Advanced Semicarbazone Manufacturing: Overcoming Hydrazine Hazards for Scalable Production
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high efficiency with stringent safety standards. Patent CN1934076B introduces a transformative methodology for the preparation of semicarbazone compounds, specifically addressing the critical bottlenecks associated with traditional synthesis pathways. This innovation centers on a novel condensation strategy where a substituted hydrazone compound reacts directly with an aniline derivative, bypassing the need for hazardous free hydrazine or toxic isocyanate intermediates. By shifting the synthetic logic to a safer hydrazone-aniline coupling mechanism, the patent offers a pathway that not only enhances operator safety but also delivers superior yields and purity profiles essential for high-value active ingredients. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for fungicides and pharmaceutical intermediates, ensuring compliance with increasingly rigorous environmental and safety regulations while maintaining cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of semicarbazone derivatives has been plagued by significant safety and efficiency challenges that hinder large-scale commercialization. As illustrated in the prior art, traditional routes often rely on the direct reaction of phenylpropiophenone with N-carbamoyl aniline, a process known to suffer from disappointingly low yields that render it economically unviable for industrial application. 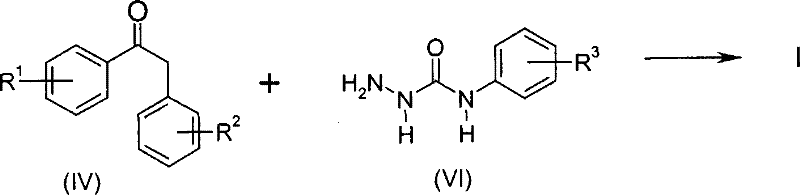 Furthermore, alternative conventional pathways depicted in Scheme 2 necessitate the use of free hydrazine in the initial steps to form hydrazone intermediates.
Furthermore, alternative conventional pathways depicted in Scheme 2 necessitate the use of free hydrazine in the initial steps to form hydrazone intermediates. 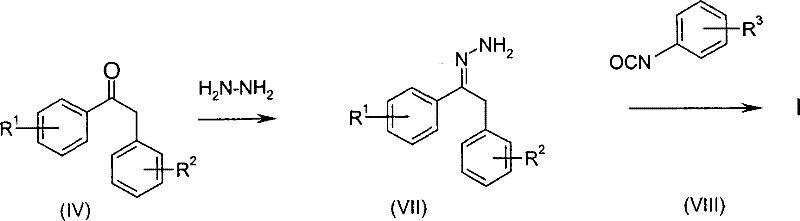 The reliance on hydrazine introduces severe safety liabilities, as it is a recognized potential carcinogen prone to spontaneous decomposition and gas formation upon contact with metallic substances. Additionally, these older methods frequently require the use of phenyl isocyanates, which are highly toxic and demand ultra-safe handling measures, thereby inflating operational costs and complicating waste management protocols due to the generation of hazardous decomposition byproducts.
The reliance on hydrazine introduces severe safety liabilities, as it is a recognized potential carcinogen prone to spontaneous decomposition and gas formation upon contact with metallic substances. Additionally, these older methods frequently require the use of phenyl isocyanates, which are highly toxic and demand ultra-safe handling measures, thereby inflating operational costs and complicating waste management protocols due to the generation of hazardous decomposition byproducts.
The Novel Approach
In stark contrast to these hazardous legacy methods, the patented process introduces a streamlined two-step sequence that fundamentally alters the risk profile and efficiency of semicarbazone manufacturing. The core innovation lies in the reaction of a pre-formed hydrazone compound (Formula II) with a substituted aniline (Formula III), effectively eliminating the need to handle free hydrazine or toxic isocyanates during the critical coupling step. This approach allows for the use of stable hydrazide precursors, such as methyl carbazate, which are significantly safer and easier to handle on a multi-ton scale. By decoupling the formation of the hydrazone moiety from the final condensation with the aniline, the process achieves a remarkable improvement in reaction control. The result is a synthesis that not only mitigates the extreme safety risks associated with hydrazine gas but also simplifies the downstream purification processes, leading to a more robust and commercially attractive manufacturing protocol for complex organic intermediates.
