Optimized Industrial Synthesis of SN158: High-Purity Antidiabetic Intermediate Manufacturing
Introduction to Advanced SN158 Manufacturing Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for potent antidiabetic agents, particularly those targeting PPAR α/γ receptors. Patent CN110937987B introduces a groundbreaking synthetic methodology for producing SN158, a dual partial agonist with significant potential in managing type 2 diabetes and metabolic disorders. This technical disclosure outlines a refined chemical pathway that addresses critical bottlenecks in prior art, specifically focusing on yield optimization and purification simplicity. By leveraging a strategic sequence of bromination, selective protection, methylation, and Lewis acid-catalyzed condensation, the inventors have established a process capable of delivering the target molecule with exceptional purity. The significance of this development lies not merely in the chemical transformation itself but in its direct applicability to commercial manufacturing environments where efficiency and cost control are paramount. This report analyzes the technical merits of this route, providing R&D and procurement leaders with a clear understanding of its value proposition in the competitive landscape of antidiabetic drug intermediates.
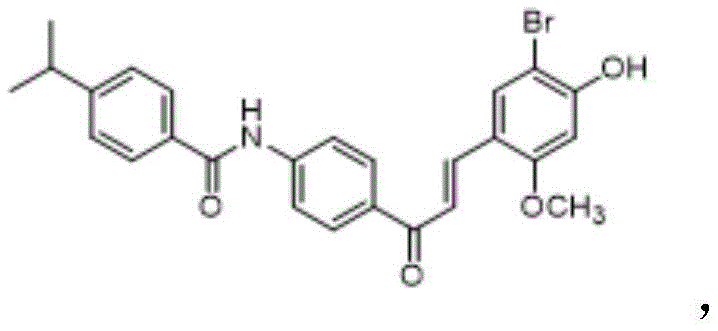
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex chalcone derivatives like SN158 has been plagued by inefficiencies that hinder large-scale production. Traditional methodologies often rely heavily on column chromatography for purification, a technique that is notoriously difficult to scale beyond the laboratory bench. Column chromatography consumes vast quantities of silica gel and organic solvents, creating substantial waste streams and driving up operational expenditures significantly. Furthermore, the reliance on such separation techniques often results in variable recovery rates, leading to inconsistent batch-to-batch yields that complicate supply chain planning. In the context of SN158, prior art routes suffered from low overall synthesis yields and required tedious purification steps to remove impurities generated during the Claisen-Schmidt condensation. These technical defects render conventional methods economically unviable for the tonnage-scale production required to meet global demand for next-generation antidiabetic therapies.
The Novel Approach
The innovative route disclosed in the patent fundamentally reengineers the synthesis strategy to bypass these traditional pitfalls. By introducing a specific sequence of protective group manipulations—specifically the use of methoxymethyl (MOM) groups—the process achieves high regioselectivity during the functionalization of the benzaldehyde core. This precision minimizes the formation of side products, thereby reducing the burden on downstream purification. Most critically, the final product is isolated via simple recrystallization rather than chromatography, achieving a purity level exceeding 99%. This shift from chromatographic purification to crystallization is a hallmark of process chemistry maturity, indicating a route that is inherently stable and reproducible. The ability to obtain high-purity SN158 directly from reaction mixtures through filtration and washing represents a paradigm shift in manufacturing efficiency, drastically reducing solvent consumption and processing time while maximizing material throughput.
Mechanistic Insights into Lewis Acid-Catalyzed Claisen-Schmidt Condensation
The cornerstone of this synthetic success is the optimized Claisen-Schmidt condensation step, which links the functionalized benzaldehyde intermediate with the acetophenone derivative. In this novel protocol, the reaction is catalyzed by Lewis acids such as boron trifluoride diethyl etherate (BF3·OEt2) or hydrochloric acid in ethanol (HCl-EtOH). The mechanism involves the activation of the carbonyl group of the aldehyde by the Lewis acid, increasing its electrophilicity and facilitating nucleophilic attack by the enolizable ketone. This activation lowers the energy barrier for the condensation, allowing the reaction to proceed under milder conditions compared to traditional strong base catalysis. The use of acidic conditions is particularly advantageous here as it prevents the degradation of sensitive functional groups, such as the phenolic hydroxyl and the amide linkage, which might be susceptible to hydrolysis or oxidation under harsh alkaline environments. This mechanistic finesse ensures that the reaction proceeds with high chemoselectivity, favoring the formation of the desired (E)-isomer while suppressing polymerization or self-condensation side reactions.
