Advanced Total Synthesis of Houttuynoid Hybrid Flavonoids for Commercial Scale-Up
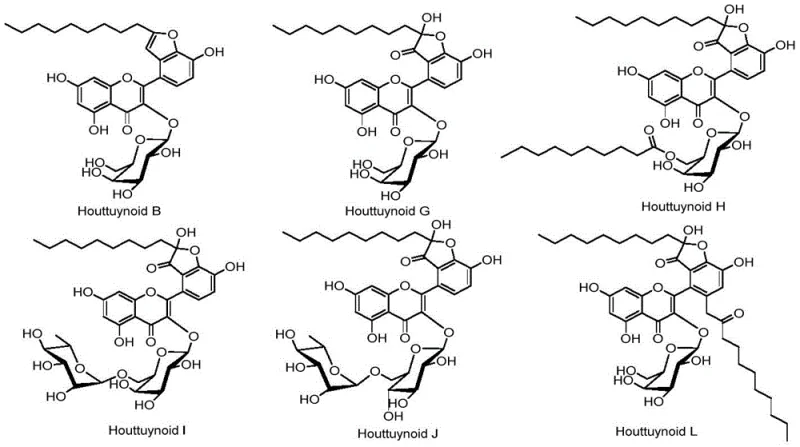
The pharmaceutical industry is constantly seeking reliable sources for complex bioactive molecules, particularly those with potent antiviral properties. Patent CN108047291B discloses a groundbreaking synthetic method for houttuynin sodium bisulfite heterozygous flavonoid compounds, specifically targeting the Houttuynoid family including Houttuynoid B, G, H, I, J, and L. These natural products, originally isolated from Houttuynia cordata, have demonstrated significant anti-herpes virus (HSV) activity, acting on viral infection stages potentially involving envelope proteins gB, gD, gH, and gL. However, relying on natural extraction presents severe limitations regarding yield, seasonal availability, and purification complexity. This patent provides a robust, ten-step total synthesis strategy that transforms simple aromatic aldehydes and alkynes into these high-value pharmaceutical intermediates. By establishing a fully synthetic pathway, manufacturers can bypass the bottlenecks of agricultural sourcing, ensuring a stable supply chain for drug development programs focused on novel antiviral mechanisms distinct from existing nucleoside analogs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining Houttuynoid hybrid flavonoids required extensive extraction and isolation from the medicinal plant Houttuynia cordata. This biological sourcing method is inherently unstable, subject to variations in plant growth conditions, harvest seasons, and geographical origin, leading to inconsistent batch-to-batch quality. Furthermore, the concentration of these specific hybrid flavonoids in the raw plant material is often trace-level, necessitating the processing of massive quantities of biomass to obtain milligram amounts of pure compound. The downstream purification involves complex chromatographic separations to remove structurally similar impurities and other plant metabolites, which drastically increases the cost of goods sold (COGS) and extends the lead time for research materials. For pharmaceutical companies aiming to conduct preclinical or clinical trials, this reliance on natural extraction creates a significant supply chain risk, where demand cannot be met by the slow rate of natural accumulation, effectively stalling drug development pipelines due to material shortages.
