Scalable Green Synthesis of Chiral Bicyclic Intermediates via Aqueous Organocatalysis
Scalable Green Synthesis of Chiral Bicyclic Intermediates via Aqueous Organocatalysis
The pharmaceutical and fine chemical industries are increasingly demanding sustainable manufacturing processes that do not compromise on stereochemical purity or yield. Patent CN101525291A introduces a groundbreaking green asymmetric synthesis method for producing chiral bicyclic compounds, which serve as critical precursors in the development of complex active pharmaceutical ingredients (APIs). This technology leverages an aqueous-phase organocatalytic Diels-Alder reaction, utilizing chiral secondary amine catalysts derived from proline to achieve exceptional enantioselectivity under mild conditions. By shifting away from traditional volatile organic solvents to water, this method addresses both environmental concerns and cost-efficiency, positioning it as a highly attractive route for reliable pharmaceutical intermediate supplier networks seeking to modernize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral bicyclic scaffolds has relied heavily on stoichiometric chiral auxiliaries or expensive transition metal catalysts operating in hazardous organic solvents. Traditional Diels-Alder reactions often require strict anhydrous conditions and cryogenic temperatures to maintain stereocontrol, leading to significant energy consumption and complex waste streams. Furthermore, the removal of heavy metal residues from the final product adds substantial downstream processing costs and regulatory hurdles for API manufacturers. Previous organocatalytic approaches, while metal-free, frequently suffered from low reaction rates in non-polar solvents or required high catalyst loadings that were economically unviable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The innovative methodology described in the patent overcomes these barriers by employing water as the primary reaction medium, which acts not merely as a solvent but as a promoter for reactivity and selectivity. The process utilizes readily available cyclohexenone derivatives and nitroolefin derivatives reacting in the presence of a specific proline-based chiral secondary amine catalyst and a protonic acid co-catalyst. This system operates effectively at temperatures ranging from -20°C to 40°C, drastically reducing energy requirements compared to cryogenic protocols. The use of water facilitates the "on-water" effect, where hydrophobic interactions accelerate the reaction rate while the hydrogen-bonding network stabilizes the transition state, leading to superior diastereo- and enantioselectivity without the need for toxic organic media.
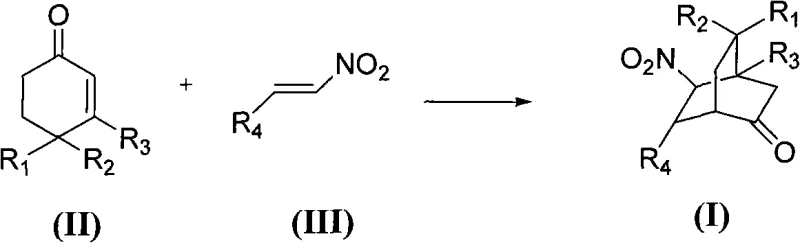
This reaction pathway transforms simple starting materials into highly functionalized chiral bicyclic ketones with multiple stereocenters established in a single step. The versatility of the substrate scope allows for various substituents on the aromatic rings, including electron-donating and electron-withdrawing groups, making it a robust platform for generating diverse chemical libraries. By eliminating the need for inert atmospheres and specialized dry solvents, this approach significantly simplifies the operational complexity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where supply chain simplicity is paramount.
Mechanistic Insights into Proline-Catalyzed Asymmetric Diels-Alder Reaction
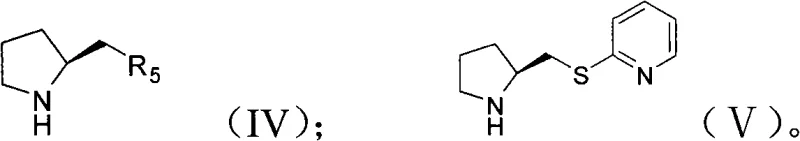
The core of this technological advancement lies in the precise design of the chiral secondary amine catalyst, specifically the proline derivatives represented by formula (IV) and preferably formula (V). These catalysts function through an enamine activation mechanism, where the secondary amine condenses with the ketone substrate to form a nucleophilic enamine intermediate. This activation lowers the LUMO energy of the dienophile or raises the HOMO energy of the diene, facilitating the cycloaddition. The chiral environment provided by the pyrrolidine ring and the bulky substituents directs the approach of the nitroolefin, ensuring that the new carbon-carbon bonds are formed with high facial selectivity. The presence of water is crucial here, as it helps organize the transition state through hydrogen bonding, further tightening the stereochemical control.
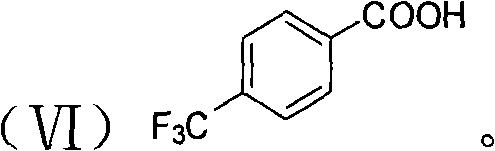
Complementing the amine catalyst is the role of the protonic acid co-catalyst, such as trifluoromethylbenzoic acid (formula VI), which protonates the nitro group of the electrophile to increase its reactivity. This dual-activation strategy ensures that the reaction proceeds efficiently even at ambient temperatures. The synergy between the chiral amine and the Brønsted acid creates a highly organized catalytic pocket that minimizes the formation of unwanted byproducts and regioisomers. Impurity control is inherently built into the mechanism; the high specificity of the organocatalyst means that side reactions like polymerization or non-selective background reactions are suppressed, resulting in a cleaner crude reaction profile that requires less intensive purification.
