Revolutionizing Clotrimazole Production: A Green One-Step Synthetic Route for Commercial Scale-Up
Revolutionizing Clotrimazole Production: A Green One-Step Synthetic Route for Commercial Scale-Up
The pharmaceutical landscape is constantly evolving towards greener, more efficient synthetic pathways, and the recent disclosure in patent CN107629006B marks a significant paradigm shift in the manufacturing of Clotrimazole, a widely used antifungal agent. This groundbreaking technology abandons the cumbersome, multi-step protocols of the past in favor of a direct, one-pot nucleophilic substitution facilitated by the unique properties of hexafluoroisopropanol (HFIP). By leveraging this advanced solvent system in conjunction with mild inorganic bases, manufacturers can now achieve exceptional conversion rates under ambient conditions, effectively bypassing the severe safety hazards and energy-intensive requirements associated with legacy Grignard methodologies. For R&D directors and process chemists, this represents a tangible opportunity to streamline production workflows while simultaneously adhering to increasingly stringent environmental regulations regarding waste generation and solvent toxicity. The implications for supply chain stability are profound, as the simplification of the synthetic route directly correlates to reduced batch cycle times and lower capital expenditure on specialized cryogenic equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Clotrimazole has been plagued by inherent inefficiencies and safety concerns that drive up operational costs and complicate regulatory compliance. Traditional routes often rely on the formation of Grignard reagents, which necessitate strictly anhydrous conditions, the use of highly flammable ethers, and often cryogenic temperatures to control exothermic reactions, creating a significant safety burden for plant operators. Furthermore, alternative pathways starting from o-chlorotoluene involve multiple chlorination and Friedel-Crafts steps, resulting in a cumulative yield that rarely exceeds 65% and generates substantial quantities of acidic and organic waste streams that require costly treatment. These multi-step processes not only extend the lead time for API production but also introduce multiple points of failure where impurities can accumulate, thereby complicating downstream purification and potentially compromising the final drug substance quality. The reliance on hazardous reagents and complex thermal profiles makes scaling these conventional methods a logistical challenge, often requiring dedicated containment systems and extensive safety interlocks that erode profit margins.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a remarkably elegant solution by utilizing 2-chlorotrityl chloride and imidazole as direct coupling partners in a HFIP medium. This innovative strategy eliminates the need for pre-formed organometallic species, allowing the reaction to proceed smoothly at room temperature (25°C) over a 48-hour period without external heating or cooling. The use of sodium carbonate as a stoichiometric base provides a mild yet effective means of scavenging the hydrochloric acid byproduct, ensuring that the reaction equilibrium is driven towards completion without degrading sensitive functional groups. This one-step convergence not only simplifies the reactor setup but also dramatically improves the atom economy of the process, as evidenced by the reported yields reaching as high as 92%. By collapsing several synthetic operations into a single unit operation, this approach offers a compelling value proposition for manufacturers seeking to reduce their carbon footprint while maximizing throughput in existing infrastructure.
Mechanistic Insights into HFIP-Promoted Nucleophilic Substitution
The exceptional performance of this synthetic route can be attributed to the unique physicochemical properties of hexafluoroisopropanol (HFIP), which acts as more than just an inert solvent medium. HFIP possesses a high ionizing power and a strong ability to form hydrogen bond networks, which significantly stabilizes the developing positive charge on the trityl carbocation intermediate during the rate-determining step of the substitution. This stabilization lowers the activation energy barrier for the nucleophilic attack by imidazole, allowing the reaction to proceed efficiently even at ambient temperatures where traditional solvents would fail to generate sufficient electrophilic character. Additionally, the fluorinated alcohol environment enhances the nucleophilicity of the imidazole nitrogen by organizing the solvent shell around the transition state, effectively catalyzing the coupling without the need for expensive transition metal catalysts or aggressive Lewis acids. This mechanistic advantage ensures high selectivity for the desired N-alkylation product, minimizing the formation of regioisomers or poly-alkylated byproducts that often plague similar alkylation reactions in less polar media.
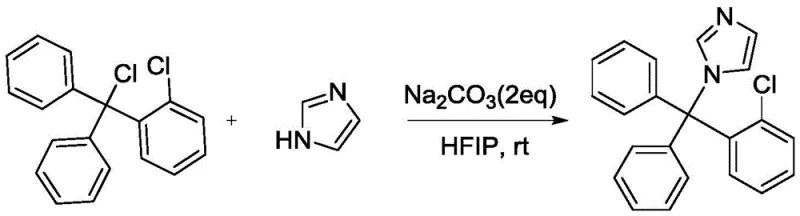
From an impurity control perspective, the choice of sodium carbonate as the base plays a pivotal role in maintaining the integrity of the reaction profile. Unlike stronger organic bases such as triethylamine, which showed lower yields in comparative studies within the patent data, sodium carbonate provides a buffered alkaline environment that prevents the degradation of the acid-sensitive trityl moiety. The heterogeneous nature of the inorganic base also facilitates easy removal post-reaction, reducing the risk of base-catalyzed side reactions during the workup phase. Furthermore, the high conversion rate (>92%) implies that very little starting material remains, simplifying the purification burden and allowing for straightforward isolation techniques such as concentration and column chromatography or crystallization. This level of control over the reaction trajectory is essential for meeting the rigorous purity specifications required for pharmaceutical intermediates, ensuring that the final API meets global pharmacopoeia standards with minimal reprocessing.
