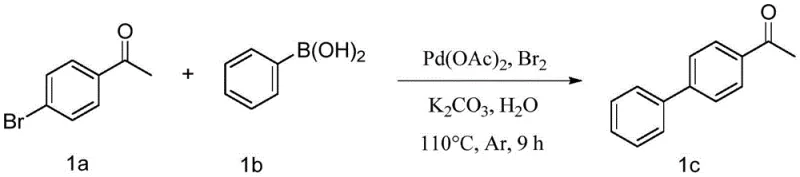
Scaling Green Suzuki Coupling: Ultra-Low Palladium Water-Phase Technology
The pharmaceutical and fine chemical industries are currently undergoing a rigorous transformation towards sustainable manufacturing practices, driven by both regulatory pressure and the economic necessity of reducing waste. Patent CN114031475A introduces a groundbreaking methodology for the synthesis of biphenyl compounds, which are critical structural motifs in numerous active pharmaceutical ingredients (APIs) and agrochemicals. This technology leverages a bromine simple substance-promoted, extremely-low-dose palladium-catalyzed water-phase Suzuki coupling reaction. By shifting the reaction medium from traditional volatile organic solvents to pure water and drastically reducing the catalyst loading to the parts-per-million (ppm) range, this innovation addresses the dual challenges of environmental compliance and production cost efficiency. The ability to achieve high yields under these mild conditions represents a significant leap forward for process chemists seeking to optimize synthetic routes for commercial scale-up.
The strategic implementation of this water-phase technology offers a compelling value proposition for manufacturers of high-purity pharmaceutical intermediates. Traditional cross-coupling reactions often rely on expensive and toxic organic solvents such as THF or dioxane, which require complex recovery systems and pose significant safety hazards. In contrast, the method described in CN114031475A utilizes water as the sole solvent, inherently enhancing the safety profile of the manufacturing process. Furthermore, the use of elemental bromine as a promoter allows for the use of commercially available palladium sources at concentrations as low as 5 ppm to 500 ppm. This reduction in precious metal usage not only lowers the raw material cost but also simplifies the downstream purification process, ensuring that the final product meets stringent heavy metal residue specifications required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the Suzuki-Miyaura coupling reaction has been the gold standard for constructing biaryl bonds, yet its conventional implementation suffers from several critical drawbacks that hinder efficient commercial production. Most traditional protocols require the use of organic solvents to ensure the solubility of reactants and the stability of the catalyst, leading to significant environmental burdens associated with solvent disposal and emission control. Additionally, to achieve acceptable reaction rates and yields, these methods typically necessitate palladium catalyst loadings in the range of 1 to 5 mol%, which is economically inefficient given the high market price of palladium. High catalyst loading also results in substantial metal residues in the crude product, necessitating expensive and time-consuming purification steps such as scavenging or recrystallization to meet pharmaceutical purity standards. These factors collectively inflate the cost of goods sold (COGS) and extend the manufacturing lead time, creating bottlenecks in the supply chain for key drug intermediates.
The Novel Approach
The novel approach detailed in the patent data overcomes these limitations through a synergistic combination of aqueous phase chemistry and bromine promotion. By employing pure water as the reaction medium, the process eliminates the need for hazardous organic solvents, thereby reducing the environmental footprint and operational risks associated with flammability and toxicity. The introduction of liquid bromine acts as a powerful promoter that activates the catalytic cycle, enabling the reaction to proceed efficiently with ultra-low palladium concentrations. This drastic reduction in catalyst loading, down to the ppm level, directly translates to lower raw material costs and significantly reduced metal contamination in the final product. The method demonstrates wide substrate applicability, tolerating various functional groups such as ketones, ethers, and nitriles, which ensures its versatility for synthesizing complex molecular structures required in modern drug discovery and development.
Mechanistic Insights into Bromine-Promoted Aqueous Suzuki Coupling
The mechanistic foundation of this technology lies in the unique role of elemental bromine in facilitating the oxidative addition and transmetallation steps within an aqueous environment. In traditional palladium-catalyzed cycles, the activation of the aryl halide can be sluggish in polar protic solvents like water due to catalyst deactivation or poor solubility of organic substrates. The presence of bromine species helps to maintain the active palladium species in solution and may assist in the generation of more reactive organopalladium intermediates. This promotion effect allows the reaction to proceed at moderate temperatures, typically between 60°C and 150°C, without the need for specialized ligands or microwave irradiation. The robustness of this catalytic system ensures that the reaction kinetics remain favorable even at extremely low catalyst concentrations, providing a reliable pathway for consistent batch-to-batch performance in a manufacturing setting.
Impurity control is another critical aspect where this mechanistic approach offers distinct advantages over conventional methods. High catalyst loading in traditional Suzuki reactions often leads to the formation of palladium black or other metal aggregates that are difficult to remove and can catalyze unwanted side reactions. By operating at ppm-level catalyst concentrations, the formation of such metal-based impurities is inherently minimized. Furthermore, the use of water as a solvent facilitates the separation of organic products through simple extraction, as the inorganic salts and residual catalyst components remain in the aqueous phase. This phase separation capability simplifies the workup procedure, reducing the number of unit operations required and minimizing the loss of product during purification. The result is a cleaner crude product profile that requires less intensive downstream processing to achieve the high purity levels demanded by the pharmaceutical industry.