Furthermore, the mechanistic robustness allows for a broad tolerance of functional groups, which is essential for late-stage functionalization in drug discovery. The ability to tune the electronic properties of the catalyst and the acid co-catalyst provides chemists with fine control over the reaction outcome. This level of mechanistic understanding translates directly to process reliability, as the reaction parameters are well-defined and reproducible. For R&D teams, this means that scaling from milligram to kilogram quantities does not introduce unpredictable variables, ensuring that the high enantiomeric excess values observed in the lab (often >90% ee) are maintained during commercial production.
How to Synthesize Chiral Bicyclic Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this green chemistry solution in a production setting. The process begins with the preparation of the reaction mixture containing the cyclohexenone derivative, the nitroolefin derivative, the chiral catalyst, and the acid additive in water. The mixture is then stirred at a controlled temperature, typically around 25°C, for a duration of 12 to 36 hours depending on the specific substrate reactivity. Upon completion, the product is isolated via simple extraction with ethyl acetate, followed by solvent removal and purification using standard column chromatography techniques. This straightforward workup procedure avoids complex distillation or crystallization steps often required in traditional organic synthesis.
- Mix cyclohexenone derivatives and nitroolefin derivatives in water with a chiral secondary amine catalyst and protonic acid.
- Maintain the reaction temperature between -20°C and 40°C for 3 to 72 hours to ensure optimal stereoselectivity.
- Extract the product with ethyl acetate, remove solvent, and purify via column chromatography to obtain the target chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this aqueous organocatalytic process represents a strategic opportunity to optimize both cost structures and supply resilience. The elimination of expensive, anhydrous organic solvents and the reduction in energy consumption due to mild reaction temperatures directly contribute to significant cost savings in manufacturing operations. Moreover, the use of water as a solvent removes the safety hazards associated with flammable volatile organic compounds (VOCs), lowering insurance premiums and facility compliance costs. The simplicity of the raw material sourcing, relying on commodity chemicals like cyclohexenones and nitroolefins, ensures a stable and continuous supply chain free from the geopolitical risks often associated with rare earth metals or specialized reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the reaction medium and workup procedures. By replacing costly organic solvents with water, the expense of solvent purchase, recovery, and disposal is virtually eliminated. Additionally, the high catalytic efficiency means that lower loading of the chiral catalyst is required to achieve high yields, reducing the cost per kilogram of the final API intermediate. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, thereby reducing the burden on downstream purification units and increasing overall throughput capacity without additional capital investment.
- Enhanced Supply Chain Reliability: Adopting this synthetic route mitigates risks associated with the availability of specialized reagents. The catalysts and substrates are derived from abundant feedstocks, ensuring that production schedules are not disrupted by raw material shortages. The robustness of the aqueous system also means that the process is less sensitive to minor fluctuations in environmental conditions, such as humidity, which can plague moisture-sensitive traditional reactions. This reliability translates to more predictable lead times for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to plan their inventory with greater confidence and reduce safety stock levels.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with global sustainability goals. The absence of heavy metals and halogenated solvents simplifies waste treatment and discharge compliance, avoiding the hefty fines and remediation costs associated with toxic effluent. The process is inherently scalable; the exothermic nature of the reaction is easily managed in water due to its high heat capacity, allowing for safe scale-up from pilot plants to multi-ton commercial reactors. This scalability ensures that as demand for the chiral intermediate grows, the supply can be expanded rapidly without the need for extensive process re-engineering or new regulatory filings for hazardous materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the key advantages of using water as a solvent in this asymmetric synthesis?
A: Using water significantly reduces costs associated with organic solvents and simplifies waste treatment. Additionally, water can enhance reaction rates and selectivity through hydrophobic effects and hydrogen bonding interactions.
Q: What level of enantiomeric excess (ee) can be achieved with this method?
A: The patent data demonstrates that this method can achieve high enantiomeric excess values, often exceeding 90% ee, with specific examples reaching up to 96% ee under optimized conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of mild temperatures (-20 to 40°C), inexpensive catalysts, and water as a green solvent makes this process highly scalable and compliant with modern environmental regulations for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bicyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green organocatalysis in modern drug development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high standards of purity and stereochemical integrity defined in the lab are met in the plant. We possess stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the quality of every batch. Our commitment to sustainability mirrors the principles of this patent, making us an ideal partner for companies looking to produce high-purity pharmaceutical intermediates with a reduced environmental footprint.
We invite you to discuss how our manufacturing capabilities can support your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By collaborating with us, you gain access to a supply chain that prioritizes innovation, quality, and long-term partnership, ensuring your drug development pipeline remains uninterrupted and cost-effective.