Mechanistic Insights into Hydrazone-Aniline Condensation
The mechanistic elegance of this invention relies on a nucleophilic substitution-condensation sequence driven by thermodynamic equilibrium control. The reaction initiates with the interaction between the nucleophilic nitrogen of the aniline derivative (Formula III) and the electrophilic center of the hydrazone intermediate (Formula II).  Crucially, the reaction is performed in high-boiling aromatic solvents like toluene or xylene at elevated temperatures ranging from 70°C to 150°C. Under these conditions, the reaction generates an alcohol byproduct (R-H), typically methanol or ethanol, depending on the hydrazide precursor used. The driving force for the reaction completion is the continuous removal of this alcohol byproduct via azeotropic distillation. By employing a reaction vessel equipped with a distillation column, the system continuously shifts the equilibrium towards the product side according to Le Chatelier's principle. This dynamic removal of volatiles ensures that the conversion proceeds to near-completion without requiring a vast excess of reagents, thereby maximizing atom economy and minimizing waste generation in the final reaction mass.
Crucially, the reaction is performed in high-boiling aromatic solvents like toluene or xylene at elevated temperatures ranging from 70°C to 150°C. Under these conditions, the reaction generates an alcohol byproduct (R-H), typically methanol or ethanol, depending on the hydrazide precursor used. The driving force for the reaction completion is the continuous removal of this alcohol byproduct via azeotropic distillation. By employing a reaction vessel equipped with a distillation column, the system continuously shifts the equilibrium towards the product side according to Le Chatelier's principle. This dynamic removal of volatiles ensures that the conversion proceeds to near-completion without requiring a vast excess of reagents, thereby maximizing atom economy and minimizing waste generation in the final reaction mass.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the stereochemical outcome of the semicarbazone product. The process naturally favors the formation of the E-isomer over the Z-isomer, with reported ratios often exceeding 4:1, which is highly desirable for biological activity in sterilant applications. 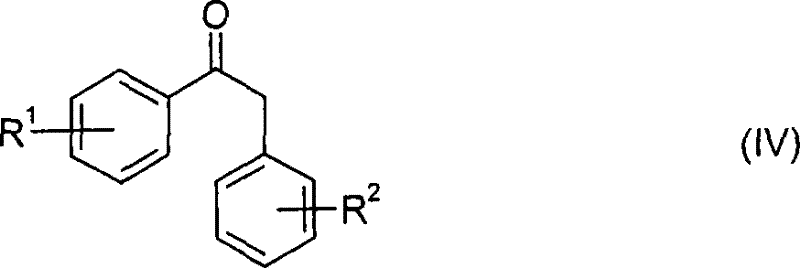 The use of specific acid catalysts, such as p-toluenesulfonic acid or acetic acid, can further refine the reaction kinetics, although the patent notes that the reaction can proceed efficiently even in the absence of added acid under optimized thermal conditions. The high purity of the crude product, often surpassing 90% as determined by HPLC, suggests that side reactions such as hydrolysis or polymerization are effectively suppressed. This inherent selectivity reduces the burden on downstream crystallization steps, allowing manufacturers to achieve specification-grade material with fewer unit operations, directly translating to reduced processing time and lower energy consumption per kilogram of finished product.
The use of specific acid catalysts, such as p-toluenesulfonic acid or acetic acid, can further refine the reaction kinetics, although the patent notes that the reaction can proceed efficiently even in the absence of added acid under optimized thermal conditions. The high purity of the crude product, often surpassing 90% as determined by HPLC, suggests that side reactions such as hydrolysis or polymerization are effectively suppressed. This inherent selectivity reduces the burden on downstream crystallization steps, allowing manufacturers to achieve specification-grade material with fewer unit operations, directly translating to reduced processing time and lower energy consumption per kilogram of finished product.
How to Synthesize Semicarbazone Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of mixing, heating, and distillation to ensure optimal yield and safety. Operators begin by preparing the hydrazone intermediate through the reaction of a substituted phenylpropiophenone with a hydrazide in an alcoholic solvent, a step that can be performed with high conversion rates. Following the isolation or direct use of the hydrazone, it is combined with the specific aniline derivative in an aromatic solvent system. The mixture is then heated to reflux, and the critical phase of azeotropic distillation begins, where the alcohol byproduct is continuously removed to drive the reaction forward.
