Advanced Refining Technology for Bendazac Lysine: Ensuring Ultra-High Purity for Global Ophthalmic Supply Chains
Advanced Refining Technology for Bendazac Lysine: Ensuring Ultra-High Purity for Global Ophthalmic Supply Chains
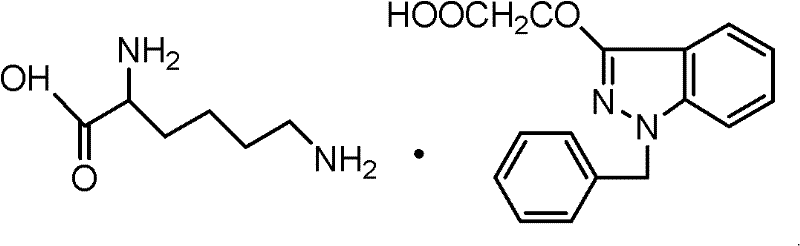
The global pharmaceutical landscape for ophthalmic treatments demands exceptionally high purity standards, particularly for active ingredients targeting chronic conditions like cataracts. Patent CN102206185A introduces a groundbreaking refinement protocol for Bendazac Lysine (BDZL) and its structural analogs, addressing critical quality bottlenecks that have long plagued manufacturers. This technology leverages a sophisticated dual-solvent crystallization strategy, utilizing a polar aprotic solvent system for initial salt formation followed by a polar protic solvent system for final recrystallization. By strictly controlling solvent polarity and concentration, this method effectively eliminates stubborn impurities such as 3-hydroxy-1-benzyl indazole, which are notoriously difficult to remove using traditional single-solvent techniques. For R&D directors and procurement specialists seeking a reliable ophthalmic intermediate supplier, understanding the mechanistic advantages of this process is crucial for securing a supply chain that meets stringent pharmacopoeia requirements without compromising on yield or cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
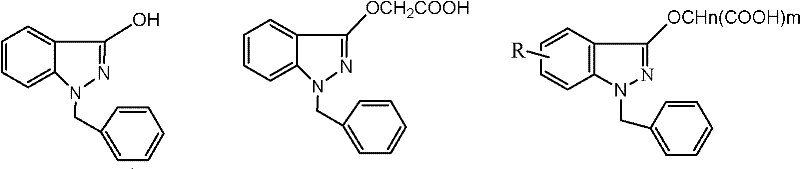
Historically, the production of Bendazac Lysine has been hindered by the persistence of specific synthetic byproducts that degrade the therapeutic efficacy and safety profile of the final drug. Conventional purification methods typically rely on forming salts and recrystallizing within a single solvent system, often using aqueous ethanol throughout the entire process. Experimental data indicates that when polar protic solvents like methanol or ethanol are used exclusively for the salt formation step, the resulting crude product exhibits low melting points and unacceptably high levels of related substances, often ranging between 1% and 5%. The primary culprit is 3-hydroxy-1-benzyl indazole, an intermediate from the Bendazac Acid synthesis that co-precipitates with the desired lysine salt due to insufficient solubility differentiation in protic environments. Furthermore, attempting to purify this contaminated crude through repeated recrystallization in the same solvent system yields diminishing returns, failing to reduce impurity levels below the critical 0.2% threshold mandated by modern quality standards, thereby rendering large batches unsuitable for clinical use.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally alters the solvation environment to exploit the differential solubility profiles of the target molecule versus its impurities. Instead of a uniform solvent system, the process initiates salt formation in a polar aprotic solvent, specifically aqueous acetonitrile with a concentration of 90-95% (v/v). In this environment, the hydrophobic 3-hydroxy-1-benzyl indazole impurity remains highly soluble and is retained in the mother liquor during the precipitation of the Bendazac Lysine salt. Following the isolation of this high-purity crude salt, the process transitions to a polar protic solvent, specifically aqueous ethanol at 91-94% (v/v), for the final recrystallization. This second step is not merely for further purification but is critical for controlling the crystal lattice formation, ensuring the product achieves the sharp melting point range of 179-182°C and the characteristic white crystalline powder appearance required for regulatory approval. This sequential solvent switch represents a significant leap in process chemistry, enabling yields exceeding 80% while maintaining assay values between 99.6% and 101%.
Mechanistic Insights into Solvent Polarity and Impurity Rejection
The success of this refining protocol lies in the precise manipulation of intermolecular forces through solvent selection. During the initial salt formation, the use of acetonitrile, a polar aprotic solvent, creates a medium where the ionic interaction between Bendazac Acid and L-Lysine is strong enough to precipitate the salt, yet the solvent's specific dielectric properties prevent the co-precipitation of the neutral, less polar indazole derivative. The patent data demonstrates that deviating from this aprotic environment, such as using methanol or ethanol for salt formation, results in a crude product where the impurity is physically trapped within the crystal matrix or adsorbed onto the surface, leading to "related substance" levels that are significantly elevated. Conversely, the subsequent switch to ethanol, a polar protic solvent, facilitates hydrogen bonding interactions that stabilize the crystal growth of the lysine salt in a thermodynamically favorable conformation. This step effectively washes away residual pigment impurities and trace solvents that might have been occluded during the rapid precipitation in acetonitrile, ensuring the final product is not only chemically pure but also possesses the correct physical properties for downstream tableting or formulation into eye drops.