The Novel Approach
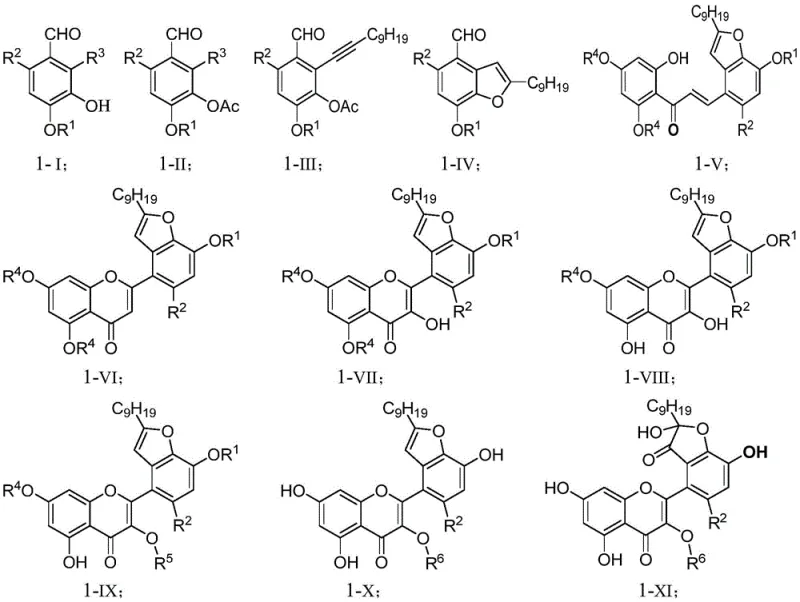
The patented synthetic route offers a transformative solution by constructing the complex houttuynoid skeleton from readily available chemical building blocks through a convergent and linear sequence. As illustrated in the general synthetic scheme, the process begins with the functionalization of a benzaldehyde derivative, followed by a palladium-catalyzed Sonogashira coupling to introduce the undecyne side chain, which mimics the houttuynin moiety. Subsequent steps involve a base-mediated condensation to form the chalcone intermediate, followed by a critical iodine-mediated oxidative cyclization in DMSO to construct the flavonoid core. This chemical approach allows for precise control over stereochemistry and substitution patterns, enabling the synthesis of specific analogues like Houttuynoid G and H with high purity. By shifting from extraction to synthesis, manufacturers gain the ability to scale production based on market demand rather than harvest cycles, significantly reducing the cost reduction in pharmaceutical intermediate manufacturing and securing the supply of these critical antiviral candidates.
Mechanistic Insights into Pd-Catalyzed Coupling and Oxidative Cyclization
The success of this synthesis hinges on two pivotal mechanistic events: the formation of the carbon-carbon triple bond linkage and the construction of the heterocyclic flavonoid ring system. The Sonogashira coupling step utilizes a dual catalyst system of bis(triphenylphosphine)palladium(II) chloride and cuprous iodide to couple the aryl iodide with 1-undecyne. This reaction proceeds under mild conditions (room temperature) with high efficiency, creating the lipophilic tail essential for the biological activity of the final molecule. Following this, the chalcone intermediate undergoes an iodine-catalyzed oxidative cyclization in dimethyl sulfoxide (DMSO) at elevated temperatures (110°C). In this transformation, iodine acts as a mild oxidant to facilitate the intramolecular attack of the phenolic hydroxyl group onto the enone system, closing the pyrone ring to form the flavone backbone. This method avoids the use of harsh acidic conditions often required for traditional flavonoid synthesis, thereby preserving sensitive functional groups and minimizing side reactions that could generate difficult-to-remove impurities.
Impurity control is further enhanced in the later stages of the synthesis, particularly during the glycosylation and final oxidation steps. The introduction of the sugar moiety is achieved via nucleophilic substitution using a specific bromosugar intermediate under basic conditions, which ensures high regioselectivity for the glycosidic bond formation. The final step involves the oxidation of the methyl group to a lactone using osmium tetroxide (OsO4) and N-methylmorpholine N-oxide (NMO). This catalytic oxidation is highly selective, preventing over-oxidation of the electron-rich flavonoid rings. The use of buffered conditions during the earlier oxone oxidation step (pH 9-12) also prevents the degradation of the sensitive furan ring system. These carefully optimized reaction parameters ensure that the final product meets stringent purity specifications required for biological testing, reducing the burden on downstream purification teams and increasing the overall yield of the process.
How to Synthesize Houttuynoid G Efficiently
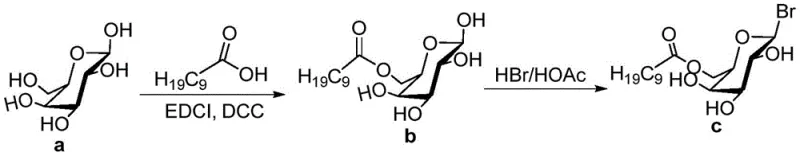
The synthesis of Houttuynoid G serves as a representative example of the platform technology described in the patent, demonstrating the feasibility of producing these complex molecules in a laboratory setting. The process integrates standard organic transformations such as acetylation, cross-coupling, and hydrogenolysis, making it accessible to most fine chemical manufacturing facilities. The route is designed to maximize atom economy in key steps while utilizing protecting group strategies (benzyl and acetyl groups) to manage reactivity. For process chemists looking to replicate or scale this work, attention must be paid to the specific stoichiometry of the oxidants and the temperature control during the cyclization phases to ensure optimal conversion. The detailed standardized synthesis steps below outline the precise conditions, reagents, and workup procedures required to achieve the reported yields, providing a clear roadmap for technology transfer from lab bench to pilot plant.
