Scalable Bartoli Synthesis of 7-Methyl-5-Oxygen Substituted Indole Intermediates for Pharma
Introduction to Advanced Indole Synthesis Technologies
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes. A pivotal advancement in this domain is detailed in patent CN102718692B, which discloses a robust preparation method for 7-methyl-5-oxygen substituted indole compounds. These specific heterocyclic structures are of immense value in medicinal chemistry, serving as critical scaffolds for the development of 5-hydroxytryptamine receptor affinity agents and treatments for nervous system disorders. The traditional approaches to constructing this specific substitution pattern have historically been plagued by complexity and poor yield, limiting their utility to small-scale laboratory experiments. However, the novel methodology presented in this intellectual property leverages the power of the Bartoli indole synthesis, utilizing 2-methyl-5-oxo nitrobenzene compounds and vinyl Grignard reagents to achieve a direct, one-step cyclization. This represents a significant paradigm shift from multi-step sequences to a streamlined process that is inherently more suitable for the rigorous demands of modern supply chains.
For R&D directors and process chemists, the implications of this technology are profound. By shifting the synthetic strategy to a Grignard-based cyclization, the process eliminates several intermediate isolation steps that traditionally contribute to yield loss and increased waste generation. The reaction operates in tetrahydrofuran (THF) at controlled low temperatures, ranging from -50°C to 20°C, providing a manageable thermal profile that can be replicated in standard stainless steel reactors. This patent does not merely describe a theoretical possibility; it provides concrete embodiments demonstrating the successful synthesis of key derivatives like 5-methoxy-7-methyl-1H-indole and 5-benzyloxy-7-methyl-1H-indole. As a reliable pharmaceutical intermediate supplier, understanding and implementing such patented methodologies allows us to offer clients a distinct competitive advantage in terms of both material cost and timeline reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indole rings bearing specific oxygen and methyl substituents at the 5 and 7 positions has relied heavily on classical methodologies such as the Leimgruber-Batcho indole synthesis or variations of the SASK method. While these methods are well-documented in academic literature, they suffer from significant drawbacks when applied to this specific structural motif. The Leimgruber-Batcho route, for instance, typically involves the formation of an enamine followed by a reduction and cyclization sequence. For the 7-methyl-5-oxygen substituted targets, this pathway often encounters steric hindrance and electronic deactivation issues that drastically reduce reaction efficiency. Furthermore, the reagents required for these traditional routes can be expensive or hazardous, and the multi-step nature of the synthesis necessitates multiple purification stages. Each additional step introduces opportunities for yield erosion, accumulation of impurities, and increased solvent consumption, rendering the overall process economically unviable for large-scale industrial implementation.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data utilizes a direct Bartoli reaction strategy that fundamentally simplifies the molecular construction. By reacting a 2-methyl-5-oxo nitrobenzene derivative directly with a vinyl Grignard reagent, the indole nucleus is formed in a single operational step. This convergence of complexity is a hallmark of efficient process chemistry. The reaction mechanism bypasses the need for pre-functionalized enamines or harsh reduction conditions often associated with older methods. Instead, it relies on the nucleophilic attack of the Grignard reagent on the nitro group, followed by a spontaneous cyclization that builds the five-membered pyrrole ring onto the benzene core. This streamlined workflow not only reduces the total processing time but also minimizes the physical footprint required for manufacturing. The ability to access these valuable intermediates through such a direct route transforms them from niche, high-cost materials into accessible building blocks for drug discovery and development programs.
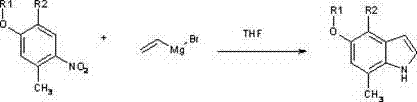
Mechanistic Insights into Vinyl Grignard-Mediated Cyclization
The core of this technological breakthrough lies in the precise mechanistic execution of the Bartoli indole synthesis. The reaction initiates with the nucleophilic addition of the vinyl Grignard reagent to the electron-deficient nitro group of the 2-methyl-5-oxo nitrobenzene substrate. This initial attack generates a transient nitronate intermediate, which subsequently undergoes a rearrangement and cyclization sequence. The presence of the ortho-methyl group is critical, as it directs the cyclization to occur at the correct position to form the 7-methyl substituted indole. The solvent choice, anhydrous tetrahydrofuran (THF), plays a dual role: it solvates the magnesium cation effectively to enhance the nucleophilicity of the vinyl anion and provides a stable medium for the highly reactive intermediates. Temperature control is another mechanistic imperative; maintaining the reaction between -50°C and 0°C during the addition phase prevents the decomposition of the Grignard reagent and suppresses competing side reactions such as polymerization or over-addition. This tight control over reaction parameters ensures that the kinetic pathway favors the desired indole formation over thermodynamic byproducts.
From an impurity control perspective, this mechanism offers distinct advantages. The primary impurities in such Grignard reactions typically arise from protonation of the reagent or incomplete cyclization. However, the protocol specifies a quenching step using aqueous ammonium chloride, which effectively neutralizes excess Grignard reagent and facilitates the separation of magnesium salts into the aqueous phase. The subsequent workup involves extraction with ethyl acetate and drying over anhydrous sodium sulfate, a standard yet effective purification train. For higher purity requirements, the patent describes a two-stage silica gel column chromatography or recrystallization process. This flexibility in purification allows manufacturers to tailor the final purity specifications based on the intended application, whether it be early-stage research requiring gram quantities or late-stage clinical supply demanding kilogram batches with stringent impurity profiles. The structural integrity of the final product, such as the 5-methoxy-7-methyl-1H-indole shown below, is confirmed through rigorous analytical techniques including NMR and Mass Spectrometry.
