Advanced Manufacturing of Upadacitinib Intermediates: A Safer, Scalable Synthetic Route
Introduction to the Novel Synthetic Pathway
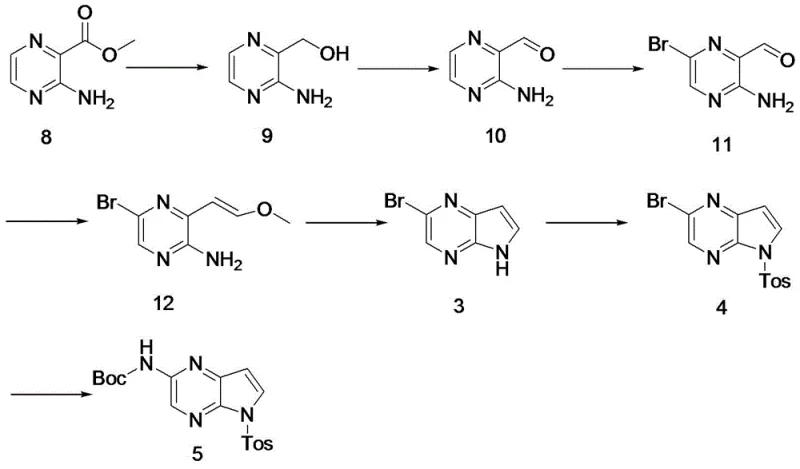
The pharmaceutical industry is constantly seeking robust, scalable, and safe methods for producing complex heterocyclic intermediates, particularly for high-value targets like JAK inhibitors. Patent CN116396299A, published in July 2023, discloses a groundbreaking method for preparing a key intermediate for Upadacitinib (also known as Wu Pati Nib), specifically tert-butyl 5-tosyl-5H-pyrrolo[2,3-b]pyrazine-2-carbamate. This molecule serves as a critical building block in the synthesis of Upadacitinib, a potent JAK1 inhibitor approved for treating rheumatoid arthritis and atopic dermatitis. The disclosed technology represents a significant leap forward in process chemistry, moving away from hazardous reagents and expensive catalytic systems towards a more economical and operationally simple 7-step sequence. By starting from the readily available methyl 3-aminopyrazine-2-carboxylate, the inventors have constructed a route that achieves a total yield of 40% and a final purity exceeding 99%, addressing major pain points in the existing supply chain regarding safety and cost.
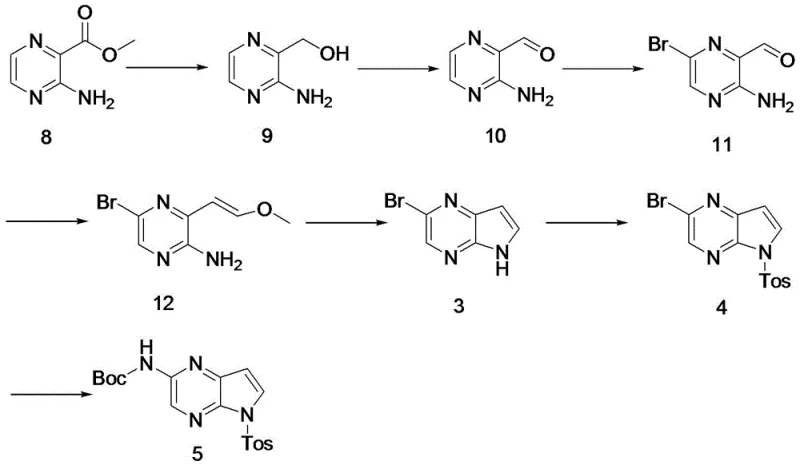
This new methodology is particularly relevant for reliable pharmaceutical intermediate suppliers looking to optimize their manufacturing portfolios. The route effectively bypasses the limitations of previous patents, such as US2015118229, which relied on harsh conditions and scarce starting materials. The strategic shift to a Wittig reaction followed by acid-catalyzed cyclization not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing three-waste treatment requirements. For procurement and R&D teams, understanding this pathway is essential for securing a stable supply of high-quality Upadacitinib precursors in an increasingly competitive market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing the pyrrolo[2,3-b]pyrazine core have been plagued by significant technical and economic hurdles. The most prominent existing route, described in US2015118229, utilizes 3,5-dibromopyrazin-2-amine as a starting material. This approach necessitates a Sonogashira cross-coupling reaction involving palladium and copper catalysts to introduce the alkyne functionality required for ring closure. While chemically feasible, this method suffers from several critical drawbacks in an industrial setting. Firstly, the starting material, 3,5-dibromopyrazin-2-amine, is not commercially abundant and often requires additional synthesis steps, adding to the lead time and cost. Secondly, the reliance on Pd/Cu catalysis introduces the risk of catalyst poisoning and necessitates rigorous purification steps to remove trace heavy metals, which is a stringent requirement for API intermediates.
