Advanced Synthesis of N-Aryl-1,2,4-Triazolopyrimidine-2-Sulfonamides for Commercial Herbicide Production
Introduction to Patent CN1033642C and Technological Breakthrough
The global demand for high-efficacy herbicides continues to drive innovation in the synthesis of heterocyclic sulfonamides, specifically the N-(aryl)-1,2,4-triazolopyrimidine-2-sulfonamide class. Patent CN1033642C represents a pivotal advancement in this domain, addressing long-standing synthetic challenges associated with coupling electron-deficient aromatic amines with triazolopyrimidine sulfonyl halides. This intellectual property outlines a novel catalytic system that utilizes pyridine bases in the presence of catalytic amounts of dimethyl sulfoxide (DMSO) to facilitate these difficult couplings. The significance of this technology lies in its ability to bypass the severe reactivity limitations inherent in traditional methods, particularly when dealing with arylamines substituted with strong electron-withdrawing groups or bulky ortho-substituents. By enabling direct coupling under mild conditions, this process offers a robust pathway for the reliable agrochemical intermediate supplier seeking to optimize production lines for next-generation weed control agents. The structural versatility of the resulting compounds, as illustrated in the general formula below, underscores their potential utility across a broad spectrum of agricultural applications.
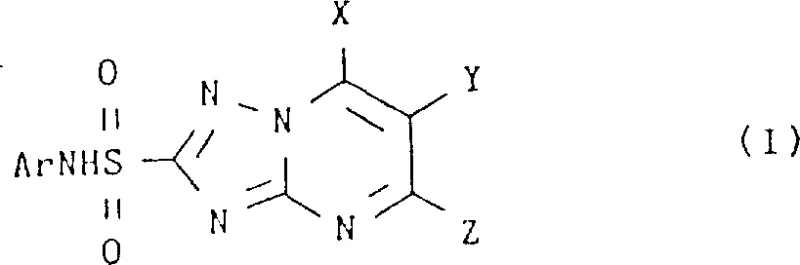
Furthermore, the patent details the preparation of isomeric forms, specifically the 1,2,4-triazolo(1,5-c)-pyrimidine derivatives, expanding the chemical space available for biological optimization. This comprehensive approach ensures that manufacturers can access a diverse library of bioactive molecules without being constrained by synthetic bottlenecks. The integration of this catalytic methodology into existing manufacturing frameworks promises to enhance the cost reduction in herbicide manufacturing by streamlining reaction sequences and minimizing the reliance on exotic reagents. As we delve deeper into the technical specifics, it becomes evident that this invention is not merely a laboratory curiosity but a commercially viable solution designed for the rigorous demands of industrial organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl-1,2,4-triazolopyrimidine-2-sulfonamides has been plagued by significant inefficiencies when attempting to couple sterically hindered or electronically deactivated amines. Conventional protocols typically rely on the use of tertiary amine bases or a large excess of the aniline reactant to drive the reaction to completion. However, these methods frequently fail when the aromatic amine possesses electron-withdrawing substituents, such as halogens or nitro groups, particularly at the ortho-position relative to the amino functionality. In such scenarios, the nucleophilicity of the amine is drastically reduced, leading to sluggish reaction kinetics and unacceptably low yields of the desired sulfonamide product. To circumvent these issues, prior art strategies have resorted to the use of highly basic reagents, such as alkali metal alkyls or hydrides, to generate reactive metal aniline derivatives.
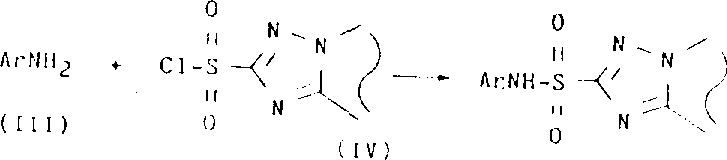
While effective in principle, these alternative approaches introduce severe operational hazards and economic burdens, including the necessity for cryogenic reaction conditions below 0°C and the handling of pyrophoric materials. Another workaround involves converting the aniline into an N-trialkylsilyl derivative to enhance nucleophilicity, but this adds an extra synthetic step and generates stoichiometric amounts of silica waste that require complex recovery and recycling procedures. These limitations render conventional methods unsuitable for the cost reduction in herbicide manufacturing on a commercial scale, as they compromise safety, increase waste disposal costs, and extend production lead times. The inability to efficiently process a wide range of substituted anilines restricts the chemical diversity available to formulators, thereby limiting the development of improved crop protection solutions.
