Scalable Synthesis of 2-Methylthio-4-pyrimidinone for High-Purity Rilpivirine Manufacturing
Scalable Synthesis of 2-Methylthio-4-pyrimidinone for High-Purity Rilpivirine Manufacturing
The pharmaceutical industry continuously seeks robust and cost-effective pathways for synthesizing critical antiretroviral intermediates, particularly for HIV-1 treatments like Rilpivirine. A significant technological breakthrough in this domain is detailed in patent CN114933567B, which outlines a novel preparation method for 2-methylthio-4-pyrimidinone. This compound serves as a pivotal building block in the construction of non-nucleoside reverse transcriptase inhibitors (NNRTIs). The disclosed methodology represents a paradigm shift from traditional alkylation strategies, offering a streamlined approach that enhances both economic viability and operational safety. By leveraging stable starting materials and optimizing reaction conditions, this process addresses long-standing challenges in heterocyclic chemistry, providing a reliable foundation for large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methylthio-4-pyrimidinone has been plagued by reliance on hazardous and costly reagents that complicate supply chain management and increase production overheads. The most prevalent conventional method involves the use of 2-thiouracil derivatives reacting with methyl iodide under reflux conditions. While this pathway can achieve yields around 82%, the dependence on methyl iodide introduces severe drawbacks. Methyl iodide is not only prohibitively expensive due to global regulatory restrictions on volatile organic compounds but also poses significant health and safety risks in an industrial setting. Furthermore, alternative routes utilizing ethyl formylacetate suffer from the inherent instability of the starting materials, necessitating complex in-situ generation protocols that reduce overall process reliability and batch-to-batch consistency.
The Novel Approach
In stark contrast, the innovative strategy presented in the patent data circumvents these issues by utilizing S-methylisothiourea sulfate and diethyl ethoxymethylenemalonate as primary feedstocks. These materials are commercially abundant, thermally stable, and significantly more affordable than their halogenated counterparts. The new route eliminates the need for dangerous alkylation steps, replacing them with a controlled cyclization followed by hydrolysis and decarboxylation. This structural redesign of the synthetic pathway not only simplifies the purification workflow but also drastically reduces the environmental footprint of the manufacturing process. The result is a highly efficient protocol that maintains high yields while ensuring the safety of personnel and the stability of the supply chain.
Mechanistic Insights into Pyrimidine Ring Construction and Functionalization
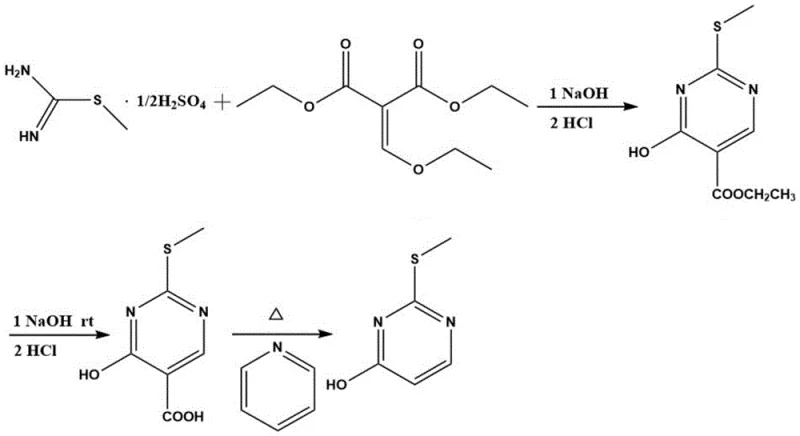
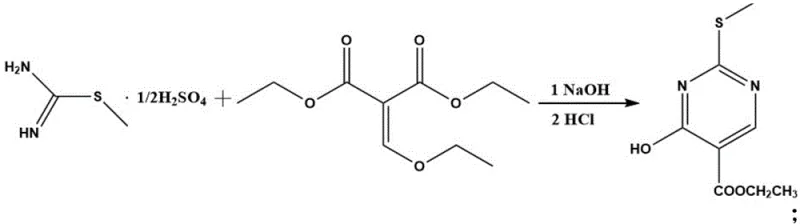
The core of this synthesis lies in a meticulously optimized three-step sequence that constructs the pyrimidine scaffold with high precision. The initial step involves the condensation of S-methylisothiourea sulfate with diethyl ethoxymethylenemalonate in an ethanol-sodium hydroxide medium. This cyclization occurs efficiently at mild temperatures ranging from 0°C to 30°C over a period of 4 to 10 hours. The use of a specific molar ratio, typically between 1:1 and 1:1.2, ensures complete consumption of the limiting reagent, minimizing the formation of unreacted starting materials. Following the reaction, careful acidification to a pH of 3-4 induces the precipitation of the ethyl 4-hydroxy-2-methylthiopyrimidine-5-carboxylate intermediate, allowing for easy isolation via filtration and washing.
