Optimizing Topiroxostat Production: A Technical Breakthrough for High-Purity API Intermediate Manufacturing
Optimizing Topiroxostat Production: A Technical Breakthrough for High-Purity API Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical active pharmaceutical ingredients, particularly for treatments addressing chronic conditions like hyperuricemia and gout. Patent CN113121503A introduces a significant advancement in the synthesis of Topiroxostat, a highly selective non-purine xanthine oxidase inhibitor. This technical disclosure outlines a streamlined three-step process that addresses longstanding challenges in yield optimization and impurity control. By fundamentally restructuring the sequence of functional group introductions, specifically delaying the cyano group addition until after the initial hydrazinolysis, the method mitigates the formation of stubborn byproducts that have historically plagued industrial scale-up efforts. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable API intermediate supplier capable of delivering consistent quality. The following analysis dissects the chemical logic, operational parameters, and commercial implications of this novel approach, providing a comprehensive roadmap for integrating this technology into existing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
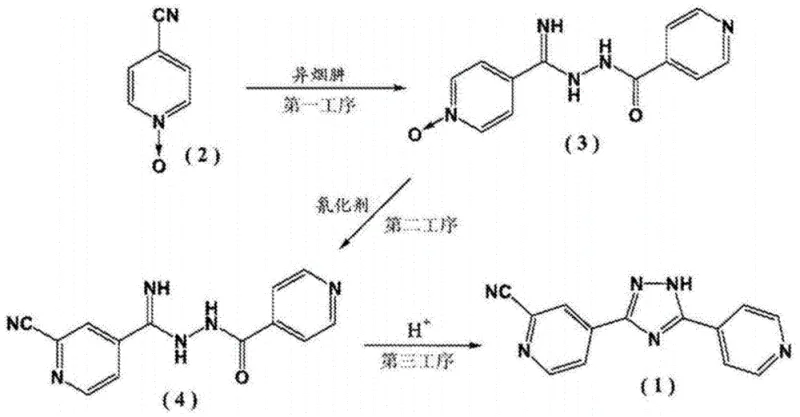
Historical synthetic routes for Topiroxostat, such as those disclosed in earlier patents like CN104411686B, often relied on introducing the cyano group prior to the cyclization step. This sequence presents severe thermodynamic and kinetic disadvantages during manufacturing. When the cyano group is added early, the subsequent ring-closing reaction under high-temperature conditions tends to generate a complex mixture of unreacted intermediates and structural isomers. These byproducts possess solubility profiles remarkably similar to the target molecule, rendering standard recrystallization techniques ineffective for purification. Furthermore, certain prior art methods utilize alcohol and water mixtures that result in the formation of hydrate crystal forms. Converting these hydrates into the medicinally required Crystal Form I necessitates aggressive vacuum drying at temperatures exceeding 80°C, which introduces the risk of thermal degradation and often results in a mixed crystal system rather than a pure polymorph. Such inefficiencies translate directly into increased production costs and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method described in CN113121503A adopts a strategic reversal of the synthetic sequence to bypass these inherent bottlenecks. The process initiates with the hydrazinolysis of 4-cyanopyridine to form a hydrazine intermediate, followed by a condensation reaction with 2-cyano-4-pyridine carboxylic acid. Crucially, the cyclization to form the triazole ring occurs as the final step in an acetic acid medium. This late-stage cyclization ensures that the sensitive cyano functionality is preserved without exposure to harsh conditions prematurely. The use of acetic acid as both solvent and catalyst facilitates a clean ring closure that precipitates the product directly as Crystal Form I upon cooling. This eliminates the need for energy-intensive dehydration steps and significantly simplifies the isolation process. For supply chain heads, this translates to a more predictable manufacturing timeline and reduced dependency on specialized drying equipment, thereby enhancing overall supply chain reliability.