The Novel Approach
The inventive process described in CN1033642C fundamentally transforms this landscape by introducing a mild, catalytic system that obviates the need for harsh bases or protective group chemistry. By employing a pyridine base in conjunction with a catalytic quantity of dimethyl sulfoxide (DMSO), the reaction facilitates the direct coupling of substituted 1,2,4-triazolopyrimidine-2-sulfonyl halides with even the least nucleophilic arylamines. This breakthrough allows for the successful synthesis of targets containing challenging substituents like ortho-chloro, ortho-fluoro, or trifluoromethyl groups, which were previously difficult to access with high efficiency. The reaction proceeds smoothly in inert aprotic solvents such as acetonitrile at temperatures ranging from 0°C to 60°C, with room temperature being optimal for most substrates. This operational simplicity translates directly into enhanced supply chain reliability, as it eliminates the need for specialized low-temperature reactors and hazardous reagent handling protocols.
Moreover, the catalytic nature of the DMSO additive means that it is not consumed in stoichiometric quantities, reducing material costs and simplifying the downstream purification process. The method is broadly applicable to various triazolopyrimidine scaffolds, including both 1,5-a and 1,5-c fused ring systems, providing a versatile platform for the commercial scale-up of complex agrochemical intermediates. By removing the synthetic barriers associated with electron-deficient amines, this approach empowers research and development teams to explore a wider chemical space for herbicidal activity without being constrained by manufacturability concerns. The result is a streamlined, economically attractive process that aligns perfectly with the goals of modern green chemistry and sustainable manufacturing practices.
Mechanistic Insights into DMSO-Catalyzed Nucleophilic Substitution
The efficacy of this novel synthetic route hinges on the unique synergistic interaction between the pyridine base and the dimethyl sulfoxide catalyst. While the precise mechanistic pathway may involve multiple transient species, it is hypothesized that DMSO acts to activate the sulfonyl halide electrophile or stabilize the transition state of the nucleophilic attack, thereby lowering the activation energy required for the bond formation. In traditional nucleophilic substitution reactions involving sulfonyl chlorides, the rate-determining step is often the attack of the amine on the sulfur center. When the amine is deactivated by electron-withdrawing groups, this attack becomes energetically unfavorable. The presence of DMSO, a polar aprotic solvent with high donor number properties even in catalytic amounts, likely enhances the electrophilicity of the sulfur atom or assists in the departure of the chloride leaving group. This catalytic effect is sufficient to overcome the kinetic barriers imposed by the substituents on the aromatic ring, allowing the reaction to proceed at ambient temperatures where it would otherwise be inert.
From an impurity control perspective, this mechanism offers distinct advantages over metal-mediated alternatives. By avoiding the use of strong metal bases, the process eliminates the risk of metal contamination in the final active pharmaceutical ingredient or agrochemical product, a critical quality attribute for regulatory compliance. Furthermore, the mild conditions minimize side reactions such as hydrolysis of the sulfonyl chloride or decomposition of sensitive functional groups on the heterocyclic core. The reactants involved, specifically the triazolopyrimidine sulfonyl halides of Formula (VI) shown below, are stable and readily accessible, further contributing to the robustness of the process. The ability to tolerate a wide array of substituents (X, Y, Z) on the triazolopyrimidine ring without compromising yield demonstrates the generality of this catalytic system.
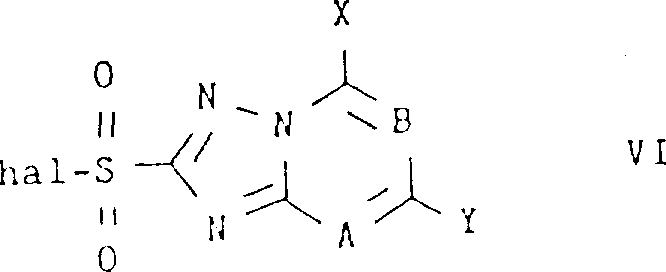
This mechanistic understanding is crucial for process chemists aiming to optimize reaction parameters for specific substrates. For instance, the ratio of pyridine to sulfonyl halide can be tuned to ensure complete consumption of the valuable electrophile, while the catalytic loading of DMSO is kept low (0.05 to 0.5 molar equivalents) to prevent potential side reactions or difficulties in product isolation. The choice of solvent, typically acetonitrile or ethyl acetate, also plays a role in maintaining the homogeneity of the reaction mixture and facilitating heat transfer. By mastering these variables, manufacturers can achieve consistent high-purity outputs, ensuring that the final herbicide intermediates meet the stringent specifications required for field application. This level of control is essential for maintaining the reputation of a reliable agrochemical intermediate supplier in a competitive global market.
