Scalable Production of Drimane Sesquiterpenoids via Novel Sclareolide Pathway
Scalable Production of Drimane Sesquiterpenoids via Novel Sclareolide Pathway
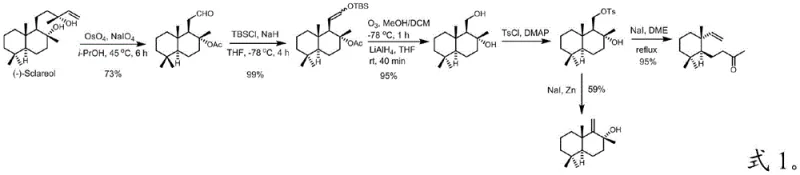
The pharmaceutical and fragrance industries are constantly seeking robust, scalable routes for complex natural product scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN114751814B, which discloses a highly efficient preparation method for sesquiterpenoids, specifically targeting the drimane skeleton. This technology addresses critical bottlenecks in the supply chain of high-value intermediates like 4-((1S,6R)-2,2,6-trimethyl-6-vinylcyclohexyl)butan-2-one and (+)-drim-9(11)-en-8α-ol. By utilizing sclareolide as a readily available starting material, the patented process achieves superior total yields through a streamlined four-step sequence involving reduction, iodination, hydrolysis, and elimination. For R&D directors and procurement specialists, this represents a pivotal shift away from inefficient extraction methods toward a reliable, cost-effective synthetic strategy that ensures consistent quality and supply continuity for complex terpene derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of drimane-type sesquiterpenoids has been plagued by significant technical and economic hurdles. Traditional methods relying on direct extraction from plant sources often suffer from extremely low reaction efficiency and unavoidable product isomerization, leading to inconsistent purity profiles that are unacceptable for fine chemical applications. Furthermore, earlier synthetic attempts, such as those reported by Enzell et al. or Kametani et al., involved excessively long reaction sequences ranging from six to nine steps, which inherently accumulate yield losses at every stage. As illustrated in prior art reaction schemes, these legacy pathways often resulted in dismal total yields, sometimes as low as 40% for key alcohol intermediates, while relying on precious starting materials that required their own complex preparation. The reliance on expensive noble metal catalysts in some conventional routes further exacerbated production costs, making large-scale commercialization economically unviable for many potential applications in agrochemicals and pharmaceuticals.
The Novel Approach
In stark contrast to these legacy challenges, the methodology described in CN114751814B introduces a radically simplified four-step synthetic route that maximizes atom economy and operational simplicity. By initiating the synthesis from sclareolide, a cheap and abundant feedstock, the process bypasses the need for complex chiral pool construction. The core innovation lies in the strategic use of a photo-induced radical iodination followed by a tunable elimination step, which allows for the selective generation of either the vinyl ketone or the unsaturated alcohol depending on reaction conditions. This flexibility is a game-changer for supply chain managers who require specific isomers for downstream coupling reactions. The new approach eliminates the need for transition metal catalysts entirely, replacing them with inexpensive inorganic bases and iodine, thereby drastically reducing the cost of goods sold (COGS) and simplifying waste treatment protocols. The result is a robust manufacturing process capable of delivering high-purity intermediates with total yields reaching up to 74.6% for specific targets, a substantial improvement over the historical benchmarks.
Mechanistic Insights into Photo-Induced Radical Iodination and Elimination
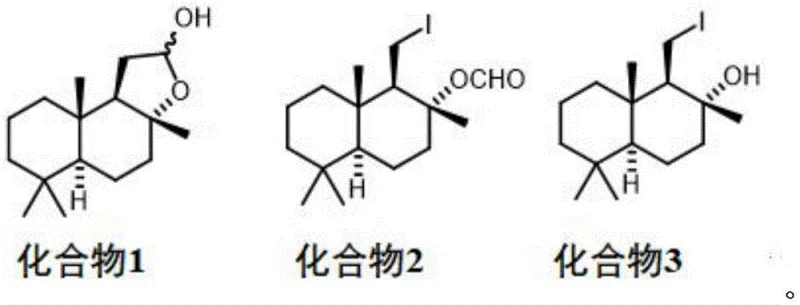
The heart of this synthetic innovation is the precise control over the functionalization of the drimane skeleton, particularly during the iodination and elimination phases. The process begins with the reduction of the lactone moiety in sclareolide to a lactol (Compound 1) using hydride reagents like DIBAL-H at cryogenic temperatures (-78°C to 0°C), ensuring the preservation of the sensitive stereocenters. Subsequently, the introduction of the iodine atom is achieved not through traditional electrophilic substitution, but via a radical mechanism mediated by iodobenzene diacetate and molecular iodine under metal halide lamp irradiation. This photo-chemical activation generates iodine radicals that selectively abstract hydrogen atoms, facilitating the installation of the iodine group at the desired position with high regioselectivity. This step is critical because it sets up the leaving group necessary for the final elimination, and the use of light rather than harsh thermal conditions minimizes side reactions and decomposition of the terpene framework.
