Advanced Organocatalytic Synthesis of Chromanone Spiro Indole Intermediates for Oncology
Advanced Organocatalytic Synthesis of Chromanone Spiro Indole Intermediates for Oncology
The relentless pursuit of novel therapeutic agents in the pharmaceutical industry often hinges on the ability to efficiently construct complex heterocyclic scaffolds that mimic natural product architectures. A significant breakthrough in this domain is documented in patent CN111875612A, which discloses a sophisticated class of chromanone splicing pyrrole spiro epoxidized indole skeleton compounds bearing trifluoromethyl groups. These molecules represent a convergence of three distinct pharmacophores known for their biological potency, offering a rich chemical space for drug discovery programs focused on oncology. The strategic fusion of a chromanone backbone with a pyrrole spiro-oxindole core, further functionalized with a trifluoromethyl moiety, creates a privileged structure capable of interacting with diverse biological targets. This technical insight report analyzes the synthetic methodology and commercial viability of producing these high-value intermediates, providing critical intelligence for R&D directors and procurement strategists seeking reliable sources for advanced pharmaceutical building blocks.
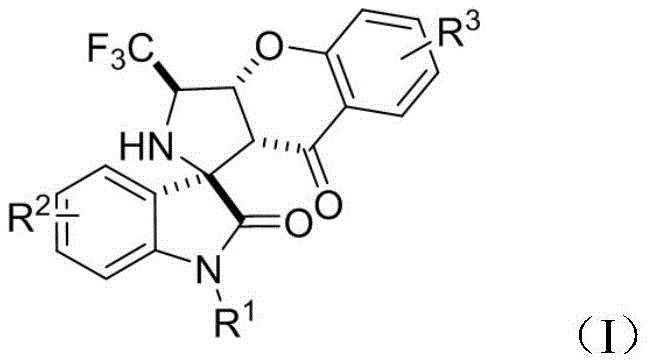
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of spiro-oxindole derivatives containing multiple fused rings has relied heavily on transition metal catalysis or harsh reaction conditions that pose significant challenges for large-scale manufacturing. Conventional routes often necessitate the use of expensive palladium or rhodium catalysts, which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding residual metal limits in final drug substances. Furthermore, many classical methods require elevated temperatures or inert atmospheres, increasing energy consumption and operational complexity. The purification of products from these reactions frequently involves tedious chromatographic separations to remove metal residues and side products, leading to reduced overall yields and extended production timelines. For supply chain managers, these factors translate into higher volatility in pricing and potential bottlenecks in securing consistent quality material for clinical trials. The reliance on moisture-sensitive reagents in older methodologies also compromises the robustness of the process, making technology transfer from lab to plant a risky endeavor.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent utilizes a metal-free organocatalytic strategy that fundamentally reshapes the economic and operational landscape of synthesizing these complex intermediates. The core transformation involves a [3+2] cycloaddition reaction between 3-trifluoromethylmethyleneimine substituted oxindoles and 3-formyl chromones, mediated by simple organic small molecule tertiary amine catalysts. As illustrated in the reaction scheme below, this approach leverages the nucleophilic activation of the dipole by catalysts such as DMAP, enabling the reaction to proceed smoothly at room temperature. This mild condition profile eliminates the need for specialized high-pressure equipment or cryogenic cooling, drastically simplifying the engineering requirements for production vessels. The use of commodity chemicals like chloroform as the solvent further enhances the accessibility of the process, allowing for straightforward scale-up without the need for exotic reagents. By avoiding transition metals entirely, the downstream processing is significantly streamlined, as there is no requirement for costly metal scavenging resins or additional washing steps to meet ICH Q3D guidelines.
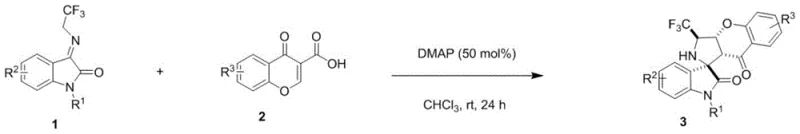
Mechanistic Insights into DMAP-Catalyzed [3+2] Cycloaddition
The success of this synthetic route lies in the precise mechanistic orchestration of the 1,3-dipolar cycloaddition, which is elegantly facilitated by the organic base catalyst. The reaction initiates with the deprotonation or nucleophilic activation of the 3-trifluoromethylmethyleneimine substituted oxindole, generating a reactive 1,3-dipole species in situ. This dipole then engages with the electron-deficient double bond of the 3-formyl chromone in a concerted manner, forming two new sigma bonds and establishing the intricate spirocyclic framework in a single operational step. The stereochemical outcome of the reaction is controlled by the spatial arrangement of the transition state, where the catalyst likely stabilizes specific conformers through hydrogen bonding or ion-pairing interactions, leading to high diastereoselectivity as evidenced by the reported dr values ranging from 5:1 to 15:1. Understanding this mechanism is crucial for R&D teams aiming to optimize the process further, as it highlights the importance of catalyst loading and solvent polarity in maintaining the integrity of the dipole. The presence of the trifluoromethyl group adds an electronic dimension to the reactivity, enhancing the electrophilicity of the dipole and driving the cycloaddition forward with favorable kinetics even under ambient conditions.