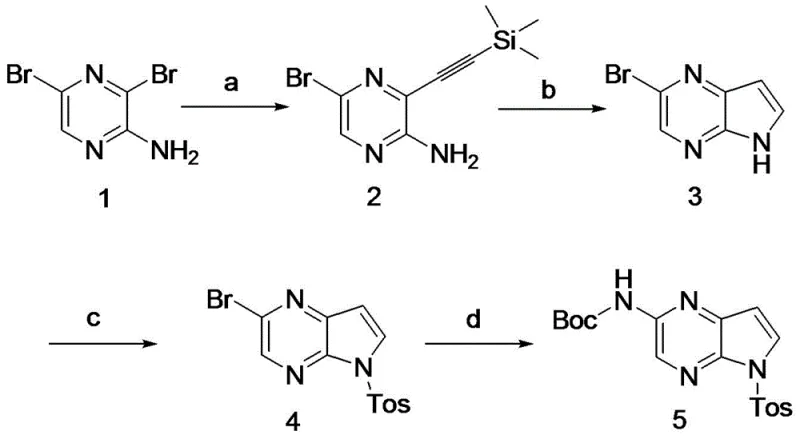
Furthermore, the ring-closing step in conventional methods typically employs sodium hydride (NaH) in DMF under strong basic conditions. Sodium hydride is a pyrophoric solid that poses severe safety hazards, especially during scale-up, requiring specialized equipment and handling protocols. The use of such dangerous reagents increases operational costs and insurance liabilities. Additionally, the overall yield of these traditional routes is reported to be around 18%, which is economically inefficient for large-scale production. The combination of low yield, high safety risk, and expensive catalysts makes these conventional methods suboptimal for modern cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The method disclosed in CN116396299A offers a transformative solution by completely redesigning the synthetic logic. Instead of building the ring via alkyne coupling, the new route constructs the pyrrole ring through a Wittig reaction followed by an acid-mediated cyclization. This strategy begins with methyl 3-aminopyrazine-2-carboxylate, a commodity chemical that is cheap and easy to source. The elimination of the Sonogashira coupling removes the dependency on precious metal catalysts entirely for the core construction, thereby simplifying the impurity profile and reducing raw material costs. The replacement of the hazardous sodium hydride cyclization with a mild acid-catalyzed process (using hydrochloric acid) fundamentally changes the safety profile of the manufacturing process, making it far more suitable for commercial scale-up of complex pharmaceutical intermediates.
Moreover, the new route incorporates a strategic bromination step using pyridinium tribromide, which offers better control and safety compared to handling elemental bromine directly. The sequential optimization of each step—reduction, oxidation, bromination, Wittig olefination, cyclization, protection, and coupling—results in a cumulative yield of 40%, more than double that of the prior art. This substantial improvement in efficiency, coupled with the ability to achieve >99% purity, positions this novel approach as the preferred method for industrial application. It directly addresses the need for reducing lead time for high-purity intermediates by streamlining the synthesis and minimizing purification bottlenecks.
Mechanistic Insights into the Wittig-Acid Cyclization Sequence
The heart of this innovative synthesis lies in the transformation of the brominated aldehyde (Compound 11) into the fused pyrrolopyrazine system (Compound 3). This is achieved through a two-stage cascade involving a Wittig reaction and subsequent acid-catalyzed cyclization. In the first stage, Compound 11 reacts with (methoxymethyl)triphenylphosphonium chloride in the presence of potassium tert-butoxide. The choice of potassium tert-butoxide is critical; it provides a moderately alkaline environment that facilitates the formation of the phosphorus ylide without promoting side reactions that stronger bases might induce. This step installs the necessary two-carbon unit with a methoxy group, creating the vinyl ether intermediate (Compound 12). The reaction is highly selective, ensuring that the amino group on the pyrazine ring remains intact and does not interfere with the phosphonium salt.
Following the Wittig olefination, the reaction mixture undergoes acid-catalyzed cyclization. Upon exposure to acidic conditions (such as 3N hydrochloric acid) and heat (75°C), the vinyl ether moiety hydrolyzes and subsequently attacks the adjacent amino group on the pyrazine ring. This intramolecular nucleophilic attack closes the five-membered pyrrole ring, releasing methanol and forming the aromatic 5H-pyrrolo[2,3-b]pyrazine core (Compound 3). This mechanism is superior to the base-mediated cyclization of prior art because it avoids the generation of hydrogen gas and the exothermic risks associated with sodium hydride. Furthermore, the acidic workup naturally precipitates the product, simplifying isolation and improving the purity profile to 99.4% at this stage. This mechanistic elegance ensures that the process is not only chemically efficient but also operationally robust for large-scale reactors.
Impurity control is another hallmark of this mechanism. By avoiding transition metal catalysts in the ring-forming steps, the potential for metal-catalyzed side reactions or residual metal contamination is eliminated. The use of pyridinium tribromide for the preceding bromination step ensures a clean conversion to Compound 11 with minimal poly-brominated byproducts. The subsequent steps, including the Tos protection and the final Buchwald-Hartwig coupling (which does use Pd but in a controlled, late-stage manner), are designed to maintain this high level of purity. The final product, Compound 5, is obtained with a purity of >99%, demonstrating the effectiveness of this mechanistic pathway in delivering high-purity pharmaceutical intermediates suitable for sensitive downstream coupling reactions.
