Advanced Green Oxidation Technology for High-Purity 2-Nitro-4-Methylsulfonyl Benzoic Acid Production
Advanced Green Oxidation Technology for High-Purity 2-Nitro-4-Methylsulfonyl Benzoic Acid Production
The global agrochemical industry is constantly seeking more sustainable and efficient pathways for producing critical intermediates, and patent CN108530326B presents a groundbreaking solution for the synthesis of 2-nitro-4-methylsulfonyl benzoic acid. This compound serves as a pivotal precursor for mesotrione, a widely utilized HPPD-inhibitor herbicide that has gained significant traction in global agriculture due to its efficacy against broadleaf weeds. The traditional manufacturing landscape for this intermediate has long been plagued by severe environmental challenges and high operational costs, primarily driven by the reliance on stoichiometric oxidants and toxic heavy metal catalysts. This new patented methodology introduces a catalytic aerobic oxidation system that utilizes molecular oxygen as the terminal oxidant, fundamentally shifting the paradigm towards greener chemistry. By leveraging N-hydroxylamine compounds in conjunction with nitrate co-catalysts, the process achieves high conversion rates under moderate conditions, effectively addressing the pain points of waste generation and equipment corrosion that have historically burdened producers. For R&D directors and procurement strategists alike, this technology represents a viable pathway to secure a more robust and compliant supply chain for high-value agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-nitro-4-methylsulfonyl benzoic acid has relied heavily on oxidation methods that pose significant environmental and safety risks. One prevalent approach involves the use of heavy metal salt oxidants, such as potassium dichromate, which, while effective in terms of yield, generates substantial quantities of hazardous heavy metal waste that requires complex and costly disposal procedures. Another common industrial method utilizes nitric acid as the oxidant, often catalyzed by vanadium pentoxide; although nitric acid is inexpensive, this route produces vast amounts of waste acid and nitrogen oxides, creating immense pressure on wastewater treatment facilities and increasing the risk of equipment corrosion. Furthermore, alternative methods using hydrogen peroxide often suffer from low atom economy and poor yields due to the excessive consumption of the oxidant, while air oxidation methods employing cobalt acetate or metalloporphyrins have historically struggled with low oxidation efficiency and poor selectivity. These conventional limitations result in inflated production costs, regulatory compliance burdens, and potential supply disruptions due to stringent environmental inspections, making the search for a superior alternative not just desirable but essential for long-term business continuity.
The Novel Approach
The innovative process described in patent CN108530326B overcomes these historical deficiencies by employing a catalytic system driven by molecular oxygen, which is both abundant and environmentally benign. This method utilizes 2-nitro-4-methylsulfonyltoluene as the starting material and employs aliphatic carboxylic acids, such as acetic acid, as the reaction medium, creating a homogeneous system that facilitates efficient mass transfer. The core of this breakthrough lies in the synergistic combination of N-hydroxylamine catalysts and nitrate co-catalysts, which together activate molecular oxygen to selectively oxidize the methyl group on the aromatic ring to a carboxylic acid group. This approach completely eliminates the need for toxic heavy metal catalysts like vanadium pentoxide and drastically reduces the generation of inorganic waste salts and acidic effluents. Moreover, the process allows for the recycling of the solvent system, further enhancing the economic viability and sustainability of the operation. By operating at temperatures between 60-180°C and pressures of 0.1-5 MPa, the method balances reaction kinetics with safety, offering a scalable solution that aligns perfectly with modern green chemistry principles and the increasing demand for cost reduction in agrochemical manufacturing.
