Advanced Lithiation Strategy for 5-Substituted-3-Furfural: Scaling High-Purity Pharmaceutical Intermediates
Introduction to Patent CN1023802C: A Breakthrough in Furan Functionalization
The pharmaceutical industry constantly seeks more efficient routes to complex heterocyclic scaffolds, and Patent CN1023802C presents a transformative approach to synthesizing 5-substituted-3-furfurals, which are critical precursors for bioactive furanones such as manoalide. This patent details a novel chemical process that fundamentally alters the synthetic landscape by enabling direct regioselective lithiation at the 5-position of 3-furfural without the cumbersome necessity of blocking the 2-position. Historically, achieving high regioselectivity in furan systems has been a significant challenge due to the similar reactivity of the alpha positions, often forcing chemists into multi-step protection strategies that degrade overall yield and increase waste. By leveraging a specific lithium-morpholinide complex followed by sec-butyllithium treatment, this invention achieves unprecedented control over the substitution pattern, offering a streamlined pathway that is both chemically elegant and commercially viable for the production of high-value anti-inflammatory intermediates.
For R&D directors and process chemists evaluating new technologies, the significance of this patent lies in its ability to condense what was previously a multi-stage sequence into a single-pot operation. The traditional reliance on blocking groups not only adds synthetic steps but also introduces additional purification burdens and potential safety hazards associated with handling sulfur-containing intermediates. In contrast, the methodology disclosed in CN1023802C utilizes readily available organolithium reagents and common solvents like tetrahydrofuran to generate the active nucleophile in situ. This shift represents a move towards atom-economical synthesis, where the focus is on maximizing the incorporation of starting materials into the final product while minimizing auxiliary substances. As we analyze the technical specifics, it becomes clear that this process offers a robust platform for generating diverse libraries of 5-substituted furans, thereby accelerating the discovery and development of next-generation therapeutic agents targeting inflammatory pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation described in this patent, the synthesis of 5-substituted-3-furfurals was heavily reliant on indirect strategies that were inherently inefficient and resource-intensive. As documented in earlier literature by researchers such as Goldsmith and Katsumura, the standard protocol involved a tedious sequence where the 2-position of the furan ring had to be first blocked, typically with a phenylthio group, to prevent unwanted side reactions during subsequent functionalization. This blocking step not only consumed valuable reagents and time but also necessitated a final deprotection step to remove the blocking group after the desired 5-substitution was achieved. Furthermore, these multi-step processes often suffered from moderate yields due to losses incurred during each isolation and purification stage, particularly when dealing with sensitive intermediates that could degrade under harsh reaction conditions or during chromatographic separation. The accumulation of byproducts from the blocking and deblocking reagents also complicated waste management, posing environmental challenges that are increasingly scrutinized in modern green chemistry initiatives.
The Novel Approach
The methodology introduced in Patent CN1023802C circumvents these historical bottlenecks by employing a sophisticated directed metalation strategy that exploits the coordinating ability of the aldehyde oxygen atom. By pre-forming a lithium-morpholinide species and subsequently treating the system with sec-butyllithium at cryogenic temperatures, the process generates a highly reactive lithiated intermediate specifically at the 5-position. This eliminates the absolute need for any 2-position blocking group, effectively collapsing a three-to-four step sequence into a single continuous operation. The result is a dramatic simplification of the workflow, where the crude reaction mixture can often proceed directly to quenching with the electrophile without intermediate isolation. This "one-pot" characteristic is a game-changer for process scalability, as it reduces the number of unit operations, minimizes solvent consumption, and significantly lowers the labor costs associated with manual handling and transfer of materials between reactors. For procurement teams, this translates to a more resilient supply chain with fewer points of failure and a reduced dependency on specialized blocking reagents that may have volatile market availability.
Mechanistic Insights into Lithium-Morpholinide Directed Lithiation
At the heart of this technological advancement is a precise understanding of organolithium coordination chemistry and how it influences regioselectivity in heteroaromatic systems. The mechanism begins with the formation of a lithium-morpholinide complex, which acts as a non-nucleophilic base capable of interacting with the carbonyl oxygen of the 3-furfural substrate. This coordination creates a transient chelate that effectively shields the adjacent positions and directs the incoming sec-butyllithium to the distal 5-position of the furan ring. The use of sec-butyllithium is critical here, as its steric bulk and high basicity ensure rapid and complete deprotonation at the targeted site before any competing nucleophilic addition to the aldehyde can occur. Maintaining the reaction temperature at approximately -78°C is essential during this phase to stabilize the lithiated intermediate and prevent thermal decomposition or rearrangement, ensuring that the kinetic product is formed exclusively. This level of mechanistic control allows for the introduction of a wide variety of electrophilic groups with high fidelity, preserving the integrity of the sensitive furan aldehyde moiety throughout the transformation.
Understanding the impurity profile is equally important for ensuring the quality of the final pharmaceutical intermediate. In conventional methods, incomplete blocking or premature deprotection often leads to mixtures of 2,5-disubstituted or 2-substituted byproducts that are difficult to separate due to their similar physical properties. However, the directed lithiation approach described here inherently suppresses substitution at the 2-position because the coordination sphere of the lithium-morpholinide complex sterically hinders access to that site. Consequently, the primary impurities are likely to be unreacted starting material or over-lithiated species, both of which are easier to manage through standard aqueous workups or crystallization techniques. The ability to produce a cleaner crude product significantly reduces the burden on downstream purification processes, such as flash chromatography or distillation, which are often the most costly and time-consuming parts of fine chemical manufacturing. This inherent purity advantage is a key selling point for contract development and manufacturing organizations (CDMOs) aiming to deliver high-specification materials to regulated markets.
