Advanced Convergent Synthesis of Ceftizoxime Alapivoxil Hydrochloride for Commercial Scale-Up
Introduction to Patent CN103059047A and Technological Breakthrough
The pharmaceutical industry constantly seeks more efficient pathways for producing complex beta-lactam antibiotics, particularly oral prodrugs that offer improved bioavailability. Patent CN103059047A discloses a groundbreaking method for preparing ceftizoxime alapivoxil hydrochloride, a third-generation cephalosporin prodrug known for its broad antibacterial spectrum and stability against beta-lactamases. This invention represents a significant paradigm shift from traditional linear synthetic strategies to a highly efficient convergent synthesis approach. By strategically decoupling the synthesis of the cephem nucleus and the acyl side chain, the process achieves a total yield exceeding 64%, drastically outperforming historical methods that often struggled to reach 35%. For R&D directors and process chemists, this patent offers a robust blueprint for manufacturing high-purity API intermediates while mitigating the risks associated with toxic reagents and difficult purification protocols.
The core innovation lies in the meticulous design of the reaction sequence, which prioritizes atom economy and operational simplicity. Traditional methods frequently relied on harsh conditions, such as low-temperature anhydrous operations with phosphorus oxychloride or the use of expensive trifluoroacetic acid for deprotection. In contrast, the methodology described in CN103059047A utilizes mild reaction conditions ranging from -20°C to 25°C, making it inherently safer and more scalable for industrial production. Furthermore, the avoidance of AE active esters eliminates the persistent issue of 2-mercaptobenzothiazole residues, a critical quality attribute for meeting stringent regulatory standards in global markets. This technical advancement positions the process as a premier solution for reliable pharmaceutical intermediates supplier networks aiming to enhance supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ceftizoxime alapivoxil has been plagued by inefficiencies inherent to linear synthetic routes. As illustrated in prior art such as US Patent US5389625, early methods required the protection of both the amino and carboxyl groups of 7-ANCA using silane groups, followed by condensation with phosphorus oxychloride under strictly anhydrous conditions. These processes not only demanded specialized equipment and rigorous environmental controls but also resulted in low total yields, often reported around 23.3% or even lower depending on the specific pathway. Additionally, the reliance on column chromatography for purification at multiple stages introduced significant product loss and increased labor intensity, rendering these methods economically unviable for large-scale manufacturing.
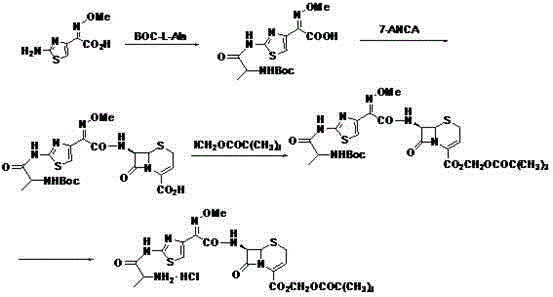
Another major drawback of conventional techniques, specifically those utilizing AE active esters as seen in Chinese patents CN1958591B and CN101613357B, is the generation of hazardous by-products. The use of benzothiazole-based activators inevitably leads to the residual presence of 2-mercaptobenzothiazole (Accelerator M) in the final drug substance. Given that the US FDA has imposed strict limits on such residues due to potential toxicity, this poses a severe compliance risk for manufacturers. Moreover, these linear approaches often necessitate the use of costly reagents like trifluoroacetic acid for deprotection steps, further inflating the cost of goods sold. The cumulative effect of these limitations is a fragile supply chain vulnerable to yield fluctuations and quality failures, highlighting the urgent need for a more robust synthetic strategy.
The Novel Approach
The novel approach detailed in CN103059047A fundamentally restructures the synthesis into a convergent model, effectively bypassing the bottlenecks of linear progression. Instead of building the molecule step-by-step on a single scaffold, this method independently prepares two key fragments: the modified cephem nucleus (7-ANCA-POM) and the activated side chain (Boc-L-Ala-ATMA). These fragments are then coupled in a final condensation step, a strategy that inherently minimizes the propagation of errors and impurities from early stages to the final product. The visual representation of this convergent pathway demonstrates how the 7-amino group of 7-ANCA is first Boc-protected, esterified with iodomethyl pivalate, and then deprotected to yield the versatile 7-ANCA-POM intermediate.