Mechanistic Insights into N-Hydroxylamine Catalyzed Aerobic Oxidation
The success of this synthesis route hinges on the unique radical mechanism facilitated by the N-hydroxylamine catalysts, specifically the formation of the phthalimide-N-oxyl (PINO) radical. As illustrated in the structural diversity of the catalysts provided in the patent, compounds containing the N-OH moiety, such as N-hydroxyphthalimide (NHPI) and its derivatives, play a critical role in initiating the oxidation cycle. Under the reaction conditions, the N-hydroxylamine catalyst undergoes a single electron transfer or hydrogen abstraction to generate the stable PINO radical species. This radical is highly electrophilic and capable of abstracting a hydrogen atom from the benzylic position of the 2-nitro-4-methylsulfonyltoluene substrate, generating a carbon-centered radical on the methyl group. This step is the rate-determining phase of the reaction, and the presence of the electron-withdrawing nitro and sulfonyl groups on the aromatic ring typically makes this hydrogen abstraction difficult; however, the high reactivity of the PINO radical overcomes this electronic deactivation.
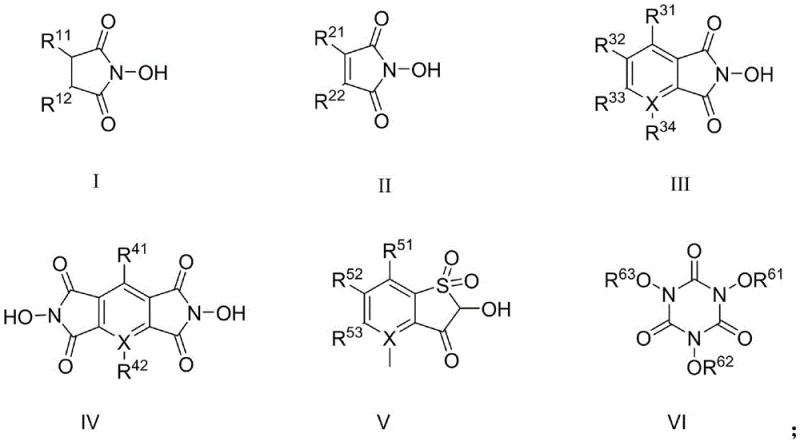
Once the benzylic radical is formed, it rapidly reacts with molecular oxygen dissolved in the solvent to form a peroxy radical, which subsequently undergoes further transformation through the assistance of the nitrate co-catalyst to yield the final carboxylic acid product while regenerating the N-hydroxylamine catalyst. The inclusion of nitric acid, nitrates, or nitrites as co-catalysts is crucial, as they facilitate the decomposition of intermediate hydroperoxides and accelerate the turnover of the catalytic cycle, preventing the accumulation of inactive species. This mechanistic pathway ensures high selectivity for the desired carboxylic acid, minimizing the formation of over-oxidized byproducts or incomplete oxidation intermediates like aldehydes or alcohols. For R&D teams, understanding this mechanism is vital for optimizing reaction parameters such as oxygen pressure and catalyst loading to maximize yield and purity, ensuring that the final product meets the stringent specifications required for downstream herbicide synthesis.
How to Synthesize 2-Nitro-4-Methylsulfonyl Benzoic Acid Efficiently
Implementing this advanced oxidation protocol requires precise control over reaction parameters to ensure optimal performance and safety. The process begins with charging a pressure reactor with the substrate, 2-nitro-4-methylsulfonyltoluene, and the solvent, typically glacial acetic acid or a mixture with water, followed by the addition of the N-hydroxylamine catalyst and the nitrate co-catalyst. The detailed standardized synthesis steps involve pressurizing the system with oxygen or air to the specified range of 0.1-5 MPa and heating the mixture to a temperature between 60-180°C while maintaining vigorous stirring to ensure adequate gas-liquid contact. Reaction times can vary from 0.5 to 15 hours depending on the specific catalyst variant and conditions employed, after which the mixture is cooled, filtered, and the solid product is isolated through alkaline dissolution and subsequent acidification.
- Charge the reactor with 2-nitro-4-methylsulfonyltoluene, aliphatic carboxylic acid solvent, N-hydroxylamine catalyst, and nitrate co-catalyst.
