through a clever bioconjugation strategy that integrates D-biotin directly into the photosensitizer framework. This novel approach capitalizes on the overexpression of biotin receptors on the surface of many tumor cells, thereby facilitating receptor-mediated endocytosis and selective accumulation within the malignant tissue. More critically, the electron-donating nature of the biotin moiety alters the electronic state of the photosensitizer, promoting a Type I electron transfer pathway that generates superoxide anion radicals (O2-·) independent of high oxygen concentrations. This mechanism ensures that the therapeutic agent remains potent even under severe hypoxic conditions, effectively turning the tumor's own defensive microenvironment against itself. The result is a versatile class of compounds that combines targeted delivery with oxygen-independent cytotoxicity, representing a substantial advancement in the field of oncological intermediates.
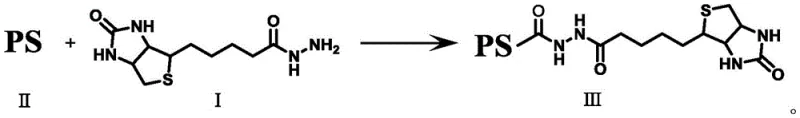
Mechanistic Insights into Amide Coupling and Electronic Modulation
The core of this synthetic innovation lies in the efficient formation of an amide bond between the hydrazide group of the biotin derivative and the carboxylic acid functionality of the photosensitizer. This condensation reaction is typically mediated by carbodiimide chemistry, utilizing reagents such as EDCI (1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide) and HOBt (Hydroxybenzotriazole) to activate the carboxyl group without racemization or side reactions. The reaction proceeds through the formation of an active ester intermediate, which is then nucleophilically attacked by the hydrazine nitrogen of the biotin linker. This mild coupling protocol is advantageous for preserving the delicate conjugated systems of complex photosensitizers like porphyrins or fluorophores, which might otherwise degrade under harsher acylation conditions. The use of anhydrous DMF as a solvent ensures optimal solubility for both the hydrophobic photosensitizer and the polar biotin intermediate, driving the reaction to completion with high conversion rates.
Beyond the synthetic mechanics, the electronic modulation imparted by the biotin unit is the key driver of the enhanced biological activity. Cyclic voltammetry studies indicate that the biotinylated conjugates exhibit a lower reduction potential compared to their non-conjugated counterparts. This anodic shift in reduction potential thermodynamically favors the acceptance of electrons from surrounding substrates upon photoexcitation, thereby facilitating the Type I electron transfer process. Theoretical calculations suggest that the folded conformation of the biotinylated molecule allows for effective intramolecular electronic communication between the biotin moiety and the photosensitizer core. This structural arrangement supports the rapid generation of superoxide radicals, which can subsequently undergo dismutation to produce hydrogen peroxide and hydroxyl radicals, creating a cascade of cytotoxic species that overwhelm the tumor cell's antioxidant defenses regardless of oxygen tension.
How to Synthesize Biotin-Conjugated Photosensitizers Efficiently
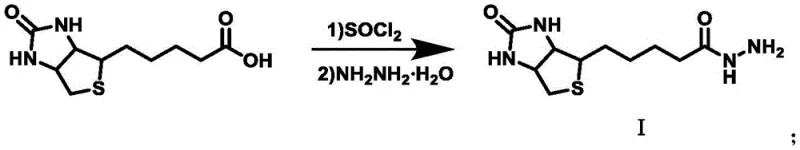
The preparation of these advanced therapeutic intermediates follows a streamlined two-step sequence that begins with the activation of commercially available D-biotin. In the initial phase, D-biotin is converted into a reactive hydrazide derivative (Compound I) through a chlorination-hydrazinolysis sequence, yielding a stable white powder that serves as the universal linker for subsequent conjugations. This intermediate is then coupled with a variety of terminal carboxyl-bearing photosensitizers under inert atmosphere conditions to prevent oxidation of sensitive functional groups. The detailed standardized synthesis steps outlined below provide a robust framework for laboratory-scale preparation and can be readily adapted for larger batch production with appropriate process engineering controls.