How to Synthesize 5-Tosyl-5H-pyrrolo[2,3-b]pyrazine-2-carbamate Efficiently
The synthesis of this key Upadacitinib intermediate is a multi-step process that requires precise control over reaction conditions to maximize yield and safety. The patented route outlines a clear progression from a simple ester to the complex protected heterocycle. The process begins with the reduction of the ester to an alcohol, followed by oxidation to an aldehyde, setting the stage for the crucial carbon-carbon bond formation. Each step has been optimized for industrial feasibility, utilizing common solvents like THF, DCM, and ethanol, and reagents that are manageable on a multi-kilogram scale. The detailed标准化 synthesis steps见下方的指南 provide a roadmap for replicating this high-efficiency pathway in a GMP environment.
- Reduce methyl 3-aminopyrazine-2-carboxylate (Compound 8) to the corresponding alcohol (Compound 9) using sodium borohydride and a Lewis acid.
- Oxidize Compound 9 to the aldehyde (Compound 10) using manganese dioxide, followed by bromination to form Compound 11.
- Perform a Wittig reaction on Compound 11 to generate the vinyl ether (Compound 12), followed by acid-catalyzed cyclization to form the pyrrolopyrazine core (Compound 3).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic route described in CN116396299A offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By shifting away from specialized dibromo-precursors to commodity chemicals like methyl 3-aminopyrazine-2-carboxylate, manufacturers can mitigate supply risks and leverage better pricing from bulk chemical suppliers. This change significantly enhances supply chain reliability, ensuring that production schedules are not disrupted by the scarcity of niche starting materials. Furthermore, the elimination of hazardous reagents like sodium hydride reduces the regulatory burden and safety compliance costs associated with storage and handling, leading to substantial indirect cost savings.
- Cost Reduction in Manufacturing: The new route delivers significant economic value by removing the need for expensive palladium and copper catalysts in the early stages of synthesis. In traditional methods, these precious metals represent a major cost center, and their removal requires additional purification steps like scavenging, which adds time and expense. By replacing the Sonogashira coupling with a Wittig reaction using inexpensive phosphonium salts, the direct material cost is lowered. Additionally, the doubling of the overall yield from 18% to 40% means that less raw material is wasted per kilogram of final product, effectively halving the material cost basis for the intermediate. This efficiency gain translates directly to improved margins and a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a more resilient supply chain. Methyl 3-aminopyrazine-2-carboxylate is a standard building block with multiple global suppliers, reducing the risk of single-source dependency. Moreover, the simplified process flow, which avoids complex cryogenic conditions or strictly anhydrous environments required for sodium hydride reactions, allows for manufacturing in a broader range of facilities. This flexibility increases the available capacity in the market, reducing lead times and ensuring consistent delivery of the intermediate to downstream API manufacturers. The robustness of the acid-cyclization step also means fewer batch failures due to operational variances, further stabilizing the supply.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is vastly superior. The avoidance of sodium hydride eliminates the generation of hydrogen gas and the associated explosion risks, making the process inherently safer for scale-up to 100 MT levels. The reduction in three-waste treatment volume, particularly by avoiding large amounts of elemental bromine and heavy metal residues, aligns with stricter global environmental regulations. This compliance reduces the cost of waste disposal and minimizes the risk of production shutdowns due to environmental audits. The process is designed to be green and sustainable, which is increasingly a prerequisite for partnerships with top-tier pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Upadacitinib intermediates using this novel methodology. These insights are derived directly from the technical specifications and experimental data provided in patent CN116396299A, offering clarity on the feasibility and advantages of this route for potential partners and licensees.
Q: What are the safety advantages of this new synthetic route compared to prior art?
A: The new route eliminates the use of sodium hydride (NaH) for ring closure, which is a pyrophoric and hazardous reagent difficult to handle on an industrial scale. It also avoids the use of large amounts of elemental bromine in grinding processes, significantly reducing safety risks and waste treatment burdens.
Q: How does this method improve cost efficiency for Upadacitinib production?
A: By utilizing methyl 3-aminopyrazine-2-carboxylate as a starting material instead of expensive dibromo-precursors, and by avoiding precious metal catalysts like Palladium and Copper in the early Sonogashira coupling steps, the overall raw material and catalyst costs are substantially reduced.
Q: What is the expected purity and yield of the final intermediate?
A: According to patent CN116396299A, the total yield of the 7-step synthetic route is approximately 40%, with the final product achieving a high purity of greater than 99%, making it suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Tosyl-5H-pyrrolo[2,3-b]pyrazine-2-carbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. The technology described in CN116396299A aligns perfectly with our commitment to delivering high-value intermediates through innovative chemistry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe management of Wittig reagents and acid-catalyzed cyclizations, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs utilize advanced analytical techniques to verify the >99% purity of the final intermediate, guaranteeing quality that supports your regulatory filings.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your Upadacitinib projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this essential intermediate, driving your project forward with confidence and efficiency.
