Advanced Catalytic Halogen Exchange for Commercial Scale-Up of Complex Iodoaromatic Intermediates
Introduction to Advanced Iodoaromatic Synthesis
The development of efficient synthetic routes for functionalized aromatic compounds is a cornerstone of modern electronic material manufacturing. Patent CN1690024A introduces a transformative methodology for the preparation of monoiodinated aromatic compounds, which serve as critical precursors for triarylamine-based hole transport materials used in electrophotographic imaging and potentially in organic electronics. This technology addresses the longstanding economic and technical challenges associated with traditional iodination methods by leveraging a copper-catalyzed halogen exchange mechanism. By shifting from stoichiometric oxidants to a catalytic system utilizing inexpensive metal iodides, the process achieves high yields exceeding 90% while maintaining exceptional purity levels necessary for semiconductor applications. For industry leaders seeking a reliable electronic chemical supplier, understanding this shift from classical electrophilic substitution to transition-metal catalysis is essential for optimizing supply chains and reducing the cost of goods sold for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl iodides for charge transport applications relied heavily on direct electrophilic iodination using elemental iodine and strong oxidants such as periodic acid in acidic media. This conventional approach suffers from significant drawbacks, primarily the lack of selectivity which frequently results in the formation of di-iodinated byproducts alongside the desired mono-iodo compound. The presence of these di-iodo impurities is particularly detrimental in the subsequent Ullmann condensation reactions used to build triarylamine structures, as they lead to complex mixtures that are difficult to separate. Furthermore, the purification of mono-iodo compounds from di-iodo contaminants often necessitates energy-intensive distillation under high vacuum or costly column chromatography, drastically inflating production costs. The reliance on elemental iodine, a volatile and relatively expensive reagent compared to bromine sources, further exacerbates the economic inefficiency of these legacy processes, creating bottlenecks in the manufacturing of high-purity OLED material precursors.
The Novel Approach
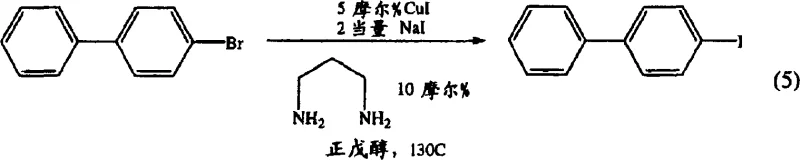
In stark contrast, the novel approach detailed in the patent utilizes a catalytic halogen exchange reaction, effectively converting readily available and cheaper aryl bromides into the corresponding aryl iodides. This method employs a copper(I) catalyst in conjunction with a specific diamine ligand, such as 1,3-propylenediamine, to facilitate the substitution of the bromine atom with iodine from sodium iodide. The reaction proceeds in high-boiling alcoholic solvents like n-amyl alcohol at temperatures around 130°C, ensuring complete conversion without the formation of poly-iodinated side products. This strategic switch not only eliminates the need for hazardous oxidants but also simplifies the downstream purification process to a straightforward recrystallization or washing step. The result is a robust, scalable pathway that delivers high-purity intermediates with yields reaching up to 92%, representing a significant advancement in cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Copper-Catalyzed Halogen Exchange
The core of this technological breakthrough lies in the sophisticated interplay between the copper catalyst and the nitrogen-based ligand, which creates a highly active catalytic cycle capable of activating the carbon-bromine bond. In this mechanism, the copper(I) species, stabilized by the bidentate diamine ligand, undergoes oxidative addition with the aryl bromide substrate to form an aryl-copper(III) intermediate. This step is crucial as it overcomes the kinetic barrier typically associated with aryl halide activation under mild conditions. Subsequently, the iodide ion from the sodium iodide source participates in a transmetallation or ligand exchange process, followed by reductive elimination to release the desired aryl iodide product and regenerate the active copper(I) catalyst. The choice of 1,3-propylenediamine is particularly effective because its bite angle and electronic properties optimize the stability of the copper center, preventing catalyst deactivation and ensuring sustained turnover numbers throughout the reaction duration.
From an impurity control perspective, this catalytic mechanism offers superior selectivity compared to electrophilic aromatic substitution. Because the reaction is driven by the thermodynamic stability of the carbon-iodine bond formation via the metal center rather than the electronic activation of the aromatic ring, there is minimal risk of over-iodination. The steric and electronic environment created by the ligand-copper complex directs the reaction exclusively towards the mono-substituted product, effectively suppressing the formation of di-iodo byproducts that plague traditional methods. This inherent selectivity means that the crude reaction mixture contains significantly fewer impurities, allowing for simpler isolation techniques such as aqueous washing and recrystallization. For R&D teams focused on purity profiles, this mechanism ensures that the final API intermediate or electronic material precursor meets stringent specifications without requiring extensive chromatographic purification, thereby preserving yield and reducing solvent waste.
