Advanced Catalytic Coupling for High-Purity Triazolopyrimidine Sulfonamide Herbicide Intermediates
Advanced Catalytic Coupling for High-Purity Triazolopyrimidine Sulfonamide Herbicide Intermediates
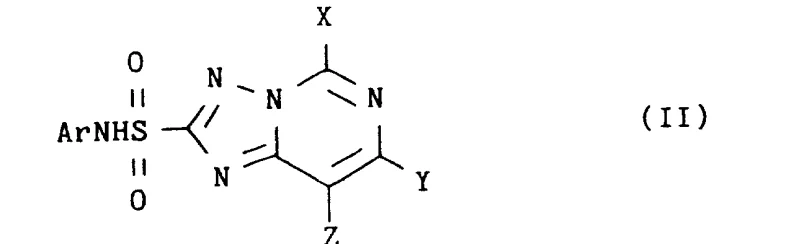
The development of efficient synthetic routes for complex heterocyclic sulfonamides is a critical bottleneck in the agrochemical industry, particularly for the production of next-generation herbicides. Patent CN1071429A introduces a transformative methodology for the preparation of N-aryl-1,2,4-triazolo-pyrimidine-2-sulfonamides, a class of compounds renowned for their potent weed-control properties in farm crops. This invention addresses the longstanding challenge of coupling deactivated arylamines with triazolopyrimidine sulfonyl halides, a reaction that has historically suffered from poor kinetics and low yields when using conventional tertiary amine bases. By leveraging a unique catalytic system involving pyridine base and dimethyl sulfoxide (DMSO), this technology enables the direct synthesis of a wide range of sulfonamide derivatives under mild conditions, bypassing the need for harsh reagents or extreme temperatures.
For R&D directors and process chemists, the significance of this patent lies in its ability to access chemical space that was previously difficult to navigate. The core innovation allows for the successful coupling of arylamines bearing strong electron-withdrawing groups, such as nitro or halogen substituents at the ortho position, which typically suppress nucleophilic activity to the point of reaction failure in standard protocols. This breakthrough not only expands the library of accessible analogs for biological screening but also provides a robust, scalable pathway for commercial manufacturing. As a reliable agrochemical intermediate supplier, understanding these mechanistic nuances is essential for optimizing production lines and ensuring the consistent delivery of high-purity intermediates required for final herbicide formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl-1,2,4-triazolo-pyrimidine-2-sulfonamides has relied on conventional coupling reactions described in prior art such as United States Patent 4,740,233. These traditional methods typically employ tertiary amine bases or a large excess of the aniline reactant to drive the nucleophilic substitution of the sulfonyl halide. While this experimental arrangement yields gratifying results when using unsubstituted anilines or those with proximate reactive behavior, it fails dramatically when the arylamine substrate contains electron-withdrawing substituents. Specifically, the presence of groups like halogens or nitro groups at the ortho position relative to the amino functionality drastically reduces the nucleophilicity of the nitrogen atom, leading to sluggish reaction rates and unacceptably low yields of the desired sulfonamide product.
To circumvent these reactivity issues, alternative strategies have been explored, such as converting the substituted aniline into a highly basic metal derivative using alkali metal alkyls or hydrides. Although this approach can successfully activate the amine for coupling, it introduces severe operational hazards and complexities, including the requirement for cryogenic conditions below 0°C and the handling of pyrophoric reagents. Furthermore, other methods involving the conversion of anilines into N-trialkylsilyl derivatives, as described in United States Patent 5,003,096, add unnecessary synthetic steps. These silylation protocols require the preparation of specialized reagents and subsequent recovery and recycling of silica byproducts, creating significant waste streams and increasing the overall cost of goods sold, which is detrimental for large-scale commercial production.
The Novel Approach
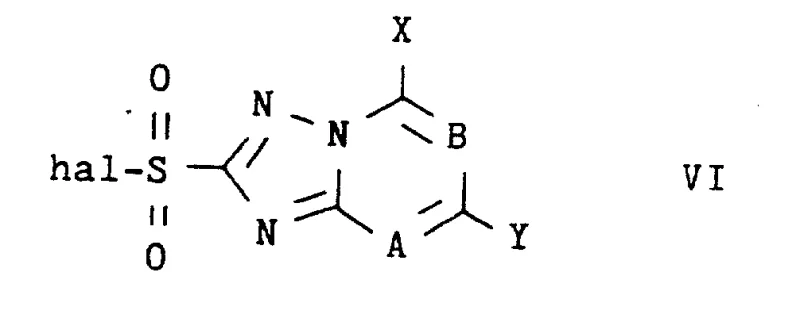
The methodology disclosed in patent CN1071429A represents a paradigm shift by utilizing a catalytic amount of dimethyl sulfoxide (DMSO) in the presence of a pyridine base to facilitate the coupling reaction. This novel approach eliminates the need for strong bases, cryogenic cooling, or extra derivatization steps, allowing the reaction to proceed smoothly at room temperature or slightly elevated temperatures ranging from 0 to 60°C. The key advantage is the dramatic enhancement of nucleophilic activity of the arylamine without altering its chemical structure permanently, enabling the efficient synthesis of sulfonamides containing electrophilic substituents that were previously inaccessible or economically unviable to produce.
By operating under these mild conditions, the new process significantly simplifies the engineering requirements for commercial scale-up. The elimination of hazardous metal reagents and the avoidance of sub-zero temperatures reduce the safety risks associated with the manufacturing process. Moreover, the use of catalytic DMSO means that the reagent load is minimal compared to stoichiometric silylating agents, leading to a cleaner reaction profile and simplified downstream purification. This efficiency translates directly into cost reduction in herbicide manufacturing, as it lowers both raw material consumption and energy expenditure for cooling and waste treatment, making it an ideal candidate for industrial adoption by forward-thinking chemical enterprises.
