Advanced Chiral Iridium Catalysts for Efficient Asymmetric C-H Functionalization and Commercial Scale-Up
The landscape of asymmetric synthesis is undergoing a transformative shift with the advent of novel transition metal catalysts capable of activating inert chemical bonds with high precision. Patent CN109970713B introduces a groundbreaking class of amino acid-derived chiral ligands containing bidentate coordination groups, specifically designed to overcome the historical limitations of Iridium(III) catalysis. This technology addresses the critical challenge of stereoselective C(sp3)-H bond functionalization, a reaction type that has long eluded efficient catalytic control due to the high energy barrier and lack of effective chiral environments. By leveraging abundant and inexpensive natural amino acids as the chiral pool source, this invention provides a sustainable and economically viable pathway for constructing complex chiral molecules, particularly gamma-lactams, which are ubiquitous scaffolds in pharmaceutical intermediates and agrochemicals.
The significance of this development extends beyond academic curiosity; it offers a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. Traditional methods often rely on stoichiometric chiral auxiliaries or expensive, difficult-to-synthesize ligands that limit industrial applicability. In contrast, the catalysts described in this patent utilize a modular design where the chiral information is encoded directly into the ligand backbone via simple derivatization. This approach not only diversifies the library of available chiral catalysts but also ensures that the production of these high-value reagents remains cost-effective, aligning perfectly with the industry's demand for cost reduction in API manufacturing without compromising on optical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the application of amino acids as ligands for Iridium(III) catalysts has been fraught with significant challenges, primarily stemming from inadequate stereocontrol. In conventional models, amino acids typically coordinate to the metal center through a nitrogen atom from the amine and an oxygen atom from the carboxylate group. While this N,O-coordination mode is common, it suffers from a critical geometric flaw: the chiral alpha-carbon substituent (the R group) is positioned distally relative to the iridium catalytic center. This spatial separation means that the steric bulk of the amino acid side chain cannot effectively shield one face of the metal center during the bond-forming event. Consequently, the energy difference between the competing diastereomeric transition states is minimal, leading to poor enantiomeric excess (ee) values and often resulting in a mixture of catalyst diastereomers that are difficult to separate. Furthermore, the lability of the carboxylate coordination can lead to catalyst decomposition under harsh reaction conditions, limiting the substrate scope and turnover numbers.
The Novel Approach
The innovation presented in Patent CN109970713B fundamentally reengineers the coordination geometry to solve these intrinsic problems. Instead of relying on the native carboxylate, the inventors have introduced a robust bidentate nitrogen-containing directing group, such as an 8-aminoquinoline moiety, onto the amino acid skeleton. This modification forces the ligand to coordinate through two nitrogen atoms (N,N-coordination), forming a stable five-membered chelate ring with the Iridium(III) center. 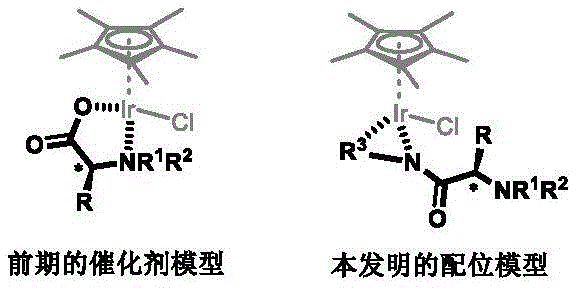 This structural rearrangement pulls the chiral environment of the amino acid backbone into immediate proximity with the metal center. The result is a rigid, well-defined chiral pocket that exerts powerful steric influence over the incoming substrate. This new coordination mode not only stabilizes the catalyst against decomposition but also ensures that a single diastereomer of the catalyst is formed predominantly, thereby delivering exceptional stereocontrol in downstream reactions. This represents a paradigm shift from passive chirality transfer to active, proximal stereochemical enforcement.
This structural rearrangement pulls the chiral environment of the amino acid backbone into immediate proximity with the metal center. The result is a rigid, well-defined chiral pocket that exerts powerful steric influence over the incoming substrate. This new coordination mode not only stabilizes the catalyst against decomposition but also ensures that a single diastereomer of the catalyst is formed predominantly, thereby delivering exceptional stereocontrol in downstream reactions. This represents a paradigm shift from passive chirality transfer to active, proximal stereochemical enforcement.
Mechanistic Insights into Ir(III)-Catalyzed C-H Activation
The efficacy of these novel catalysts lies in their ability to facilitate a sophisticated catalytic cycle involving C-H bond cleavage and functionalization. The mechanism begins with the coordination of the substrate, typically an oxime ester or similar directing group-containing molecule, to the electrophilic Iridium(III) center. This coordination displaces a labile ligand and positions the target C-H bond for activation. The catalyst then promotes the cleavage of the inert C(sp3)-H bond via a concerted metalation-deprotonation (CMD) pathway or a sigma-bond metathesis mechanism, generating a cyclometallated Ir(III) intermediate. This step is the turnover-limiting phase in many C-H functionalizations, but the electron-rich nature of the bidentate nitrogen ligands enhances the electron density at the metal center, facilitating this difficult bond breaking process.