Following iodination, the pathway diverges based on the desired final product, showcasing remarkable mechanistic versatility. The hydrolysis of the iodo-lactol intermediate (Compound 2) under mild basic conditions yields the iodo-alcohol (Compound 3) with excellent retention of configuration. The final elimination of hydrogen iodide is where the true selectivity is engineered. By modulating the strength and stoichiometry of the base—ranging from mild carbonates to stronger alkoxides—and adjusting the temperature between 25°C and 80°C, chemists can steer the elimination towards either the exocyclic double bond (forming the vinyl ketone) or the endocyclic double bond (forming the drimenol derivative). This level of control over the impurity profile is essential for R&D teams, as it ensures that the final API or fragrance ingredient meets stringent regulatory specifications without requiring extensive and yield-eroding chromatographic purification. The avoidance of heavy metals throughout this cascade further guarantees a clean impurity profile, free from toxic metal residues.
How to Synthesize Drimane Sesquiterpenoids Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature and reagent stoichiometry to maximize the yield of the target intermediates. The process is designed to be operationally simple, utilizing standard laboratory equipment such as aluminum sand baths and metal halide lamps, which translates easily to pilot and production scales. The following guide outlines the critical operational windows defined in the patent to ensure reproducibility and high purity.
- Reduce sclareolide using DIBAL-H or LDA at -78°C to 0°C to obtain the lactol intermediate (Compound 1).
- Perform radical iodination on Compound 1 using iodobenzene diacetate and iodine under metal halide lamp irradiation at 70-90°C to form Compound 2.
- Hydrolyze Compound 2 with a basic reagent like potassium carbonate in methanol to yield the iodo-alcohol intermediate (Compound 3).
- Execute elimination of hydrogen iodide on Compound 3 using a second base to selectively produce either the vinyl ketone or the drimenol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by utilizing sclareolide, a commodity chemical derived from clary sage, manufacturers can secure a stable and cost-effective feedstock compared to the volatile pricing of exotic natural extracts. The elimination of noble metal catalysts from the process flow removes a significant cost center and mitigates the risk of supply disruptions associated with rare earth elements. Furthermore, the high overall yield of the process means that less raw material is required to produce the same amount of finished goods, directly translating to substantial cost savings in manufacturing. This efficiency also reduces the volume of chemical waste generated per kilogram of product, aligning with increasingly strict environmental regulations and lowering disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive catalytic systems with inexpensive iodine and base reagents. By avoiding the use of palladium or other precious metals, the process eliminates the need for costly metal scavenging steps and rigorous testing for residual metals, which are mandatory in pharmaceutical production. Additionally, the high conversion efficiency at each step minimizes the loss of valuable chiral intermediates, ensuring that the cost per kilogram of the final sesquiterpenoid is significantly lower than that of products synthesized via traditional multi-step routes. This lean manufacturing approach allows for more competitive pricing in the global market for fragrance and pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Dependence on plant extraction subjects supply chains to seasonal variations, climate change impacts, and geopolitical instability in sourcing regions. In contrast, this fully synthetic route relies on stable, industrially produced chemicals that are available year-round from multiple global suppliers. The robustness of the reaction conditions, which tolerate standard solvents like methanol and dichloromethane, ensures that production can be maintained consistently without the risk of crop failure or harvest variability. This reliability is crucial for long-term contracts with major pharmaceutical clients who require guaranteed delivery schedules and consistent quality batches for their own regulatory filings.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations such as liquid-liquid extraction and distillation that are standard in fine chemical plants. The absence of hazardous heavy metals simplifies the wastewater treatment process, reducing the environmental footprint and the associated compliance costs. The ability to tune the reaction to produce different isomers from a common intermediate adds flexibility to the production line, allowing manufacturers to respond quickly to shifting market demands without retooling entire facilities. This adaptability, combined with the high atom economy of the four-step sequence, positions this technology as a sustainable solution for the green chemistry initiatives of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sesquiterpenoid synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity for potential licensees and manufacturing partners.
Q: What are the key advantages of this new sesquiterpenoid synthesis method over traditional extraction?
A: Unlike traditional plant extraction which suffers from low efficiency and isomerization, this chemical synthesis offers a controlled 4-step route with significantly higher total yields (up to 74.6% for specific isomers) and avoids the use of expensive noble metal catalysts.
Q: Can this process be scaled for industrial production of fragrance intermediates?
A: Yes, the process utilizes commercially available raw materials like sclareolide and standard reagents (DIBAL-H, iodine, carbonates). The reaction conditions, such as the use of aluminum sand baths and standard organic solvents, are highly amenable to large-scale commercial scale-up.
Q: How does the method control stereochemistry during the iodination step?
A: The method employs a specific radical iodination protocol using iodobenzene diacetate and iodine under controlled light irradiation (metal halide lamp). This ensures high regioselectivity and preserves the chiral integrity of the drimane skeleton, crucial for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sesquiterpenoid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the global supply of high-value terpene intermediates. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into a robust, GMP-compliant manufacturing process. Our facilities are equipped to handle the specific requirements of this chemistry, including photo-reactors for the iodination step and cryogenic capabilities for the initial reduction. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the stereochemical integrity and impurity profile of every batch.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Whether you require the vinyl ketone intermediate for fragrance applications or the drimenol derivative for pharmaceutical synthesis, our team can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
