Revolutionizing Agrochemical Intermediates: High-Yield Synthesis of 2-Alkylthio Benzoic Acid Derivatives
Revolutionizing Agrochemical Intermediates: High-Yield Synthesis of 2-Alkylthio Benzoic Acid Derivatives
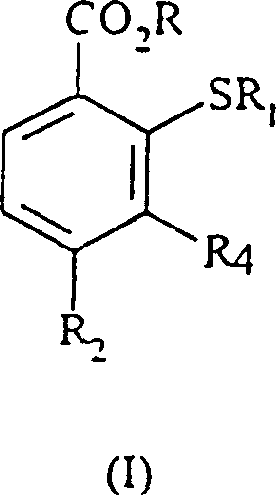
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways that balance high purity with economic viability, particularly for complex intermediates used in herbicide manufacturing. Patent CN1176071C introduces a transformative methodology for the preparation of 2-alkylthio-substituted benzoic acid derivatives, specifically addressing the longstanding challenges associated with nucleophilic aromatic substitution on sterically hindered benzoic acid scaffolds. This innovation is pivotal for the production of next-generation herbicidally active compounds, offering a route that circumvents the severe limitations of prior art techniques such as the use of unstable organolithium reagents. By leveraging a carefully controlled anhydrous environment and specific polar aprotic solvents, this process achieves exceptional conversion rates, often reaching quantitative yields, which is a rare feat in fine chemical synthesis. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkylthio benzoic acids has been plagued by significant technical hurdles that impede efficient commercial scale-up of complex agrochemical intermediates. Traditional approaches, such as those disclosed in European Patent Publication No. 527036, rely heavily on the use of organolithium reagents at extremely low temperatures, creating a hazardous operational environment that demands specialized cryogenic equipment and rigorous safety protocols. Furthermore, alternative nucleophilic substitution methods described in older literature often suffer from sluggish reaction kinetics, requiring prolonged heating periods of up to three weeks to achieve modest conversions, as noted in US Patent No. 3,714,179. These inefficiencies not only drive up energy consumption and operational costs but also introduce variability in the impurity profile, complicating downstream purification processes. The reliance on activated substrates, such as those with strong electron-withdrawing nitro groups at the 5-position, further limits the scope of applicable starting materials, restricting the versatility required for diverse herbicide scaffold development.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process defined in CN1176071C utilizes a direct nucleophilic displacement of a 2-halo or 2-nitro group on a benzoic acid derivative using an alkyl thiol or its corresponding salt. This novel approach eliminates the need for cryogenic conditions, operating instead within a moderate temperature range of -20°C to 150°C, with preferred embodiments functioning efficiently between 50°C and 100°C. The strategic selection of polar aprotic solvents, particularly N-methylpyrrolidone (NMP) or acetonitrile, facilitates the solubility of both the benzoic acid salt and the thiolate nucleophile, thereby enhancing reaction kinetics without the need for exotic catalysts. Most critically, the invention identifies water content as a decisive factor, demonstrating that maintaining anhydrous conditions with less than 5% volume of water, and ideally below 1%, prevents hydrolysis side reactions and ensures yields approaching 100%. This paradigm shift allows for cost reduction in herbicide manufacturing by simplifying the reactor setup and drastically reducing batch cycle times from weeks to mere hours.
Mechanistic Insights into Anhydrous Nucleophilic Aromatic Substitution
The core of this technological advancement lies in the precise manipulation of reaction thermodynamics and kinetics through moisture control. In nucleophilic aromatic substitution (SnAr) reactions involving benzoic acid derivatives, the presence of water can act as a competing nucleophile or lead to the protonation of the thiolate anion, effectively quenching the reactive species before it can attack the aromatic ring. The patent data explicitly illustrates that increasing water concentration from 0.01% to 20% causes a catastrophic drop in yield from 100% to merely 12%, highlighting the extreme sensitivity of the thiolate anion to protic environments. By rigorously excluding water, the reaction equilibrium is driven overwhelmingly towards the formation of the carbon-sulfur bond, leveraging the electron-withdrawing nature of substituents like the trifluoromethyl group at the 4-position to activate the 2-position for attack. This mechanistic understanding allows chemists to predict and control the formation of by-products, ensuring that the final API intermediate possesses the high purity required for regulatory compliance in agrochemical formulations.
Furthermore, the choice of the leaving group at the 2-position plays a pivotal role in the reaction pathway, with fluorine and chlorine atoms being particularly effective due to their ability to stabilize the Meisenheimer complex intermediate. The process accommodates a wide variety of substituents at the 4-position, including haloalkyl, alkoxy, and heterocyclic groups, providing a versatile platform for generating diverse libraries of bioactive molecules. The use of alkali metal salts of the benzoic acid, such as the sodium salt, enhances the solubility in polar aprotic media and increases the nucleophilicity of the carboxylate group, which can assist in stabilizing the transition state. For R&D teams focused on impurity control, this mechanism offers a clear advantage: the absence of organometallic reagents means there is no risk of heavy metal contamination, thereby simplifying the purification workflow and reducing the burden on quality control laboratories tasked with detecting trace metallic residues in the final product.