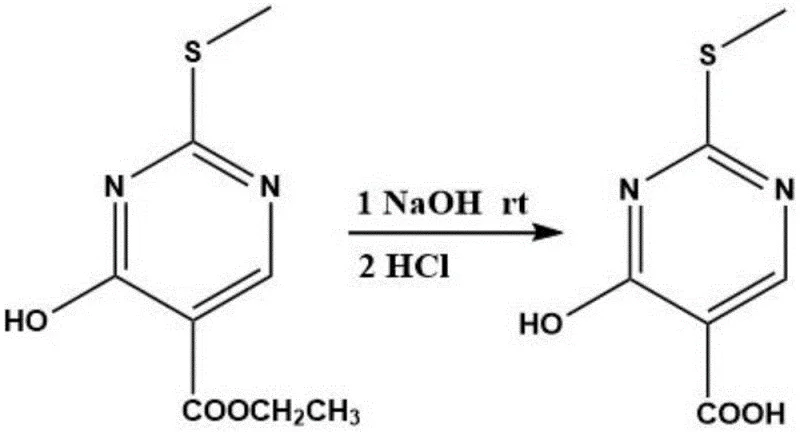
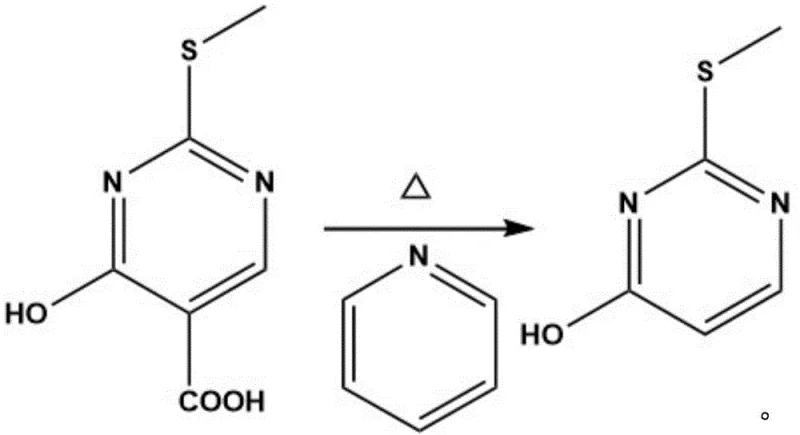
Subsequent transformation involves the hydrolysis of the ester moiety to generate the free carboxylic acid. This is achieved by dissolving the intermediate in a concentrated sodium hydroxide solution (2.7mol/L) and heating the mixture to temperatures between 70°C and 100°C. The alkaline conditions facilitate rapid saponification, converting the ethyl ester into the corresponding sodium salt, which is then acidified to recover the 4-hydroxy-2-(methylthio)pyrimidine-5-carboxylic acid. The final and perhaps most critical step is the thermal decarboxylation. By heating the carboxylic acid in the presence of a basic catalyst such as pyridine at elevated temperatures of 180°C to 230°C, the carboxyl group is cleanly removed. This decarboxylation is remarkably effective, yielding the target 2-methylthio-4-pyrimidinone with exceptional purity and minimal side reactions.