Mechanistic Insights into DMSO-Catalyzed Nucleophilic Substitution
The mechanistic underpinning of this invention involves the synergistic interaction between the pyridine base and the polar aprotic solvent DMSO to activate the nucleophile. In conventional tertiary amine systems, the base primarily acts to scavenge the hydrogen halide byproduct, but it does little to enhance the intrinsic nucleophilicity of a deactivated aniline. In contrast, the presence of DMSO, a solvent with a high dielectric constant and strong hydrogen bond accepting capability, likely stabilizes the transition state of the nucleophilic attack or solvates the protonated amine species more effectively, thereby shifting the equilibrium towards the formation of the sulfonamide bond. The pyridine base serves a dual role: it acts as a proton scavenger to neutralize the generated acid and potentially forms a transient complex with the sulfonyl halide to increase its electrophilicity.
This catalytic cycle is particularly effective for substrates where the aryl ring is substituted with electron-withdrawing groups such as fluorine, chlorine, bromine, trifluoromethyl, or nitro groups. The patent data indicates that the reaction tolerates a wide variety of substituents on the triazolopyrimidine core as well, including alkoxy, alkyl, and halogen groups at positions 5, 7, and 8. The robustness of this mechanism ensures that impurity profiles remain manageable, as side reactions typical of strong base conditions, such as hydrolysis of the sulfonyl halide or decomposition of sensitive functional groups, are minimized. For quality control teams, this means a more consistent impurity spectrum and higher crude purity, reducing the burden on final recrystallization steps.
How to Synthesize N-Aryl-1,2,4-triazolo-pyrimidine-2-sulfonamides Efficiently
The synthesis protocol outlined in the patent provides a straightforward, one-pot procedure that is amenable to both laboratory optimization and plant-scale execution. The process begins by charging a reactor with the substituted 1,2,4-triazolo-pyrimidine-2-alkylsulfonyl halide and the appropriate arylamine in an inert aprotic solvent such as acetonitrile. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and addition sequences required to maximize yield and minimize byproduct formation.
- Combine substituted 1,2,4-triazolo-pyrimidine-2-alkylsulfonyl halide and arylamine in an inert aprotic solvent such as acetonitrile.
- Add pyridine base and a catalytic amount of dimethyl sulfoxide (DMSO) to the reaction mixture under nitrogen protection.
- Stir the mixture at room temperature (0-60°C) until completion, then isolate the product via aqueous workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMSO-catalyzed technology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized silylating reagents or pyrophoric metal bases, manufacturers can source common, commodity-grade chemicals like pyridine and DMSO, which are readily available globally with stable pricing. This reduces the risk of supply disruptions caused by the scarcity of niche reagents and insulates the production budget from volatile market fluctuations associated with high-performance specialty chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of energy-intensive unit operations. Traditional methods requiring cryogenic cooling below 0°C demand significant capital investment in refrigeration infrastructure and ongoing energy costs to maintain low temperatures. By enabling the reaction to proceed at ambient or near-ambient temperatures, this novel route eliminates these energy burdens entirely. Furthermore, the avoidance of stoichiometric silyl reagents removes the cost of purchasing expensive silicon-based chemicals and the downstream costs associated with their disposal or recycling, leading to substantial overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and reduced cycle times. Processes that rely on sensitive metal derivatives are prone to failure if moisture ingress occurs or if temperature control deviates slightly, leading to batch losses and delayed deliveries. The tolerance of the DMSO-catalyzed method to standard processing variations ensures a more predictable production schedule. This reliability is crucial for maintaining continuous supply to downstream formulators, preventing stockouts of critical herbicide active ingredients during peak agricultural seasons.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns with green chemistry principles by reducing waste generation. The absence of heavy metal residues or silicon waste streams simplifies wastewater treatment and lowers the environmental compliance burden. The use of acetonitrile or similar solvents allows for efficient recovery and reuse through standard distillation, further minimizing the environmental footprint. This scalability ensures that the process can be seamlessly transferred from pilot plants to multi-ton commercial reactors without encountering the heat transfer or mixing limitations often associated with highly exothermic strong-base reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and scope of applicability for potential licensees or manufacturing partners looking to integrate this chemistry into their portfolios.
Q: Why is the DMSO-catalyzed method superior to traditional tertiary amine methods for electron-deficient anilines?
A: Traditional methods using tertiary amines or excess aniline often fail or provide low yields when the arylamine contains electron-withdrawing substituents, particularly at the ortho position. The DMSO-catalyzed protocol significantly enhances nucleophilic activity, allowing efficient coupling even with deactivated substrates.
Q: Does this process require cryogenic conditions like metal derivative methods?
A: No. Unlike methods requiring highly basic metal derivatives which must be conducted below 0°C, this novel approach operates effectively at room temperature (typically 0-60°C), drastically simplifying thermal management and equipment requirements.
Q: What are the preferred solvents for this catalytic coupling reaction?
A: The reaction is typically carried out in medium-polarity aprotic organic solvents that are inert to the reactants. Suitable examples include acetonitrile, ethers like dioxane or THF, and carboxylic acid esters such as ethyl acetate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolopyrimidine Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in patent CN1071429A for the production of high-value agrochemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite global partners to collaborate with us to leverage this cost-effective and scalable technology for their herbicide supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this DMSO-catalyzed process can optimize your manufacturing economics and secure your supply of critical triazolopyrimidine intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