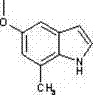
How to Synthesize 5-Methoxy-7-methyl-1H-indole Efficiently
Executing this synthesis requires adherence to strict anhydrous protocols and precise stoichiometric control. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to exclude moisture and oxygen, which are detrimental to Grignard reagents. The vinyl Grignard reagent is charged into the reactor, typically in a molar excess of 3 to 5 equivalents relative to the nitro substrate to drive the reaction to completion. The substrate, dissolved in THF, is then added dropwise while maintaining the cryogenic temperature. Following the addition, the mixture is allowed to stir for a defined period to ensure full conversion. The detailed standardized operating procedures, including specific flow rates, agitation speeds, and safety interlocks for handling pyrophoric reagents, are critical for safe scale-up. For a comprehensive breakdown of the exact experimental conditions and workup parameters, please refer to the technical guide below.
- Prepare the reaction vessel under nitrogen protection and charge with vinyl Grignard reagent in anhydrous THF, cooling the mixture to between -50°C and 0°C.
- Slowly add the solution of 2-methyl-5-oxo nitrobenzene compound dissolved in anhydrous THF to the Grignard reagent while maintaining strict temperature control.
- Quench the reaction with aqueous ammonium chloride, extract the organic phase with ethyl acetate, dry over sodium sulfate, and purify via silica gel chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel Bartoli synthesis route offers substantial strategic benefits that extend beyond simple chemistry. The primary advantage lies in the raw material supply chain. The starting materials, specifically substituted nitrotoluenes and vinyl Grignard reagents, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and price volatility often associated with exotic or custom-synthesized precursors used in older methods. Furthermore, the simplification of the synthetic route from multiple steps to a single pot reaction drastically reduces the man-hours and equipment occupancy time required per batch. This efficiency gain translates directly into lower manufacturing costs and improved throughput capacity, allowing suppliers to respond more agilely to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps significantly lowers the overall cost of goods sold (COGS). By avoiding the use of expensive catalysts or specialized reagents required in alternative pathways, the process achieves a leaner cost structure. The use of common solvents like THF and ethyl acetate further contributes to cost efficiency, as these can be readily recovered and recycled in a closed-loop system. Additionally, the high atom economy of the cyclization step means less waste is generated per unit of product, reducing disposal costs and environmental levies. This economic efficiency makes the final indole intermediate more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply continuity. Unlike processes that require ultra-low temperatures below -70°C or high-pressure hydrogenation, this method operates within a range (-50°C to 20°C) that is easily achievable with standard industrial chillers and reactors. This accessibility reduces the likelihood of equipment failure or process deviations causing production delays. Moreover, the stability of the intermediates allows for flexible scheduling; the reaction can be paused or adjusted without immediate degradation of the batch. This operational resilience ensures that delivery commitments to downstream pharmaceutical customers are met consistently, fostering long-term partnerships based on trust and reliability.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of complex exotherms that are difficult to manage in large vessels. The workup procedure relies on liquid-liquid extraction and crystallization, unit operations that are well-understood and easily scaled in multipurpose plants. From an environmental standpoint, the process generates fewer byproducts and avoids the use of heavy metal catalysts, aligning with green chemistry principles. This compliance with stringent environmental regulations simplifies the permitting process for manufacturing sites and reduces the regulatory burden on the supply chain, ensuring uninterrupted production even as global environmental standards tighten.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 7-methyl-5-oxygen substituted indoles. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent documentation. They are designed to clarify the feasibility, purity, and scalability of the method for potential partners and stakeholders evaluating this technology for their supply chains.
Q: Why is the Bartoli synthesis preferred over the Leimgruber-Batcho method for 7-methyl-5-oxygen substituted indoles?
A: The Leimgruber-Batcho method often struggles with specific substitution patterns like the 7-methyl-5-oxygen configuration, leading to difficult purification and low yields suitable only for milligram-scale lab work. The Bartoli route described in patent CN102718692B utilizes a direct one-step cyclization from readily available nitro compounds, offering a significantly simpler operational profile and better suitability for industrial scaling.
Q: What are the critical process parameters for ensuring high purity in this synthesis?
A: Maintaining anhydrous and oxygen-free conditions is paramount, typically achieved through nitrogen protection and the use of dried solvents like THF. Furthermore, strict temperature control during the addition of the nitro substrate to the Grignard reagent, specifically keeping the range between -50°C and 0°C, is essential to minimize side reactions and ensure the formation of the desired indole nucleus with high regioselectivity.
Q: Is this synthetic route viable for large-scale commercial production?
A: Yes, the process is designed for scalability. It relies on cheap and easily obtainable raw materials such as substituted nitrotoluenes and vinyl Grignard reagents. The workup procedure involves standard extraction and crystallization techniques rather than complex chromatographic separations, making it highly adaptable for multi-kilogram to ton-scale manufacturing in a GMP-compliant facility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methyl-5-Oxygen Substituted Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends heavily on the reliability and quality of the supply chain. Our expertise in implementing advanced synthetic methodologies, such as the Bartoli indole synthesis described in patent CN102718692B, positions us as a strategic partner for your drug development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to market launch. Our facilities are equipped with state-of-the-art reactors capable of handling sensitive organometallic reactions under strict temperature control, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets your exacting standards.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. By leveraging our technical capabilities, you can achieve significant cost reduction in pharmaceutical intermediate manufacturing while securing a stable supply of high-quality materials. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can add value to your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