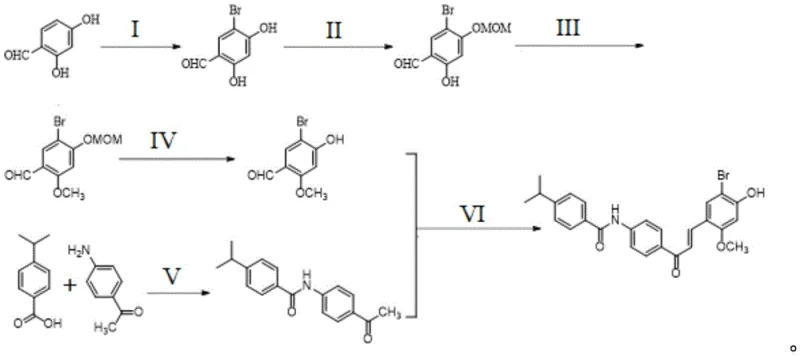
Impurity control is rigorously managed through the strategic design of the precursor synthesis. The initial bromination of 2,4-dihydroxybenzaldehyde is controlled to prevent poly-bromination, ensuring the integrity of the aromatic ring for subsequent substitutions. The protection of the para-hydroxyl group with a MOM ether serves a dual purpose: it directs the subsequent methylation exclusively to the ortho-position and protects the phenol from unwanted side reactions during the amidation and condensation steps. The final deprotection step is seamlessly integrated or managed such that the free phenol is revealed only when necessary, or the stability of the intermediate allows for direct conversion. By maintaining strict control over stoichiometry and reaction temperatures—such as keeping the condensation at 0°C initially—the process minimizes thermal degradation. The result is a crude product profile that is clean enough to allow for direct recrystallization, effectively locking out impurities within the mother liquor and yielding a crystalline solid that meets stringent pharmaceutical specifications without further chromatographic intervention.
How to Synthesize SN158 Efficiently
The synthesis of SN158 is executed through a logical six-step sequence that balances chemical complexity with operational simplicity. The process begins with the regioselective bromination of the starting material, followed by orthogonal protection strategies to differentiate the hydroxyl groups. Subsequent methylation and deprotection steps prepare the aldehyde fragment for the crucial coupling reaction. The amide fragment is synthesized separately via a high-efficiency coupling reaction using agents like HATU or EDCI, ensuring high purity before the final assembly. The convergence of these two fragments via the optimized Claisen-Schmidt condensation completes the carbon skeleton. For detailed operational parameters, including specific solvent ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent documentation.
- Brominate 2,4-dihydroxybenzaldehyde followed by selective protection of the para-hydroxyl group using chloromethyl methyl ether.
- Perform ortho-hydroxyl methylation using dimethyl sulfate or methyl iodide, then deprotect the para-position to reveal the phenol.
- Couple the resulting aldehyde with N-phenyl-4-isopropylbenzamide via Claisen-Schmidt condensation using HCl-EtOH or BF3·OEt2 catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies for antidiabetic intermediates. The elimination of column chromatography is not merely a technical improvement; it is a major economic driver. Chromatography is a resource-intensive operation requiring expensive stationary phases and large volumes of high-purity solvents, both of which contribute significantly to the cost of goods sold (COGS). By replacing this with recrystallization, the manufacturing process becomes significantly more lean, reducing waste disposal costs and solvent recovery burdens. This streamlined approach translates directly into a more competitive pricing structure for the final API intermediate, allowing pharmaceutical companies to manage their drug development budgets more effectively while maintaining high quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the drastic simplification of the purification workflow. Removing the need for silica gel columns eliminates a major variable cost and reduces the labor hours associated with packing, running, and analyzing columns. Furthermore, the high yields reported in the patent, particularly the 97% yield in the final condensation step and near-quantitative yields in intermediate steps like methylation, ensure that raw material utilization is maximized. This high atom economy means less starting material is wasted, directly lowering the input cost per kilogram of SN158 produced. The cumulative effect of these efficiencies is a substantial reduction in manufacturing overhead, making the commercial production of this potent agonist financially sustainable.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations described. The reagents used, such as dimethyl sulfate, chloromethyl methyl ether, and common Lewis acids, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The process does not rely on exotic catalysts or sensitive organometallic reagents that might have long lead times or unstable supply chains. Additionally, the ability to purify via recrystallization implies that the process is less sensitive to minor fluctuations in reaction conditions, leading to more consistent batch outcomes. This reliability ensures that procurement teams can secure long-term supply agreements with confidence, knowing that the manufacturer can consistently deliver high-quality material without unexpected production delays.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this synthetic route, which avoids unit operations that are difficult to translate from the lab to the plant. Recrystallization is a standard, easily scalable unit operation in the fine chemical industry, unlike chromatography which requires specialized equipment and significant floor space for large batches. Moreover, the reduction in solvent usage and the elimination of silica gel waste align with modern green chemistry principles and environmental regulations. This facilitates easier permitting and compliance with increasingly strict environmental, health, and safety (EHS) standards. Manufacturers adopting this route can market their production capabilities as environmentally responsible, a growing requirement for multinational pharmaceutical partners who are auditing their supply chains for sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of SN158. These answers are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these specifics helps in evaluating the feasibility of integrating this intermediate into broader drug development pipelines.
Q: What is the primary advantage of the new SN158 synthesis route over conventional methods?
A: The novel route eliminates the need for column chromatography purification, relying instead on recrystallization to achieve over 99% purity, which significantly reduces production costs and simplifies scale-up.
Q: What yields can be expected from the optimized SN158 synthetic process?
A: The optimized process demonstrates robust yields across multiple steps, with the final Claisen-Schmidt condensation step achieving yields up to 97% and an overall route yield exceeding 60%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial scalability by avoiding complex purification techniques like column chromatography and utilizing standard reagents and recrystallization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SN158 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antidiabetic therapeutics. Our team of expert process chemists has thoroughly analyzed the synthetic route disclosed in CN110937987B and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to full-scale market launch. Our facilities are equipped with state-of-the-art reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of SN158 we deliver meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how we can support your specific supply chain needs. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need to validate the feasibility of this route for your specific application, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your project timeline. Let us be your partner in bringing this promising antidiabetic candidate to the patients who need it most, leveraging our manufacturing excellence to drive your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