- Perform acetylation of the starting aldehyde followed by Sonogashira coupling with 1-undecyne to establish the carbon chain.
- Execute Claisen-Schmidt condensation and subsequent iodine-mediated oxidative cyclization in DMSO to form the flavonoid core.
- Complete the synthesis via glycosylation with specific bromosugars, deprotection, and final osmium-catalyzed oxidation to form the lactone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from natural extraction to total synthesis represents a strategic opportunity to de-risk the supply of critical antiviral intermediates. The conventional reliance on botanical sources introduces volatility in pricing and availability, whereas a synthetic route offers predictability and scalability. By adopting this patented method, organizations can secure a long-term supply of Houttuynoid derivatives without being subject to the fluctuations of the agricultural market. Furthermore, the synthetic route utilizes commodity chemicals and standard catalysts, which are readily available from global chemical suppliers, ensuring that raw material sourcing does not become a bottleneck. This stability allows for better financial planning and inventory management, as production schedules can be aligned with project milestones rather than harvest seasons.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for large-scale biomass processing and the associated solvent-intensive extraction protocols, which are notoriously expensive and environmentally taxing. By utilizing efficient catalytic steps such as the palladium-coupling and osmium oxidation, the process minimizes waste generation and reduces the consumption of raw materials per unit of product. Although specific cost percentages cannot be disclosed without a full techno-economic analysis, the removal of low-yield extraction steps logically leads to substantial cost savings. Additionally, the ability to synthesize specific analogues directly avoids the costly separation of closely related natural congeners, further driving down the effective cost of the active ingredient.
- Enhanced Supply Chain Reliability: A fully synthetic supply chain is inherently more robust against external disruptions such as climate change, crop diseases, or geopolitical issues affecting agricultural regions. The raw materials for this synthesis, including substituted benzaldehydes and alkynes, are produced by the petrochemical industry, which offers a much higher degree of supply continuity. This reliability ensures that research and development timelines are not compromised by material shortages. For clinical supply chains, this means that GMP manufacturing can be initiated with confidence, knowing that the upstream supply of the key intermediate is secure and scalable to meet the demands of late-stage trials.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to scale-up in standard stainless steel reactors, avoiding the need for specialized high-pressure or cryogenic equipment in most steps. The use of common solvents like ethanol, THF, and ethyl acetate simplifies solvent recovery and recycling processes, aligning with modern green chemistry principles. Moreover, the synthetic route allows for better control over waste streams compared to the complex organic waste generated from plant extraction. This facilitates easier compliance with environmental regulations and reduces the costs associated with waste disposal, making the process sustainable for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of houttuynoid hybrid flavonoids. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for R&D and procurement teams evaluating this technology. Understanding these details is crucial for assessing the feasibility of integrating these compounds into your drug discovery pipeline.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: The synthetic route eliminates dependence on seasonal plant harvesting and complex purification from crude extracts, ensuring consistent supply and higher purity suitable for clinical research.
Q: Which key catalytic systems are utilized in this process?
A: The process relies on Palladium-Copper catalyzed Sonogashira coupling for chain extension and Osmium tetroxide for the final stereoselective oxidation, both standard in fine chemical manufacturing.
Q: Is this method scalable for industrial production?
A: Yes, the reaction conditions utilize common solvents like THF, DMSO, and ethanol, and avoid exotic reagents, making it highly adaptable for kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Houttuynoid Supplier
The synthesis of houttuynoid hybrid flavonoids represents a sophisticated challenge in organic chemistry, requiring expertise in multistep synthesis, catalysis, and purification. NINGBO INNO PHARMCHEM stands ready to support your development needs as a trusted partner in fine chemical manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early discovery phase or preparing for market launch. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, providing you with the high-quality materials necessary for reproducible biological results.
We invite you to contact our technical procurement team to discuss how we can assist in the commercialization of this promising antiviral scaffold. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Reach out today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to clinical success with a reliable supply of high-purity houttuynoid intermediates.