- Convert D-biotin to biotin hydrazide (Compound I) using thionyl chloride and hydrazine hydrate in methanol.
- Activate the carboxyl-terminal photosensitizer (Compound II) using EDCI, HOBt, and DIEA in anhydrous DMF at 0°C.
- Couple Compound I with the activated photosensitizer at room temperature to form the final biotinylated product (Compound III).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in this patent offers distinct advantages over traditional methods that rely on complex multi-step syntheses or scarce raw materials. The primary starting material, D-biotin, is a commodity chemical produced on a massive global scale for the nutrition and pharmaceutical industries, ensuring a stable and cost-effective supply chain foundation. The coupling reagents employed, such as EDCI and HOBt, are standard inventory items in any fine chemical manufacturing facility, eliminating the need for specialized or exotic catalysts that often introduce supply bottlenecks. This reliance on ubiquitous reagents significantly de-risks the procurement process, allowing for consistent sourcing and reducing the vulnerability to market fluctuations that plague specialty chemical sectors. Furthermore, the simplicity of the workup procedure, which involves standard distillation and chromatography, facilitates easier purification and quality control.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, which are often required for cross-coupling reactions in other photosensitizer syntheses, leads to substantial cost savings in raw material expenditure. Additionally, the mild reaction conditions (0°C to room temperature) reduce energy consumption associated with heating or cryogenic cooling, further lowering the operational overhead of the manufacturing process. The high atom economy of the amide coupling reaction minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. These factors collectively contribute to a more economically viable production model for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing a modular synthesis strategy where a single biotin hydrazide intermediate can be coupled with diverse photosensitizer cores, manufacturers can maintain a flexible inventory system that responds quickly to changing demand. This modularity reduces the lead time for high-purity photosensitizer derivatives, as the rate-limiting step becomes the final coupling rather than the synthesis of the entire molecule from scratch. The robustness of the chemical process ensures high batch-to-batch consistency, which is critical for maintaining regulatory compliance and avoiding costly production delays due to out-of-specification results. This reliability is paramount for securing long-term contracts with major pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively with various substrates ranging from small molecule fluorophores to larger porphyrin structures. The absence of heavy metals simplifies the purification process, as there is no need for rigorous metal scavenging steps to meet strict residual metal limits in API intermediates. This simplification not only speeds up production cycles but also aligns with green chemistry principles by reducing the use of hazardous reagents and solvents. The ability to scale from gram quantities to multi-kilogram batches without significant process re-engineering makes this technology ideal for commercial scale-up of complex photosensitizers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this biotinylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating these intermediates into their drug development pipelines.
Q: How does biotinylation improve photosensitizer efficacy in hypoxic tumors?
A: The covalent attachment of the electron-rich biotin unit lowers the reduction potential of the photosensitizer, facilitating a Type I electron transfer mechanism that generates superoxide anions (O2-·) even in low-oxygen environments, overcoming the limitations of traditional Type II PDT.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes standard amide coupling reagents (EDCI/HOBt) and mild reaction conditions (0°C to room temperature) without requiring expensive transition metal catalysts, making it highly suitable for commercial scale-up from kilograms to metric tons.
Q: What types of photosensitizers are compatible with this conjugation strategy?
A: The method demonstrates broad universality and has been successfully applied to various carboxyl-terminal photosensitizers including delayed fluorescence agents like DCF-TFM and FL, as well as clinical protoporphyrins like PpIX.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin-Photosensitizer Conjugate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN114306624B for the next generation of oncology therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to clinical supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch of biotin-conjugated photosensitizer meets the highest standards of quality and consistency. We understand the critical nature of supply continuity in the pharmaceutical industry and are committed to being a dependable partner in your supply chain.
We invite you to collaborate with us to optimize your production costs and accelerate your time to market. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your project. By leveraging our expertise in amide coupling and bioconjugation, we can help you navigate the complexities of manufacturing these sophisticated molecules, ensuring a reliable supply of high-quality intermediates for your photodynamic therapy applications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