How to Synthesize 4-Iodobiphenyl Efficiently
The practical implementation of this catalytic halogen exchange process is designed for operational simplicity and reproducibility in both laboratory and pilot plant settings. The procedure begins by dissolving the aryl bromide starting material, such as 4-bromobiphenyl, in a polar, high-boiling solvent like n-amyl alcohol, which serves both as the reaction medium and a heat transfer fluid. To this solution, precise stoichiometric amounts of sodium iodide, copper(I) iodide, and the diamine ligand are added under an inert atmosphere to prevent oxidation of the catalyst. The detailed standardized synthesis steps see the guide below, which outlines the specific heating profiles, workup procedures involving ammonium hydroxide washing to remove copper residues, and final recrystallization protocols to achieve pharmaceutical-grade purity. This streamlined workflow minimizes unit operations and maximizes throughput, making it an ideal candidate for commercial scale-up of complex iodoaromatic compounds.
- Dissolve the aryl bromide substrate (e.g., 4-bromobiphenyl) in a high-boiling polar solvent such as n-amyl alcohol or n-hexanol under stirring.
- Add sodium iodide (2.0 equivalents), copper(I) iodide catalyst (5 mol%), and 1,3-propylenediamine ligand (10 mol%) to the reaction mixture.
- Heat the mixture to reflux (approximately 130°C) for 16-18 hours, then cool, wash with ammonium hydroxide, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic halogen exchange technology presents a compelling value proposition centered on cost efficiency and supply security. By replacing expensive elemental iodine and hazardous oxidants with commodity chemicals like sodium iodide and aryl bromides, the raw material cost structure is significantly optimized. Aryl bromides are generally more abundant and less expensive than their iodo counterparts, and the ability to convert them in-situ allows manufacturers to leverage existing supply chains for brominated intermediates. This flexibility reduces dependency on volatile iodine markets and mitigates the risk of price fluctuations associated with halogen commodities. Furthermore, the elimination of complex purification steps such as column chromatography reduces solvent consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing operation that aligns with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The transition from stoichiometric iodination to catalytic exchange fundamentally alters the cost dynamics of producing aryl iodides. By utilizing sodium iodide, a low-cost salt, instead of molecular iodine, the process drastically reduces the expense of iodine sources. Additionally, the high selectivity of the copper-catalyzed system minimizes the formation of difficult-to-remove di-iodo impurities, thereby eliminating the need for expensive and yield-loss-prone purification techniques like preparative HPLC or repeated distillation. This streamlined downstream processing translates directly into substantial cost savings per kilogram of finished product, enhancing the overall margin profile for high-value electronic intermediates.
- Enhanced Supply Chain Reliability: Sourcing high-purity aryl iodides can often be a bottleneck due to limited global suppliers and the hazards associated with transporting elemental iodine. This new method allows for the on-demand synthesis of aryl iodides from widely available aryl bromides, which are produced on a massive scale for various industrial applications. By decentralizing the production of the iodo-intermediate and integrating it into the existing bromide supply chain, companies can reduce lead time for high-purity iodoaromatic compounds and ensure a more continuous flow of materials. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by the handling of sensitive reagents, fostering a more resilient supply network.
- Scalability and Environmental Compliance: The use of high-boiling alcohols like n-amyl alcohol as solvents offers significant advantages for scale-up, as these solvents are easier to recover and recycle compared to chlorinated or ether solvents often used in traditional methods. The reaction operates at atmospheric pressure and moderate temperatures, reducing the engineering controls required for high-pressure reactors. Moreover, the absence of strong acids and oxidants simplifies wastewater treatment and reduces the generation of hazardous waste streams. This environmentally friendly profile not only lowers compliance costs but also future-proofs the manufacturing process against tightening environmental regulations, ensuring long-term operational viability for the production of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogen exchange technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction scope, catalyst loading, and product quality. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios. The answers reflect the specific advantages of the copper-diamine system in overcoming the limitations of prior art methods.
Q: Why is catalytic halogen exchange preferred over direct iodination for aryl iodides?
A: Direct iodination using elemental iodine often leads to over-iodination (di-iodo byproducts) and requires expensive oxidants like periodic acid. The catalytic exchange method described in CN1690024A offers higher selectivity for mono-iodo products and utilizes cheaper sodium iodide.
Q: What is the role of the diamine ligand in this copper-catalyzed reaction?
A: The diamine ligand, specifically 1,3-propylenediamine, coordinates with the copper catalyst to stabilize the active species and facilitate the oxidative addition and reductive elimination steps, significantly enhancing the reaction rate and yield.
Q: Can this process be scaled for industrial production of hole transport materials?
A: Yes, the process uses readily available solvents like n-amyl alcohol and avoids hazardous reagents. The workup involves simple washing and recrystallization, making it highly suitable for large-scale manufacturing of electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Iodobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation electronic and pharmaceutical products. Our technical team has extensively analyzed the catalytic halogen exchange processes described in recent literature, including CN1690024A, to optimize our own manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of 4-iodobiphenyl or related iodoaromatic compound meets the exacting standards required for hole transport material synthesis.
We invite potential partners to engage with our technical procurement team to discuss how our optimized synthesis routes can support your specific project requirements. By leveraging our expertise in copper-catalyzed transformations, we can offer a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to our supply chain can improve your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your strategic partner in delivering high-performance chemical solutions efficiently and reliably.