How to Synthesize 2-Methylthio-4-pyrimidinone Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and workup procedures to ensure maximum yield and purity. The process is designed to be scalable, moving seamlessly from laboratory benchtop to pilot plant operations without significant loss of efficiency. Operators must pay close attention to the pH adjustments during the isolation phases, as this dictates the crystallization behavior of the intermediates. For a comprehensive, step-by-step technical guide including exact reagent quantities and safety protocols, please refer to the standardized operating procedure outlined below.
- Cyclization: React S-methylisothiourea sulfate with diethyl ethoxymethylenemalonate in ethanol-sodium hydroxide solution at 0-30°C to form ethyl 4-hydroxy-2-methylthiopyrimidine-5-carboxylate.
- Hydrolysis: Heat the ethyl ester intermediate in 2.7mol/L sodium hydroxide solution at 70-100°C to hydrolyze the ester group into the corresponding carboxylic acid.
- Decarboxylation: Heat the carboxylic acid intermediate with a pyridine catalyst at 180-230°C to remove the carboxyl group and yield the final 2-methylthio-4-pyrimidinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply security. By shifting away from volatile and regulated reagents like methyl iodide, manufacturers can stabilize their raw material costs and mitigate the risk of supply disruptions caused by regulatory changes. The use of stable, salt-based starting materials ensures consistent quality and availability, which is crucial for maintaining uninterrupted production schedules in the competitive pharmaceutical market. Furthermore, the simplified workup procedures reduce the demand for specialized waste treatment infrastructure, leading to significant operational savings.
- Cost Reduction in Manufacturing: The elimination of expensive methylating agents and the use of commodity chemicals like S-methylisothiourea sulfate drastically lower the direct material costs associated with production. Additionally, the high efficiency of the decarboxylation step minimizes material loss, ensuring that a greater proportion of input mass is converted into valuable product. This lean approach to synthesis reduces the overall cost of goods sold, allowing for more competitive pricing in the global API intermediate market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents rather than hazardous liquids simplifies logistics and storage requirements. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental factors, leading to higher batch success rates. This reliability translates into shorter lead times for customers, as manufacturers can confidently commit to delivery schedules knowing that the synthesis pathway is resilient and reproducible on a commercial scale.
- Scalability and Environmental Compliance: The process is inherently designed for industrial implementation, avoiding complex cryogenic conditions or high-pressure equipment. The waste streams generated are easier to treat compared to those containing heavy metals or halogenated organics, facilitating compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also aligns with the sustainability goals of major pharmaceutical partners, enhancing the long-term viability of the supply partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this key pharmaceutical intermediate. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating route feasibility and procurement officers assessing supplier qualifications.
Q: Why is the new synthesis route preferred over traditional methyl iodide methods?
A: Traditional methods rely on expensive and hazardous methyl iodide for methylation. The patented route utilizes S-methylisothiourea sulfate, which is significantly cheaper, more stable, and eliminates the safety risks associated with volatile alkylating agents, thereby reducing overall production costs and environmental impact.
Q: What catalyst is used for the critical decarboxylation step?
A: The process employs pyridine as a basic catalyst during the thermal decarboxylation step. This choice is critical as it facilitates the removal of the carboxyl group at elevated temperatures (180-230°C) with remarkable efficiency, ensuring high conversion rates without requiring complex transition metal catalysts that are difficult to remove.
Q: How does this method improve impurity control for API manufacturing?
A: By avoiding unstable intermediates like ethyl formylacetate and using stable salt forms of starting materials, the reaction profile is much cleaner. The specific pH control (adjusting to pH 3-4) during workup ensures precise precipitation of intermediates, minimizing the carryover of side products into the final rilpivirine intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylthio-4-pyrimidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiretroviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN114933567B can be executed with precision at any volume. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methylthio-4-pyrimidinone meets the exacting standards required for downstream API synthesis, providing our partners with absolute confidence in their supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for this vital compound. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your project timelines and budgetary goals effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