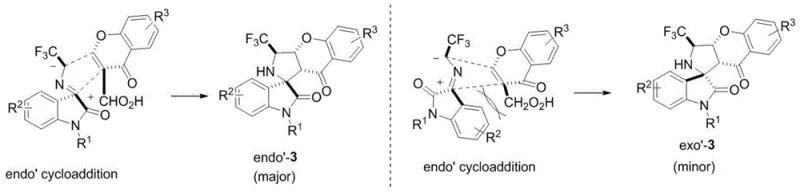
From an impurity control perspective, the organocatalytic nature of this transformation offers distinct advantages over radical or metal-mediated pathways that often generate complex mixtures of byproducts. The primary impurities typically arise from unreacted starting materials or minor diastereomers, both of which are chemically similar to the target product and can be effectively managed through standard crystallization or chromatographic techniques. The absence of metal-induced side reactions, such as homocoupling or over-reduction, ensures a cleaner crude reaction profile, which directly correlates to higher isolated yields and reduced waste generation. For quality assurance professionals, this means that the impurity profile is predictable and manageable, facilitating faster method validation and regulatory filing. The robustness of the mechanism against varying substituents on the aromatic rings (R1, R2, R3) demonstrates the versatility of the chemistry, allowing for the rapid generation of analog libraries for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new derivative.
How to Synthesize Chromanone Spiro Indole Efficiently
Implementing this synthesis in a practical setting requires careful attention to stoichiometry and mixing efficiency to maximize the formation of the desired spirocyclic product. The standard protocol involves dissolving the oxindole dipole and the chromone dipolarophile in a suitable organic solvent, followed by the addition of the tertiary amine catalyst. Maintaining a homogeneous reaction mixture is critical to ensure uniform exposure of the substrates to the catalyst, preventing localized concentration gradients that could lead to polymerization or decomposition. While the reaction proceeds at room temperature, monitoring the progress via TLC or HPLC is recommended to determine the optimal endpoint, typically achieved within 24 to 72 hours depending on the electronic nature of the substituents. Detailed standardized synthesis steps see the guide below.
- Combine 3-trifluoromethylmethyleneimine substituted oxindole and 3-formyl chromone in an organic solvent such as chloroform.
- Add an organic small molecule tertiary amine catalyst, specifically DMAP at 50 mol%, to the reaction mixture.
- Stir the reaction at room temperature for 1 to 3 days until completion, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route presents a compelling value proposition centered on cost stability and operational simplicity. The elimination of precious metal catalysts removes a major source of price volatility from the bill of materials, as the cost of DMAP or similar organic bases is negligible compared to palladium or platinum complexes. This shift not only reduces direct material costs but also mitigates the supply risk associated with geopolitically sensitive metals, ensuring a more resilient supply chain for critical intermediates. Furthermore, the ability to run the reaction at room temperature significantly lowers energy expenditures related to heating or cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. The simplified workup procedure, devoid of metal scavenging steps, reduces the consumption of auxiliary materials and shortens the batch cycle time, effectively increasing the throughput of existing manufacturing assets without requiring capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of transition metals, which eradicates the need for expensive purification technologies like scavenger resins or specialized filtration units. By utilizing commodity solvents such as chloroform and inexpensive organocatalysts, the variable cost per kilogram of the intermediate is substantially lowered, allowing for more competitive pricing in the final API market. Additionally, the high atom economy of the [3+2] cycloaddition minimizes waste generation, reducing the costs associated with solvent recovery and hazardous waste disposal. This lean manufacturing approach ensures that resources are focused on value-added transformation rather than remediation of process inefficiencies.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials and reagents ensures that production schedules are not disrupted by shortages of niche chemicals. Since the reaction conditions are mild and tolerant to various functional groups, the process is robust against minor variations in raw material quality, reducing the rejection rate of incoming shipments. This reliability translates into shorter lead times for customers, as manufacturers can maintain consistent inventory levels without the fear of batch failures due to sensitive reaction parameters. The scalability of the process from gram to ton scale is proven by the simplicity of the unit operations involved, providing confidence to supply chain planners that demand surges can be met without compromising quality.
- Scalability and Environmental Compliance: Operating at ambient pressure and temperature inherently reduces the safety risks associated with high-energy processes, simplifying the permitting and compliance landscape for manufacturing facilities. The absence of heavy metals simplifies the environmental impact assessment, as there is no risk of soil or water contamination from metal effluents, making it easier to obtain regulatory approvals in jurisdictions with strict environmental laws. The process generates minimal hazardous waste, primarily consisting of organic solvents that can be readily recycled, supporting a circular economy model within the chemical plant. This alignment with green chemistry principles not only enhances the corporate image but also future-proofs the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chromanone spiro indole intermediates. These insights are derived directly from the patent specifications and practical manufacturing experience, aiming to clarify the feasibility and benefits of this technology for potential partners. Understanding these details is essential for making informed decisions about integrating this scaffold into your drug discovery pipeline.
Q: What are the advantages of using DMAP over transition metal catalysts for this synthesis?
A: Using DMAP eliminates the risk of heavy metal contamination, which is critical for pharmaceutical intermediates. It simplifies the purification process by removing the need for expensive metal scavenging steps, thereby reducing overall production costs and environmental impact.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the reaction operates under mild conditions (room temperature) and uses readily available solvents like chloroform. The absence of sensitive reagents and the robustness of the organocatalyst make it highly suitable for scale-up from laboratory to industrial manufacturing.
Q: What is the biological significance of the chromanone spiro indole scaffold?
A: This scaffold combines three bioactive motifs: chromanone, spiro-oxindole, and trifluoromethyl groups. Patent data indicates significant inhibitory activity against human leukemia cells (K562), making it a valuable lead structure for developing new anti-tumor agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromanone Spiro Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing high-quality, complex intermediates like the chromanone spiro indole scaffold for advancing oncology research. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage discovery through to late-stage clinical supply. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to navigate the complexities of organocatalytic processes, delivering materials with consistent stereochemistry and low impurity profiles.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these critical building blocks. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this metal-free synthesis route. We are prepared to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our capabilities with your development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