How to Synthesize Clotrimazole Efficiently
Implementing this advanced synthetic protocol requires careful attention to reagent stoichiometry and solvent quality to replicate the high yields demonstrated in the patent literature. The process begins with the precise charging of 2-chlorotrityl chloride and imidazole into the reactor, followed by the addition of the HFIP solvent which must be of high purity to prevent premature hydrolysis of the acid chloride. Detailed standardized operating procedures regarding mixing speeds, addition rates, and filtration protocols are critical to ensure consistent batch-to-batch reproducibility on a commercial scale. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the technical execution guide below.
- Charge a reactor with 2-chlorotrityl chloride and imidazole in a molar ratio of 1: 2, utilizing hexafluoroisopropanol (HFIP) as the reaction solvent.
- Add 2 equivalents of sodium carbonate (Na2CO3) to the mixture to establish the necessary alkaline environment for acid scavenging.
- Stir the reaction mixture at room temperature (25°C) for 48 hours, monitor conversion via TLC, and isolate the product through concentration and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this HFIP-mediated synthesis offers distinct strategic advantages that extend beyond mere technical feasibility. The elimination of cryogenic cooling requirements and hazardous Grignard reagents translates directly into reduced utility costs and lower insurance premiums associated with handling pyrophoric materials. Moreover, the consolidation of multiple synthetic steps into a single reaction vessel significantly decreases the overall manufacturing cycle time, allowing facilities to increase their annual production capacity without the need for additional reactor volume. This efficiency gain is particularly valuable in a market characterized by fluctuating demand for antifungal medications, as it enables manufacturers to respond more agilely to supply shortages or sudden spikes in orders. The robustness of the reaction conditions also suggests a lower risk of batch failures, thereby enhancing the reliability of supply commitments to downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational workflow and the reduction in raw material waste. By achieving near-quantitative conversion in a single step, the consumption of expensive starting materials is optimized, and the volume of solvent required for intermediate isolations is drastically curtailed. The avoidance of transition metal catalysts further reduces costs by eliminating the need for specialized scavenging resins and the associated validation testing for heavy metal residues in the final product. Additionally, the mild reaction conditions reduce energy consumption related to heating and cooling, contributing to a lower overall cost of goods sold (COGS) and improved margin potential for generic manufacturers.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable raw materials such as 2-chlorotrityl chloride and imidazole mitigates the risk of supply disruptions often associated with custom-synthesized reagents or unstable organometallics. The simplicity of the process allows for easier technology transfer between different manufacturing sites, providing supply chain heads with the flexibility to dual-source or shift production locations without extensive requalification efforts. This geographic flexibility is crucial for maintaining business continuity in the face of geopolitical tensions or logistics bottlenecks, ensuring a steady flow of high-quality intermediates to the formulation plants.
- Scalability and Environmental Compliance: From an environmental standpoint, the use of HFIP, while a specialty solvent, is balanced by the significant reduction in total waste generation due to the high yield and lack of byproduct formation. The process aligns well with green chemistry principles by avoiding toxic heavy metals and reducing the E-factor of the synthesis, making it easier to obtain environmental permits for expansion. The scalability is further supported by the non-exothermic nature of the reaction at room temperature, which removes the heat transfer limitations that often hinder the scale-up of traditional exothermic alkylations, allowing for safe production in large-scale reactors ranging from pilot to multi-ton capacities.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this route for their specific portfolio needs, we have compiled answers to common inquiries regarding the process parameters and quality attributes. These insights are derived directly from the experimental data provided in the patent documentation and reflect the practical considerations necessary for successful implementation. Understanding these nuances is key to making informed decisions about process adoption and vendor selection.
Q: Why is Hexafluoroisopropanol (HFIP) critical for this synthesis?
A: HFIP acts as a unique promoter that enhances the electrophilicity of the trityl chloride through strong hydrogen bonding networks, enabling the reaction to proceed at room temperature with significantly higher yields compared to traditional solvents like TFE or Toluene.
Q: How does this method improve upon traditional Grignard-based routes?
A: Unlike traditional methods requiring hazardous Grignard reagents, cryogenic conditions, and multi-step sequences with yields around 32-65%, this novel approach operates in a single step at ambient temperature with a verified yield of 92%, drastically reducing safety risks and operational complexity.
Q: Is the sodium carbonate base recyclable in this process?
A: While the patent specifies sodium carbonate as the optimal base for neutralizing generated HCl, the heterogeneous nature of the salt allows for straightforward filtration prior to solvent recovery, facilitating a cleaner workup profile suitable for GMP manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clotrimazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this HFIP-based synthetic route and possess the technical expertise to bring it to life on an industrial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Clotrimazole meets the highest quality benchmarks required by global regulatory agencies. Our commitment to excellence ensures that our clients receive a product that is not only cost-effective but also consistently reliable for their finished dosage formulations.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, allowing you to make data-driven decisions that enhance your competitive position in the antifungal market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