How to Synthesize 2-Methylthio-4-trifluoromethylbenzoic Acid Efficiently
The practical implementation of this synthesis route is straightforward yet demands strict adherence to the specified anhydrous parameters to replicate the high yields reported in the patent examples. The general procedure involves combining a 2-halo-4-trifluoromethylbenzoic acid salt with anhydrous sodium thiomethoxide in a dry polar aprotic solvent such as NMP. The mixture is then heated to approximately 90°C for a short duration, typically around two hours, after which standard workup procedures involving acidification and organic extraction are employed to isolate the target molecule. This streamlined protocol stands in sharp contrast to multi-step sequences that require protection and deprotection strategies, offering a direct path to the desired intermediate. For detailed operational parameters, stoichiometry, and specific safety guidelines regarding the handling of thiolates, please refer to the standardized synthesis steps outlined below.
- Prepare a reaction mixture containing a 2-halo or 2-nitro substituted benzoic acid derivative (Formula II) and an alkyl thiol or its salt (Formula III) in a polar aprotic solvent.
- Maintain strictly anhydrous conditions with water content below 5% by volume, preferably below 1%, to prevent yield degradation.
- Heat the mixture to a temperature between 50°C and 100°C depending on the substrate, followed by acidification and extraction to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this anhydrous substitution process offers profound benefits for supply chain stability and overall manufacturing economics. By eliminating the dependency on cryogenic cooling systems and hazardous organolithium reagents, facilities can significantly reduce their capital expenditure on specialized infrastructure and lower their operational risk profile. The ability to run reactions at elevated temperatures in common industrial solvents like NMP or acetonitrile means that existing reactor trains can be utilized without major retrofitting, accelerating the time-to-market for new herbicide products. Moreover, the high selectivity of the reaction minimizes the generation of difficult-to-remove impurities, which translates to higher throughput in purification units and reduced solvent consumption during recrystallization or chromatography steps. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands for high-purity agrochemical intermediates without compromising on delivery schedules.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as organolithiums removes the need for costly inert atmosphere handling and cryogenic maintenance, leading to substantial operational savings. Additionally, the high reaction yields observed under anhydrous conditions mean that less raw material is wasted, optimizing the atom economy of the process and lowering the cost per kilogram of the final intermediate. The use of cost-effective solvents like acetonitrile further drives down the variable costs associated with large-scale production, making the final product more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of this chemical transformation ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream formulators. Since the starting materials, such as 2-chloro-4-trifluoromethylbenzoic acid and sodium thiomethoxide, are commercially available commodity chemicals, the risk of raw material shortages is minimized. This accessibility, combined with the process tolerance for a range of reaction temperatures, provides supply chain managers with the flexibility to adjust production schedules dynamically in response to market signals without fearing process failure.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic hazards typically associated with organometallic additions, allowing for safer operation in larger reactor volumes. The simplified workup procedure reduces the volume of aqueous waste streams generated, aligning with increasingly stringent environmental regulations regarding effluent discharge. Furthermore, the high purity of the crude product reduces the need for extensive chromatographic purification, thereby lowering the overall solvent footprint and supporting sustainability goals within the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for process engineers and technical buyers who are evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the specific conditions and outcomes observed during the development of this method, ensuring that expectations regarding yield and purity are grounded in empirical evidence rather than theoretical projections.
Q: Why is strict water control critical in this synthesis?
A: Experimental data indicates that water concentration directly inversely correlates with product yield. Maintaining water content below 1% volume is essential to achieve yields exceeding 95%, whereas higher water levels can drop yields significantly.
Q: What solvents are recommended for large-scale production?
A: While various polar aprotic solvents like DMF and DMSO are effective, N-methylpyrrolidone (NMP) and acetonitrile are preferred for large-scale manufacturing due to their optimal balance of reaction efficiency and cost-effectiveness.
Q: How does this method compare to organolithium routes?
A: Unlike conventional methods requiring hazardous organolithium reagents at cryogenic temperatures, this process operates at moderate temperatures (50-100°C) using stable salts, offering superior safety and scalability for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkylthio Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of anhydrous processing are managed with precision at every stage. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 2-alkylthio benzoic acid derivative meets the exacting standards required for agrochemical applications. Our facility is designed to handle sensitive chemistries safely, providing a secure environment for the manufacture of high-value intermediates that serve as the backbone of modern crop protection solutions.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific product needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the potential economic benefits of switching to this anhydrous process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timeline remains on track with a reliable and efficient supply of critical intermediates.