Following C-H activation, the catalytic cycle proceeds through an intramolecular migratory insertion or coupling event, depending on the specific reaction variant. In the synthesis of gamma-lactams, the intermediate undergoes a rearrangement that leads to the formation of the new C-C or C-N bond, closing the ring structure. 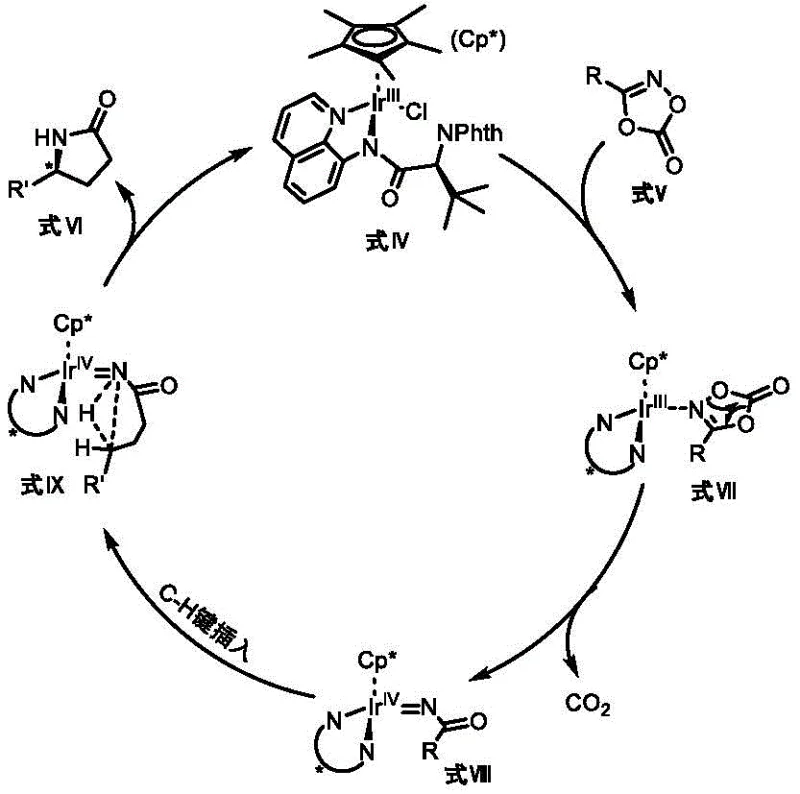 Crucially, the rigid chiral environment established by the bidentate ligand dictates the facial selectivity of this insertion step. The bulky substituents on the amino acid backbone block one approach trajectory of the substrate, forcing the reaction to proceed through the lower-energy transition state that yields the desired enantiomer. Finally, reductive elimination or protonolysis releases the chiral product and regenerates the active Ir(III) species, ready for another turnover. This mechanistic robustness allows the reaction to tolerate functional groups that would typically poison softer metal catalysts, expanding the utility of this method for synthesizing diverse high-purity pharmaceutical intermediates.
Crucially, the rigid chiral environment established by the bidentate ligand dictates the facial selectivity of this insertion step. The bulky substituents on the amino acid backbone block one approach trajectory of the substrate, forcing the reaction to proceed through the lower-energy transition state that yields the desired enantiomer. Finally, reductive elimination or protonolysis releases the chiral product and regenerates the active Ir(III) species, ready for another turnover. This mechanistic robustness allows the reaction to tolerate functional groups that would typically poison softer metal catalysts, expanding the utility of this method for synthesizing diverse high-purity pharmaceutical intermediates.
How to Synthesize Chiral Ir(III) Catalysts Efficiently
The preparation of these high-performance catalysts is remarkably straightforward, designed to minimize operational complexity and maximize yield. The process begins with the protection of commercially available amino acids, followed by amidation with the bidentate directing group precursor. This ligand synthesis can be achieved under mild conditions using standard coupling reagents, avoiding the need for cryogenic temperatures or exotic reagents. Once the ligand is isolated, the metallation step involves a simple mixing of the ligand with the iridium dimer precursor [Cp*IrCl2]2 in the presence of a mild inorganic base. 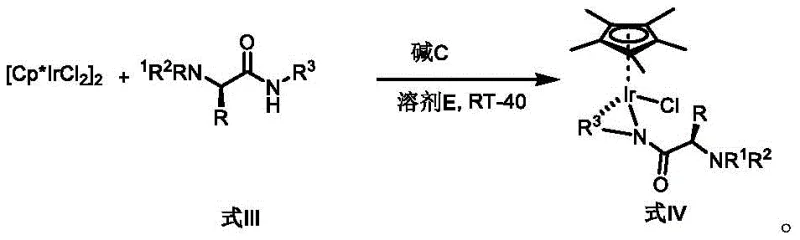 This one-pot metallation proceeds rapidly at room temperature to 40°C, yielding the monomeric catalyst in high purity after simple chromatographic purification. The detailed standardized synthesis steps for implementing this technology in your laboratory are outlined below.