Mechanistic Insights into Hydrazinolysis and Condensation Cyclization
The core of this synthetic innovation lies in the precise control of reaction conditions during the formation of Intermediate 1 and Intermediate 2. The initial step involves reacting 4-cyanopyridine with 80% hydrazine hydrate in an ethanol solvent supplemented with sodium methoxide. The alkaline environment activates the hydrazine nucleophile, facilitating a smooth attack on the nitrile carbon. Temperature control is paramount here; maintaining the reaction between 25°C and 35°C prevents excessive side reactions while ensuring complete conversion. The subsequent isolation of Intermediate 1, either as a free base or a hydrochloride salt, allows for rigorous quality control before proceeding to the next stage. This modularity is vital for R&D teams focusing on impurity谱 analysis, as it isolates potential failure points early in the process. The ability to isolate the intermediate as a stable hydrochloride salt further enhances storage stability and handling safety during transport between production units.
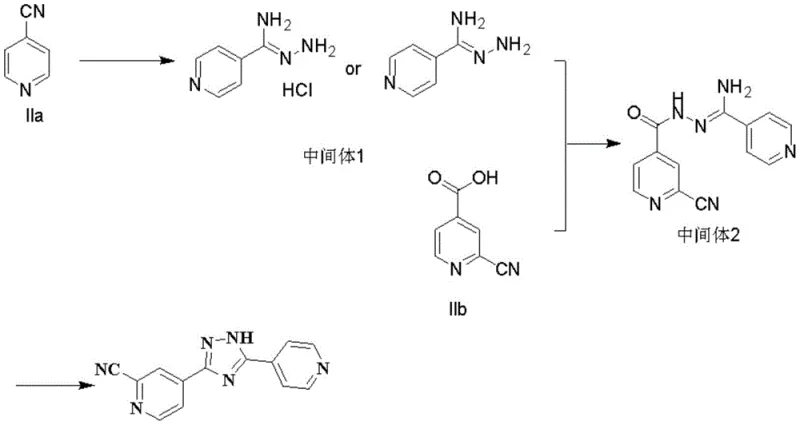
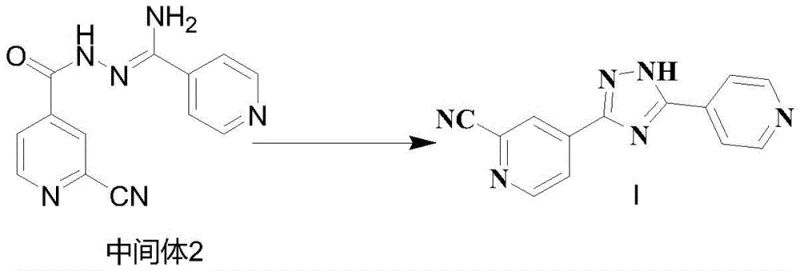
Following the preparation of the hydrazine intermediate, the condensation with 2-cyano-4-pyridine carboxylic acid represents the second critical junction. The patent specifies the use of advanced coupling agents such as EDC/HOBt or DIC/HOBt in DMF solvent. These reagents activate the carboxylic acid moiety efficiently, promoting amide bond formation with high atom economy. The reaction is conducted at controlled low temperatures, typically between 10°C and 20°C, to minimize racemization or decomposition of the activated ester species. The resulting Intermediate 2 is a linear precursor that retains the structural integrity required for the final cyclization. The final transformation involves heating Intermediate 2 in glacial acetic acid under reflux for 2 to 4 hours. This acid-catalyzed cyclocondensation drives the elimination of water and the formation of the 1,2,4-triazole ring. The low solubility of the final product in acetic acid at room temperature promotes spontaneous crystallization, yielding off-white crystals with purity levels reaching 99.9% as confirmed by HPLC analysis.
How to Synthesize Topiroxostat Efficiently
Implementing this synthesis route requires adherence to specific operational protocols to maximize yield and ensure safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton commercial production. Key parameters include the precise stoichiometric ratios of hydrazine to nitrile and the careful selection of condensing agents to minimize urea byproducts. Operators must monitor the reaction progress via thin-layer chromatography or HPLC to determine the exact endpoint for each step, preventing over-reaction which could degrade the product. The work-up procedures involve straightforward aqueous washes and filtration, avoiding complex extraction sequences that generate large volumes of organic waste. This simplicity is a key factor in reducing the environmental footprint of the manufacturing process. Below is the structured guide for executing this synthesis, detailing the critical control points for each phase of the reaction sequence.