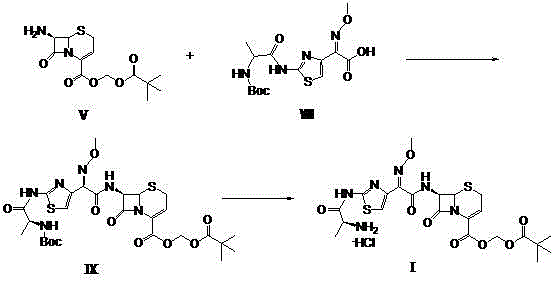
Simultaneously, the side chain is constructed by condensing N-tert-butoxycarbonyl-L-alanine with aminothioxamic acid, creating a stable and reactive intermediate ready for coupling. This modularity allows for parallel processing, which significantly reduces overall production time and enhances throughput capacity. Crucially, the final coupling and deprotection steps are performed under mild acidic conditions without the need for expensive fluorinated acids or toxic phosphorus reagents. The result is a streamlined process where intermediates can be purified via simple solvent extraction and recrystallization rather than complex chromatography. This shift not only boosts the total yield to over 64% but also ensures a cleaner impurity profile, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Convergent Coupling and Impurity Control
The mechanistic success of this synthesis hinges on the strategic use of the tert-butoxycarbonyl (Boc) protecting group and the specific activation of the carboxyl functionality. In the preparation of the nucleus, the 7-amino group of 7-ANCA is selectively protected using di-tert-butyl dicarbonate (Boc2O) in the presence of a base such as sodium carbonate or triethylamine. This protection is critical because it prevents unwanted acylation at the 7-position during the subsequent esterification of the 4-carboxyl group with iodomethyl pivalate. The reaction proceeds smoothly at temperatures between -20°C and 10°C, avoiding the degradation of the sensitive beta-lactam ring that often occurs under harsher conditions. Following esterification, the Boc group is removed using mild acids like hydrochloric acid or phosphoric acid in organic solvents, regenerating the free amine necessary for the final coupling without compromising the newly formed pivaloyloxymethyl ester.
On the side chain front, the activation of Boc-L-Ala is achieved using carbodiimide coupling agents such as DCC or EDC·HCl, often supplemented with catalysts like HOBt or DMAP. This activation converts the carboxylic acid into a reactive O-acylisourea or active ester species in situ, which then reacts efficiently with aminothioxamic acid (ATMA). The resulting Boc-L-Ala-ATMA intermediate possesses a highly reactive carboxyl group that is perfectly poised for nucleophilic attack by the 7-amino group of the 7-ANCA-POM nucleus. This convergent coupling step is the heart of the process, where the two complex fragments unite to form the full cephalosporin structure. The use of mild bases and controlled temperatures during this stage ensures stereochemical integrity is maintained, preventing epimerization at the chiral centers which could lead to inactive or toxic isomers.
Impurity control is intrinsically built into this mechanism by avoiding reagents that generate hard-to-remove by-products. Traditional methods using AE active esters generate 2-mercaptobenzothiazole, a sulfur-containing compound that is difficult to purge completely and poses toxicological risks. By eschewing these activators, the new route eliminates this specific impurity vector entirely. Furthermore, the reliance on crystallization for purification leverages differences in solubility between the product and side products, effectively filtering out unreacted starting materials and minor by-products. The final deprotection step uses hydrochloric acid to remove the Boc group and form the hydrochloride salt directly, which precipitates out of the solution, providing an additional self-purification mechanism. This multi-layered approach to purity ensures that the final high-purity pharmaceutical intermediates meet the rigorous specifications required for clinical applications.
How to Synthesize Ceftizoxime Alapivoxil Hydrochloride Efficiently
The synthesis of ceftizoxime alapivoxil hydrochloride via this convergent route involves a series of well-defined chemical transformations that balance reactivity with selectivity. The process begins with the modification of the 7-ANCA core, followed by the independent assembly of the side chain, and concludes with their union and final salt formation. Each step has been optimized to maximize yield and minimize waste, utilizing common organic solvents and readily available reagents. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for laboratory scale-up and process validation. For detailed standard operating procedures and safety data, please refer to the specific experimental sections below.