How to Synthesize N-Aryl-1,2,4-Triazolopyrimidine-2-Sulfonamides Efficiently
The practical implementation of this patented technology involves a straightforward procedure that can be easily adapted to existing reactor setups. The process begins by charging a reaction vessel with the substituted triazolopyrimidine sulfonyl halide and the chosen arylamine in a dry, inert organic solvent. Under a nitrogen atmosphere to prevent moisture ingress, the pyridine base and catalytic DMSO are added, initiating the coupling reaction. The mixture is then stirred at room temperature, with reaction progress monitored by standard analytical techniques such as HPLC or TLC. Upon completion, the workup involves simple aqueous extraction and acid washing to remove excess amine and pyridine salts, followed by crystallization to isolate the pure sulfonamide product. This operational simplicity stands in stark contrast to the multi-step, cryogenic protocols of the past.
- Combine substituted 1,2,4-triazolopyrimidine-2-sulfonyl halide and aromatic amine in an inert aprotic solvent such as acetonitrile.
- Add pyridine base and a catalytic amount of dimethyl sulfoxide (DMSO) to the reaction mixture under nitrogen protection.
- Stir the mixture at room temperature (0-60°C) until completion, then isolate the product via aqueous workup and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMSO-catalyzed synthesis offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and improved throughput. By eliminating the need for hazardous strong bases like butyllithium or sodium hydride, facilities can significantly lower their safety compliance costs and insurance premiums associated with handling pyrophoric materials. Additionally, the removal of the silyl protection step saves both time and the cost of expensive silylating reagents, while also reducing the volume of silica waste that requires disposal. These cumulative efficiencies contribute to a leaner, more agile production model capable of responding rapidly to market demands for herbicide intermediates.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling requirements represents a major energy saving, as reactions can be run effectively at ambient temperatures. Furthermore, the catalytic usage of DMSO minimizes raw material consumption compared to stoichiometric additives, and the avoidance of metal derivatives removes the need for costly metal scavenging steps during purification. This holistic reduction in resource intensity leads to substantial cost savings per kilogram of produced intermediate, enhancing the overall margin profile for the final agrochemical product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as pyridine, DMSO, and acetonitrile ensures a stable and secure supply of reagents, mitigating the risk of disruptions caused by the scarcity of specialized catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental factors, resulting in more consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting delivery commitments to downstream formulators and distributors.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction facilitates easy scale-up from pilot plant to full commercial production without the engineering challenges associated with managing exotherms at low temperatures. Moreover, the reduction in hazardous waste generation aligns with increasingly strict environmental regulations, positioning the manufacturer as a responsible partner in the sustainable supply chain. The ability to produce high volumes of complex intermediates with a smaller environmental footprint is a key differentiator in the modern agrochemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: Why is DMSO used catalytically in this sulfonamide synthesis?
A: DMSO acts as a catalyst to facilitate the coupling of sulfonyl halides with arylamines that have reduced nucleophilic reactivity, particularly those with electron-withdrawing groups, allowing the reaction to proceed efficiently at room temperature without harsh bases.
Q: What are the advantages over conventional tertiary amine methods?
A: Conventional methods often fail or provide low yields when using arylamines with ortho-substituents or electron-withdrawing groups. The DMSO-catalyzed method overcomes these steric and electronic barriers, eliminating the need for cryogenic conditions or expensive silyl protection steps.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process operates at mild temperatures (0-60°C) using common solvents like acetonitrile and avoids hazardous strong bases or moisture-sensitive reagents, making it highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl-1,2,4-Triazolopyrimidine-2-Sulfonamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for high-value agrochemical intermediates. Our team of expert process chemists has extensively evaluated the technology described in CN1033642C and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are designed to handle the specific requirements of heterocyclic chemistry, featuring rigorous QC labs and stringent purity specifications that guarantee the quality of every batch we deliver.
We invite you to collaborate with us to leverage this advanced synthesis for your herbicide development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