- Pressurize the system with molecular oxygen or air to 0.1-5 MPa and heat to 60-180°C while stirring.
- After reaction completion, cool, filter, wash the filter cake, dissolve in alkali, filter again, and acidify the filtrate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers transformative benefits that extend far beyond simple chemical synthesis. The shift from stoichiometric oxidants and toxic heavy metals to a catalytic aerobic system fundamentally alters the cost structure of production by eliminating the procurement of expensive and hazardous reagents like vanadium pentoxide and potassium dichromate. This transition not only reduces the direct cost of raw materials but also significantly lowers the indirect costs associated with waste disposal, environmental compliance, and worker safety protocols. The ability to recycle the acetic acid solvent further amplifies these savings, as it reduces the volume of fresh solvent required per batch and minimizes the waste stream that needs treatment. Consequently, manufacturers can achieve a more predictable and stable cost base, insulating their operations from the volatility of raw material markets and regulatory changes.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts and the reduction of waste acid generation lead to substantial operational expenditure savings. By avoiding the complex purification steps required to remove trace heavy metals from the final product, the downstream processing becomes simpler and less energy-intensive. Furthermore, the use of molecular oxygen, which can be sourced from air or standard industrial oxygen supplies, is vastly more economical than purchasing specialized chemical oxidants. The qualitative improvement in atom economy means that less raw material is wasted, directly translating to a lower cost per kilogram of the final active intermediate, thereby enhancing the overall margin profile for the manufacturing entity.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like acetic acid, nitric acid, and air/oxygen ensures a robust supply chain that is less susceptible to disruptions compared to sourcing specialized heavy metal salts or unstable peroxides. The simplified logistics of handling non-hazardous oxidants reduce the regulatory burden on transportation and storage, allowing for more flexible inventory management. Additionally, the high purity of the product achieved through this selective catalytic method reduces the risk of batch rejection by downstream customers, ensuring a consistent flow of revenue and strengthening relationships with key clients in the agrochemical sector who demand reliable quality.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in autoclave systems that mimic industrial high-pressure reactors, making the transition from pilot plant to commercial production seamless. The drastic reduction in three-waste emissions (waste water, waste gas, and solid waste) aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing facility against potential shutdowns due to non-compliance. This green credential is increasingly becoming a competitive advantage in B2B negotiations, as multinational corporations prioritize suppliers who can demonstrate a commitment to sustainable manufacturing practices and reduced carbon footprints.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green oxidation technology. These insights are derived directly from the experimental data and claims presented in patent CN108530326B, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this process into their existing production lines or sourcing strategies.
Q: What are the primary advantages of this aerobic oxidation method over traditional nitric acid oxidation?
A: This method eliminates the use of highly toxic vanadium pentoxide catalysts and reduces the generation of large amounts of waste acid associated with traditional nitric acid oxidation, significantly lowering environmental compliance costs and improving operator safety.
Q: Can the solvent be recycled in this process to reduce production costs?
A: Yes, the patent explicitly describes a solvent recovery process where the filtrate is treated by adding anhydrous or high-concentration solvent to restore the predetermined concentration, allowing for repeated reuse and substantial reduction in raw material consumption.
Q: What represents the key catalytic system in this synthesis route?
A: The core innovation lies in the use of N-hydroxylamine compounds, such as N-hydroxyphthalimide (NHPI) and its derivatives, combined with nitric acid or metal nitrates as co-catalysts to activate molecular oxygen for efficient side-chain oxidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Methylsulfonyl Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN108530326B to maintain competitiveness in the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is smooth and efficient. We are committed to delivering high-purity 2-nitro-4-methylsulfonyl benzoic acid that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle complex catalytic oxidations safely, leveraging our expertise in high-pressure reactions to maximize yield and minimize impurities.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our implementation of this green technology can drive value for your organization. Let us help you secure a sustainable and cost-effective source of high-quality agrochemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