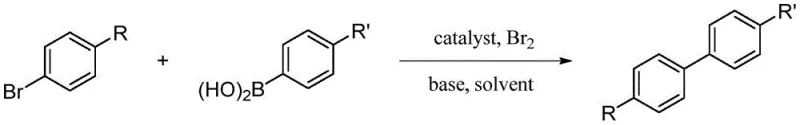
How to Synthesize Biphenyl Compounds Efficiently
The synthesis of biphenyl compounds using this patented method involves a straightforward procedure that is highly amenable to standard chemical manufacturing equipment. The process begins with the dissolution of liquid bromine under an inert atmosphere, followed by the addition of the aryl bromide and arylboronic acid substrates in the presence of a base and the ultra-low dose palladium catalyst. The reaction mixture is then heated to the specified temperature range, allowing the coupling to proceed to completion within a reasonable timeframe of 6 to 12 hours. This operational simplicity reduces the technical barrier for adoption, allowing manufacturers to implement the technology without significant capital investment in new reactor infrastructure. For detailed standardized synthesis steps and specific parameter optimization, please refer to the technical guide provided below.
- Dissolve liquid bromine in a solvent under argon protection and mix with brominated aromatic hydrocarbon and arylboronic acid.
- Add ultra-low dose palladium catalyst (5-500 ppm) and alkali base to the aqueous reaction mixture.
- Heat the reaction to 60-150°C for 6-12 hours, then extract and purify the target biphenyl compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bromine-promoted aqueous Suzuki coupling technology offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant cost reduction potential driven by the drastic decrease in palladium consumption. Since palladium is a precious metal with volatile pricing, reducing its usage from molar percentages to parts-per-million levels insulates the production cost from market fluctuations and lowers the overall material expense. Additionally, the elimination of organic solvents reduces the costs associated with solvent procurement, storage, and waste disposal, further enhancing the economic efficiency of the manufacturing process. These savings can be passed down the supply chain, offering a more competitive pricing structure for the final API intermediates without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The implementation of ultra-low dose palladium catalysis fundamentally alters the cost structure of biaryl synthesis by minimizing the consumption of high-value precious metals. By reducing catalyst loading to the ppm range, the direct material cost associated with the catalytic system is drastically lowered, providing a buffer against palladium price volatility. Furthermore, the use of water as a solvent eliminates the need for expensive organic solvents and the energy-intensive processes required for their recovery and purification. This dual reduction in catalyst and solvent costs results in a leaner manufacturing process that maximizes margin potential while maintaining high reaction yields and product quality standards.
- Enhanced Supply Chain Reliability: The simplicity and robustness of this aqueous-phase method contribute to a more resilient supply chain by reducing the risk of production delays caused by reagent shortages or equipment failures. Water is a universally available and inexpensive resource, removing the dependency on specific organic solvents that may face supply constraints or regulatory restrictions. The mild reaction conditions and tolerance to various functional groups ensure high process reliability, minimizing the occurrence of batch failures that can disrupt delivery schedules. This stability allows for more accurate production planning and inventory management, ensuring a continuous flow of high-purity intermediates to downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the use of standard reaction vessels and the absence of hazardous organic solvents. The aqueous nature of the reaction reduces the risk of fire and explosion, simplifying safety protocols and allowing for larger batch sizes without proportional increases in safety infrastructure costs. Moreover, the green chemistry profile of the process aligns with increasingly stringent environmental regulations regarding VOC emissions and heavy metal discharge. This compliance reduces the regulatory burden and potential fines associated with waste management, ensuring long-term operational sustainability and protecting the company's reputation in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bromine-promoted Suzuki coupling technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing workflows and for assessing its impact on product quality and cost efficiency.
Q: How does the bromine promoter affect catalyst loading in this Suzuki coupling?
A: The addition of elemental bromine significantly activates the catalytic cycle, allowing palladium loading to be reduced to the ppm level (5-500 ppm) while maintaining high yields, which drastically lowers metal residue concerns.
Q: What are the environmental benefits of using pure water as a solvent?
A: Replacing organic solvents with pure water eliminates volatile organic compound (VOC) emissions, reduces flammability risks, and simplifies waste treatment, aligning with strict green chemistry regulations.
Q: Is this method suitable for large-scale commercial production of API intermediates?
A: Yes, the method features simple operation, mild conditions, and easy post-treatment, making it highly scalable for commercial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain competitiveness in the global fine chemical market. Our team of expert process chemists has thoroughly evaluated the bromine-promoted water-phase Suzuki coupling method and confirmed its potential for delivering high-quality biphenyl compounds at a reduced cost. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this green synthesis route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Our commitment to technical excellence and supply chain reliability makes us the ideal partner for securing a sustainable and cost-effective source of complex pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