- Prepare the hydrazone intermediate by reacting a substituted phenylpropiophenone (Formula IV) with a hydrazide (Formula V) in an alcoholic or aromatic solvent, optionally using an acid catalyst.
- Mix the isolated or crude hydrazone compound (Formula II) with a substituted aniline (Formula III) in an aromatic solvent such as toluene or xylene.
- Heat the reaction mixture to reflux (70-150°C) while continuously removing the alcohol byproduct via azeotropic distillation to drive the equilibrium towards the semicarbazone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic advantages that extend beyond simple yield improvements. The elimination of free hydrazine from the process workflow removes a major regulatory and safety hurdle, significantly reducing the costs associated with specialized containment systems, hazardous waste disposal, and worker protection equipment. This simplification of the safety infrastructure allows for more flexible manufacturing scheduling and reduces the risk of production stoppages due to safety audits or incidents. Furthermore, the use of stable, commercially available starting materials like substituted anilines and hydrazides ensures a reliable supply chain, mitigating the volatility often associated with sourcing highly regulated hazardous reagents. The ability to produce high-purity intermediates with minimal downstream processing also translates to substantial cost reductions in utility consumption and solvent recovery, enhancing the overall margin profile of the final active ingredient.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive and hazardous reagents like phenyl isocyanates and free hydrazine, which require costly handling and disposal protocols. By utilizing stable hydrazide precursors and driving the reaction through azeotropic distillation, the method minimizes reagent excess and reduces the complexity of the workup procedure. This streamlined approach lowers the overall operational expenditure by reducing the number of purification steps required to meet purity specifications, thereby saving on solvent usage and energy costs associated with extended crystallization cycles.
- Enhanced Supply Chain Reliability: Sourcing stable intermediates such as substituted anilines and hydrazides is far more predictable than managing the supply of hazardous gases or unstable isocyanates. This stability ensures consistent production schedules and reduces the lead time for high-purity agrochemical intermediates, as manufacturers are less likely to face delays due to regulatory restrictions on hazardous material transport. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the continuity of supply for downstream customers relying on just-in-time delivery models.
- Scalability and Environmental Compliance: The avoidance of hydrazine and isocyanates drastically simplifies the environmental compliance landscape, as the waste streams generated are less toxic and easier to treat. This facilitates easier scale-up from pilot plant to commercial production volumes, as the engineering controls required are less stringent than those for conventional hydrazine-based routes. The process inherently generates less hazardous waste, aligning with green chemistry principles and reducing the carbon footprint of the manufacturing operation, which is increasingly a key criterion for selection by environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semicarbazone synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear understanding of the operational parameters and benefits.
Q: Why is the new semicarbazone synthesis method safer than conventional routes?
A: Conventional methods often require the use of free hydrazine, which is a potential carcinogen and poses explosion risks due to spontaneous decomposition. The patented process utilizes stable hydrazide precursors and avoids handling hazardous hydrazine gas or toxic isocyanates, significantly improving operational safety.
Q: What is the typical yield and purity achievable with this process?
A: The patent data indicates that the novel method consistently achieves yields of at least 80%, often exceeding 90% based on the hydrazone intermediate. Furthermore, the crude product typically demonstrates high purity levels above 90% without the need for complex recrystallization steps.
Q: How does the process control the E/Z isomer ratio of the final product?
A: The reaction conditions naturally favor the formation of the E-isomer, with ratios often exceeding 4:1 (E:Z). This is advantageous for applications requiring specific stereochemistry, such as in sterilant formulations, and the ratio can be further optimized using iodine-catalyzed isomerization if necessary.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semicarbazone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for the production of high-value intermediates. Our technical team has extensively evaluated the methodology described in CN1934076B and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art distillation units and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of semicarbazone intermediate delivered meets the exacting standards required for agrochemical and pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this optimized process can enhance your product portfolio. Let us help you navigate the complexities of chemical manufacturing with a partner dedicated to safety, quality, and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