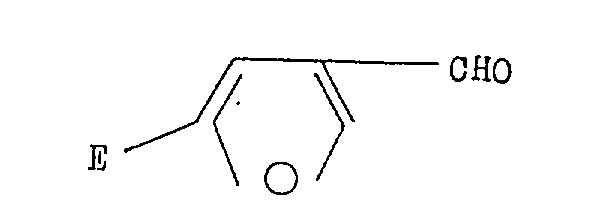
How to Synthesize 5-Substituted-3-Furfural Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly regarding moisture exclusion and temperature control, to maximize the efficiency of the organolithium reagents. The process generally begins with the preparation of the lithium-morpholinide solution in anhydrous tetrahydrofuran under an inert argon atmosphere, followed by the sequential addition of 3-furfural and sec-butyllithium at low temperatures. Once the lithiation is complete, the chosen electrophile—ranging from silyl chlorides to alkyl halides—is introduced, and the reaction mixture is allowed to gradually warm to room temperature to facilitate the coupling. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency and safety, especially given the pyrophoric nature of the reagents involved. The following guide outlines the critical operational parameters derived from the patent examples to assist technical teams in replicating this high-performance synthesis.
- Generate lithium-morpholinide in THF at -78°C under inert atmosphere by reacting n-butyllithium with morpholine.
- Add 3-furfural followed by sec-butyllithium to effect regioselective lithiation at the 5-position of the furan ring.
- Introduce the specific electrophile (e.g., silyl chloride, alkyl halide) and allow the mixture to warm to room temperature for quenching.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this lithiation technology offers profound advantages that extend well beyond the laboratory bench, directly impacting the bottom line and operational agility of chemical manufacturers. By eliminating the requirement for blocking groups and the associated deprotection steps, the process drastically reduces the raw material bill of materials (BOM) and the volume of chemical waste generated per kilogram of product. This reduction in material intensity not only lowers direct procurement costs but also diminishes the environmental footprint of the manufacturing process, aligning with increasingly stringent global sustainability regulations. For supply chain managers, the simplification of the synthetic route means fewer dependencies on niche reagents and a shorter overall cycle time from order to delivery, enhancing the reliability of supply for critical drug development programs. The robustness of the single-pot design also facilitates easier technology transfer between sites, ensuring that production can be scaled up rapidly to meet surging demand without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of the 2-position blocking group removes an entire synthetic step, along with the associated reagents, solvents, and labor costs required for that transformation. This streamlining leads to substantial cost savings by reducing the total number of unit operations and minimizing the consumption of expensive organolithium reagents through improved stoichiometric efficiency. Furthermore, the avoidance of sulfur-based blocking agents eliminates the need for specialized waste treatment protocols required for sulfide byproducts, further driving down operational expenditures related to environmental compliance and disposal. The cumulative effect of these efficiencies results in a significantly more competitive cost structure for the final 5-substituted-3-furfural intermediates, allowing manufacturers to offer better pricing to downstream pharmaceutical clients while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Simplifying the synthesis to a single-pot process reduces the number of intermediate handlings and storage requirements, thereby minimizing the risk of material degradation or contamination during production. With fewer steps comes a lower probability of batch failures or deviations, ensuring a more consistent and predictable output of high-quality intermediates. This reliability is crucial for pharmaceutical supply chains where delays in intermediate availability can stall entire drug development timelines. Additionally, the use of commodity chemicals like morpholine and standard organolithiums ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with specialized protecting group reagents, securing the continuity of supply for long-term commercial contracts.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the exothermic nature of the lithiation can be effectively managed in larger reactors through controlled dosing and cooling systems. The reduction in solvent usage and waste generation per unit of product supports greener manufacturing practices, helping companies meet their corporate social responsibility goals and adhere to strict environmental regulations. The absence of heavy metal catalysts or toxic sulfur byproducts simplifies the effluent treatment process, making it easier to obtain necessary environmental permits for large-scale production facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and sustainable partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel lithiation process for producing 5-substituted-3-furfurals. These answers are derived directly from the experimental data and claims within Patent CN1023802C, providing clarity on the scope and limitations of the technology for potential licensees or manufacturing partners. Understanding these nuances is vital for R&D teams planning to integrate this chemistry into their existing pipelines or for procurement officers evaluating the feasibility of sourcing these intermediates from new suppliers. The responses highlight the versatility of the method across different electrophiles and its suitability for producing key precursors for anti-inflammatory drugs.
Q: How does this process improve regioselectivity compared to traditional methods?
A: Unlike conventional methods that require blocking the 2-position with a phenylthio group to prevent unwanted substitution, this novel process utilizes a lithium-morpholinide complex to naturally direct lithiation to the 5-position, eliminating the need for protection-deprotection steps.
Q: What types of electrophiles are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully accommodating trialkylsilyl chlorides, trialkylstannyl chlorides, various alkyl halides, aldehydes, ketones, and isocyanates, allowing for diverse functionalization of the furan core.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the single-pot nature of the reaction without intermediate precipitation significantly simplifies processing and workup, making it highly amenable to scale-up for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted-3-Furfural Supplier
As the global demand for complex pharmaceutical intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and robust manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our deep expertise in organometallic chemistry to optimize processes like the one described in CN1023802C for industrial application. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 5-substituted-3-furfural we produce. By combining our technical prowess with a commitment to quality assurance, we provide a secure and reliable source for critical building blocks needed in the synthesis of next-generation anti-inflammatory therapeutics and other high-value fine chemicals.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this efficient lithiation technology for their specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how switching to this streamlined process can optimize your overall manufacturing budget. We encourage you to contact our technical procurement team today to request specific COA data for our available intermediates and to discuss detailed route feasibility assessments for your target molecules. Together, we can accelerate your drug development timelines and bring life-saving medicines to market faster through superior chemical innovation and supply chain excellence.