This one-pot metallation proceeds rapidly at room temperature to 40°C, yielding the monomeric catalyst in high purity after simple chromatographic purification. The detailed standardized synthesis steps for implementing this technology in your laboratory are outlined below.
- Protect the amino group of the starting amino acid using acid anhydride or acyl chloride under controlled temperature conditions to form the N-protected intermediate.
- Couple the protected amino acid with a bidentate directing group precursor, such as 8-aminoquinoline derivatives, using condensing agents to generate the chiral ligand.
- React the purified chiral ligand with [Cp*IrCl2]2 dimer in the presence of a mild base like cesium carbonate in dichloromethane to yield the final monomeric Ir(III) catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers compelling strategic advantages that go beyond mere technical performance. The primary driver for cost optimization is the reliance on natural amino acids as the starting material. Unlike synthetic chiral ligands that require multi-step asymmetric synthesis themselves, amino acids are commodity chemicals produced on a massive global scale. This abundance translates directly into significantly reduced raw material costs and eliminates the supply chain volatility associated with niche chiral building blocks. Furthermore, the simplified synthesis of the catalyst itself—requiring only a single metallation step from the ligand—reduces the manufacturing footprint and labor hours required to produce the catalyst, further driving down the cost per gram of the active reagent.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of expensive chiral auxiliaries and the reduction in catalyst loading required to achieve high conversion. Because the catalyst exhibits high turnover numbers and excellent stability, manufacturers can operate with lower catalyst loadings while maintaining high yields. Additionally, the tolerance of the reaction to air and moisture in certain steps reduces the capital expenditure required for specialized inert atmosphere equipment (such as gloveboxes or rigorous Schlenk lines). This operational flexibility allows for the use of standard reactor vessels, significantly lowering the barrier to entry for implementing this chemistry on a large scale and resulting in substantial overall process cost savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, shelf-stable reagents. The amino acid-based ligands are chemically stable and do not require the stringent cold-chain logistics often necessary for sensitive organometallic precursors. The ability to synthesize the catalyst on-demand from stable solid precursors mitigates the risk of supply disruptions for the pre-formed catalyst. Moreover, the broad substrate scope of the catalyst means that a single catalyst family can be applied to the synthesis of multiple different intermediates, allowing companies to consolidate their supplier base and reduce the complexity of their chemical inventory management. This versatility ensures a more reliable and continuous supply of critical intermediates for downstream drug production.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns with green chemistry principles. The high atom economy of C-H functionalization avoids the generation of stoichiometric waste associated with traditional cross-coupling reactions that require pre-functionalized substrates (e.g., halides). The ability to run reactions in solvent systems that can tolerate water reduces the volume of organic solvents required for drying and purification, simplifying waste stream management. The high selectivity (up to 99% ee) minimizes the formation of unwanted enantiomers, reducing the need for resource-intensive recrystallization or chiral chromatography steps to upgrade optical purity. This streamlined downstream processing facilitates easier scale-up from kilogram to tonne production while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a realistic overview of the technology's capabilities and limitations for potential adopters.
Q: Why are traditional amino acid ligands ineffective for Ir(III) catalysis?
A: Conventional amino acid ligands typically coordinate through N and O atoms, placing the chiral center too far from the iridium metal core. This distance prevents effective transmission of stereochemical information, resulting in poor enantioselectivity and the formation of diastereomeric mixtures.
Q: What is the key advantage of the bidentate coordination strategy in this patent?
A: By introducing a strong nitrogen-donor bidentate group like 8-aminoquinoline, the ligand forms a rigid five-membered chelate ring with the iridium center. This structural rigidity brings the chiral environment of the amino acid backbone into close proximity with the catalytic site, drastically enhancing stereocontrol up to 99% ee.
Q: Can these catalysts tolerate industrial reaction conditions?
A: Yes, the developed catalysts demonstrate remarkable robustness. They can operate effectively under air atmosphere and tolerate the presence of water in the solvent system, which simplifies processing requirements and reduces the need for stringent anhydrous conditions typical of organometallic chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ir(III) Catalyst Supplier
The technological potential of amino acid-derived bidentate Ir(III) catalysts represents a significant opportunity for advancing asymmetric synthesis in the pharmaceutical sector. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like Patent CN109970713B can be successfully translated into robust industrial processes. Our team of process chemists is adept at optimizing reaction parameters such as solvent selection, temperature profiles, and workup procedures to meet stringent purity specifications required for GMP manufacturing. With our rigorous QC labs and state-of-the-art analytical capabilities, we guarantee the consistent quality and batch-to-batch reproducibility essential for regulatory approval.
We invite you to explore how this advanced catalytic technology can optimize your current synthetic routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data for our catalyst libraries and comprehensive route feasibility assessments to help you accelerate your development timelines. Let us partner with you to turn complex chiral synthesis challenges into competitive commercial advantages.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