- Perform hydrazinolysis on 4-cyanopyridine using hydrazine hydrate in an alcohol solvent with an alkaline reagent to obtain Intermediate 1.
- React Intermediate 1 with 2-cyano-4-pyridine carboxylic acid in a solvent like DMF using a condensing agent such as EDC/HOBt to form Intermediate 2.
- Heat and reflux Intermediate 2 in acetic acid for 2-4 hours, then cool, filter, and dry to obtain Topiroxostat Crystal Form I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound advantages for procurement managers and supply chain directors seeking cost reduction in pharmaceutical manufacturing. The primary driver of value is the significant simplification of the purification process. By avoiding the generation of hard-to-remove byproducts associated with early cyano substitution, the need for repetitive recrystallization or preparative chromatography is drastically reduced. This reduction in downstream processing directly correlates to lower solvent consumption and decreased energy usage for heating and drying operations. Furthermore, the reagents employed, such as ethanol, acetic acid, and standard coupling agents, are commodity chemicals with stable global supply chains. This ensures that production is not vulnerable to shortages of exotic catalysts or specialized reagents, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts, which are often required in alternative cross-coupling routes, removes the necessity for expensive heavy metal scavenging steps. This not only lowers the direct cost of goods sold but also reduces the regulatory burden associated with residual metal testing. The high yield reported in the patent examples, particularly in the final cyclization step, ensures that raw material utilization is optimized, leading to substantial cost savings over the lifecycle of the product. Additionally, the direct formation of Crystal Form I avoids the yield losses typically incurred during polymorph conversion processes, further enhancing the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 4-cyanopyridine and 2-cyano-4-pyridine carboxylic acid means that sourcing risks are minimized. These precursors are produced by multiple vendors globally, preventing single-source bottlenecks. The robustness of the reaction conditions, which tolerate slight variations in temperature and stirring rates without compromising product quality, makes the process ideal for transfer between different manufacturing sites. This flexibility allows for diversified production strategies, ensuring continuous supply even if one facility faces operational disruptions. The simplified workflow also shortens the batch cycle time, allowing for more frequent production runs and better responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: The process generates significantly less three-waste compared to traditional methods, aligning with increasingly stringent environmental regulations. The use of acetic acid, which can be recovered and recycled, minimizes liquid waste discharge. The absence of hazardous reagents reduces the complexity of waste treatment protocols, lowering compliance costs. From a scalability standpoint, the exothermic nature of the reactions is manageable with standard cooling systems, and the solid products are easily filtered using conventional equipment. This ease of scale-up from 100 kgs to 100 MT annual commercial production ensures that the technology can meet growing global demand for gout medications without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Topiroxostat synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. They are intended to clarify the operational feasibility and quality advantages of this route for potential partners and technical stakeholders. Understanding these details is crucial for making informed decisions about technology transfer and long-term supply agreements.
Q: How does this synthesis method improve product purity compared to prior art?
A: By reversing the order of cyano group introduction and cyclization, this method avoids the formation of difficult-to-remove byproducts associated with high-temperature ring closure in earlier stages, resulting in purity levels reaching 99.9%.
Q: What specific crystal form is achieved through this process?
A: The process directly yields Topiroxostat Crystal Form I without the need for high-temperature vacuum drying to remove water molecules, avoiding the mixed crystal issues found in hydrate-based methods.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes common solvents like ethanol and acetic acid and avoids expensive transition metal catalysts, making it highly scalable with reduced three-waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiroxostat Supplier
The technical potential of this synthetic route is immense, offering a pathway to high-quality Topiroxostat that meets the rigorous standards of the global pharmaceutical market. NINGBO INNO PHARMCHEM stands ready to leverage this technology, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of intermediate or API delivered conforms to the highest international standards. We understand the critical nature of supply continuity in the drug development lifecycle and are committed to being a partner that delivers both technical excellence and operational reliability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this more efficient route. Our experts are available to provide specific COA data and route feasibility assessments tailored to your volume needs. By collaborating with us, you secure access to a supply chain that is not only cost-effective but also technically robust, ensuring the success of your hyperuricemia treatment formulations.