- Protect the 7-amino group of 7-ANCA with Boc, esterify the 4-carboxyl group with iodomethyl pivalate, and deprotect to form 7-ANCA-POM.
- Condense Boc-L-Ala with aminothioxamic acid (ATMA) using a carbodiimide coupling agent to form the activated side chain intermediate Boc-L-Ala-ATMA.
- Couple the 7-ANCA-POM nucleus with the Boc-L-Ala-ATMA side chain, followed by final deprotection with acid to yield the target hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this convergent synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the substantial cost savings driven by the elimination of expensive and hazardous reagents. By removing the need for trifluoroacetic acid, phosphorus oxychloride, and silanizing agents, the raw material costs are significantly reduced. Additionally, the replacement of column chromatography with simple crystallization and extraction steps drastically lowers labor costs and solvent consumption. This simplification of the downstream processing workflow translates directly into a lower cost of goods sold, allowing for more competitive pricing in the global market for oral cephalosporin intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of high-cost inputs and inefficient purification steps. Traditional linear routes often require multiple chromatographic separations, which are solvent-intensive and result in significant product loss. In contrast, this novel method relies on crystallization, a unit operation that is far more scalable and economical. The avoidance of trifluoroacetic acid, a costly corrosive reagent, further reduces the expense of raw materials and waste disposal. Moreover, the higher overall yield means that less starting material is required to produce the same amount of final product, amplifying the cost efficiency across the entire production volume.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available starting materials and robust reaction conditions. The key precursors, such as 7-ANCA and Boc-L-Ala, are commodity chemicals with stable supply lines, reducing the risk of shortages. The mild reaction conditions, which do not require strict anhydrous environments or cryogenic temperatures for extended periods, make the process less susceptible to equipment failures or utility fluctuations. This operational robustness ensures consistent production schedules and reliable delivery times for customers relying on high-purity pharmaceutical intermediates for their own drug formulations.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with green chemistry principles and modern manufacturing standards. The absence of benzothiazole residues eliminates a major regulatory hurdle, ensuring that the product meets international safety guidelines without extensive additional testing. The reduction in solvent usage and the elimination of toxic heavy metal catalysts or phosphorus reagents simplify waste treatment and lower the environmental footprint. This makes the process highly scalable, capable of transitioning smoothly from pilot plant batches to multi-ton commercial production without encountering the bottlenecks typical of more complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of ceftizoxime alapivoxil hydrochloride. These answers are derived directly from the technical specifications and advantageous effects described in patent CN103059047A. They are intended to provide clarity on the process capabilities, quality attributes, and strategic benefits for potential partners and stakeholders evaluating this technology for integration into their supply chains.
Q: Why is the convergent synthesis route superior to linear methods for ceftizoxime alapivoxil?
A: The convergent route significantly improves total yield (over 64%) compared to linear routes (often below 35%) by synthesizing the nucleus and side chain separately before coupling, reducing cumulative losses and purification steps.
Q: How does this method address benzothiazole residue concerns?
A: Unlike methods using AE active esters which leave 2-mercaptobenzothiazole residues, this novel route avoids benzothiazole-based activators entirely, ensuring higher purity and compliance with strict FDA regulations on impurities.
Q: What are the key cost-saving factors in this patented process?
A: The process eliminates expensive reagents like trifluoroacetic acid (TFA) and phosphorus oxychloride (POCl3), replaces column chromatography with simple crystallization, and uses mild reaction conditions that reduce energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftizoxime Alapivoxil Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the pharmaceutical sector. Our team of expert chemists has thoroughly analyzed the convergent route described in CN103059047A and is fully equipped to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield process are realized in actual manufacturing output. Our state-of-the-art facilities are designed to handle the specific solvent systems and reaction conditions required for beta-lactam synthesis, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical instrumentation.
We invite you to collaborate with us to leverage this innovative synthesis route for your ceftizoxime alapivoxil hydrochloride requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes quality, cost-efficiency, and regulatory compliance. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can drive value for your organization and secure your position in the market for high-quality oral cephalosporin intermediates.