Furthermore, the concentration of the solvents plays a pivotal role in the thermodynamic equilibrium of the crystallization process. The patent specifies a narrow operational window for acetonitrile concentration between 90% and 95% (v/v); concentrations below 90% fail to keep the indazole impurity in solution, causing it to crash out with the product, while concentrations above 95% require excessive solvent volumes to dissolve the reactants, negatively impacting process economics and throughput. Similarly, the ethanol concentration for recrystallization is optimized at 91-94% (v/v) to balance solubility and yield. Lower ethanol concentrations introduce pigment impurities into the final crystal lattice, whereas higher concentrations necessitate larger volumes to achieve dissolution, reducing the overall volumetric productivity of the reactor. This fine-tuned balance ensures that the process is robust against minor fluctuations in raw material quality, making it an ideal candidate for commercial scale-up where consistency is paramount.
How to Synthesize Bendazac Lysine Efficiently
The implementation of this refining process requires strict adherence to the specified solvent ratios and thermal profiles to guarantee the removal of the 3-hydroxy-1-benzyl indazole impurity. The procedure begins with the reaction of Bendazac Acid and L-Lysine in the optimized acetonitrile-water mixture under reflux, followed by hot filtration to remove any insoluble mechanical impurities before cooling induces crystallization. The resulting crude solid is then subjected to the secondary recrystallization in the ethanol-water system, where controlled cooling rates allow for the growth of large, pure crystals.
- React Bendazac Acid with L-Lysine in 90-95% aqueous acetonitrile under reflux to form the crude salt, ensuring hydrophobic impurities remain in solution.
- Perform hot filtration to remove insoluble particulates, then cool the filtrate to room temperature to precipitate the crude Bendazac Lysine salt.
- Recrystallize the dried crude product in 91-94% aqueous ethanol under reflux, followed by slow cooling to obtain white crystalline powder with >99.6% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dual-solvent refining technology offers substantial strategic advantages beyond mere technical compliance. By effectively eliminating the need for multiple, iterative recrystallization cycles that characterize older methods, the process drastically simplifies the manufacturing workflow, leading to significant reductions in processing time and labor costs. The ability to consistently produce Bendazac Lysine with impurity levels below 0.2% directly from the first major purification step minimizes the risk of batch rejection, thereby enhancing overall supply chain reliability and reducing the inventory buffer needed to account for quality failures. Moreover, the process is designed to handle not only freshly synthesized material but also sub-standard commercial raw materials, providing a valuable remediation pathway that can salvage off-spec inventory and convert it into high-value, pharmacopoeia-compliant product, thus optimizing asset utilization and reducing waste disposal costs associated with failed batches.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reliance on common, recoverable solvents like acetonitrile and ethanol significantly lowers the raw material cost base. By avoiding the need for expensive chromatographic purification or complex extraction protocols to remove the indazole impurity, the process streamlines the cost structure, allowing for more competitive pricing in the global ophthalmic intermediate market while maintaining healthy margins through high yield recovery rates exceeding 80%.
- Enhanced Supply Chain Reliability: The robustness of the solvent system against variations in crude quality ensures a steady output of high-purity material, mitigating the risk of supply disruptions caused by quality deviations. Since the process can upgrade existing commercial stock that fails to meet specifications, it provides a flexible buffer against upstream synthesis inconsistencies, ensuring that downstream formulation partners receive a consistent, high-quality input regardless of the source of the crude Bendazac Acid.
- Scalability and Environmental Compliance: The use of standard industrial solvents with well-established recovery protocols facilitates easy scale-up from pilot plants to multi-ton commercial production without requiring specialized equipment. The mild reaction conditions, involving simple reflux and cooling steps, reduce energy consumption compared to high-pressure or cryogenic alternatives, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process, which is increasingly critical for maintaining regulatory licenses in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this refining technology, based on the detailed experimental data provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing lines.
Q: What is the primary impurity targeted by this refining process?
A: The process specifically targets the removal of 3-hydroxy-1-benzyl indazole, a persistent byproduct from the synthesis of Bendazac Acid that often remains at levels of 1-5% in conventional methods.
Q: Why is a dual-solvent system (Acetonitrile then Ethanol) required?
A: Acetonitrile (polar aprotic) is essential during salt formation to keep the hydrophobic indazole impurity dissolved, while Ethanol (polar protic) is critical during recrystallization to control crystal morphology and achieve the correct melting point range of 179-182°C.
Q: Can this method be applied to commercially available raw materials?
A: Yes, the patent explicitly validates the process for refining both newly synthesized Bendazac Lysine and existing commercial raw materials that fail to meet pharmacopoeia standards due to high impurity content.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bendazac Lysine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of ophthalmic drug synthesis requires a partner with deep technical expertise and unwavering commitment to quality. Our R&D team has extensively analyzed advanced refining protocols like the one described in CN102206185A, and we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the stringent purity specifications required for Bendazac Lysine, particularly the suppression of 3-hydroxy-1-benzyl indazole to undetectable levels, demands rigorous QC labs and state-of-the-art crystallization equipment. Our facility is equipped to handle the precise solvent controls and thermal management necessary to replicate these high-yield, high-purity results on an industrial scale, ensuring that your supply of this critical cataract treatment intermediate is never compromised.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how implementing this dual-solvent refinement can optimize your specific bill of materials. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data for our current batches, and review our comprehensive route feasibility assessments to ensure your project moves from development to commercial success with maximum efficiency and minimal risk.
