Advanced Copper Photocatalytic Synthesis of 9-Trifluoromethyl-9,10-Dihydrophenanthrene for Commercial API Manufacturing
Introduction to Green Photocatalytic Fluorination Technology
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to access complex fluorinated scaffolds, which are critical for enhancing the metabolic stability and lipophilicity of modern drug candidates. Patent CN112441875B introduces a groundbreaking method for the synthesis of 9-trifluoromethyl-9,10-dihydrophenanthrene compounds, utilizing a sophisticated copper photocatalytic system. This technology represents a significant paradigm shift from traditional thermal processes to visible-light-driven chemistry, enabling the construction of these valuable heterocyclic cores under exceptionally mild conditions. By leveraging blue LED irradiation at temperatures ranging from 15°C to 40°C, this process eliminates the need for energy-intensive heating and hazardous reagents, aligning perfectly with the principles of green chemistry and modern sustainable manufacturing.
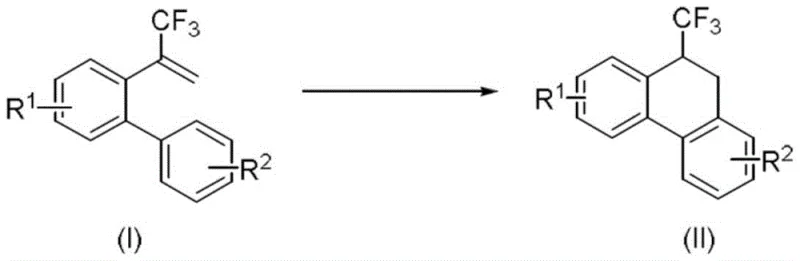
The core innovation lies in the ability to introduce the trifluoromethyl group directly into the dihydrophenanthrene framework in a single step with high atom economy. As illustrated in the general reaction scheme, the transformation proceeds via a radical cyclization mechanism initiated by the photo-excited copper catalyst. This approach not only simplifies the synthetic route but also ensures that the resulting intermediates possess the structural integrity required for downstream applications in anticancer and anti-inflammatory drug development. For R&D directors and process chemists, this patent offers a robust platform for generating diverse libraries of fluorinated phenanthrenes with superior purity profiles and reduced environmental impact compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydrophenanthrene derivatives has relied heavily on harsh thermal conditions that pose significant challenges for industrial scale-up and operator safety. A prominent example cited in the background art involves the use of 2-phenethyliodobenzene as a substrate, reacting with potassium tert-butoxide in pyridine under microwave irradiation at temperatures as high as 160°C. This conventional free-radical addition cyclization mode is fraught with limitations, primarily due to the extreme thermal energy required to overcome the activation barrier for ring closure. Furthermore, the reliance on pyridine, a solvent known for its high toxicity and unpleasant odor, creates substantial waste disposal issues and necessitates rigorous containment protocols to protect personnel and the environment.
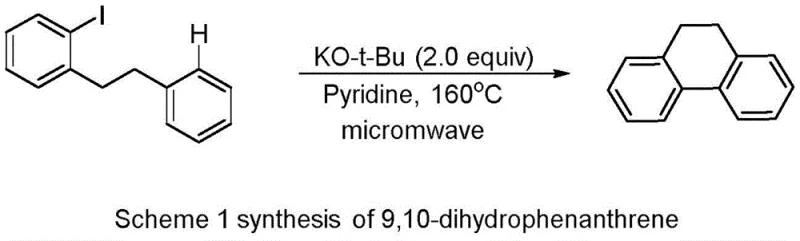
Beyond the safety concerns, the high-temperature nature of these traditional methods often leads to poor selectivity and the formation of complex impurity profiles, complicating the purification process and reducing overall yield. The energy consumption associated with maintaining such elevated temperatures, especially when using microwave reactors, is economically inefficient for large-scale production. Additionally, the substrate scope in these thermal reactions is often narrow, struggling to accommodate sensitive functional groups that might decompose under such vigorous conditions. These factors collectively hinder the cost-effective and reliable supply of high-purity dihydrophenanthrene intermediates needed for the rapid development of new pharmaceutical agents.
The Novel Approach
In stark contrast, the novel photocatalytic method described in CN112441875B utilizes visible light energy to drive the reaction, effectively bypassing the need for thermal activation. By employing a specialized copper photosensitizer in conjunction with a mild base like potassium phosphate and a greener solvent such as tetrahydrofuran, the reaction proceeds smoothly at near-ambient temperatures. This shift from thermal to photochemical activation drastically reduces the energy footprint of the process while simultaneously improving the safety profile by eliminating the risk of thermal runaway and exposure to toxic solvents. The use of blue LED light sources provides a consistent and controllable energy input that promotes the selective generation of radical intermediates necessary for cyclization.
Moreover, this innovative approach demonstrates exceptional substrate adaptability, successfully tolerating a wide array of substituents including methyl, chloro, methoxy, and phenyl groups on the aromatic rings. This versatility is crucial for medicinal chemists who need to explore structure-activity relationships (SAR) without being constrained by synthetic limitations. The reaction achieves high yields, often exceeding 70% for optimized substrates, and maintains 100% atom utilization, meaning all atoms in the starting materials are incorporated into the final product without generating stoichiometric waste. This level of efficiency and flexibility makes the novel photocatalytic route a superior choice for the commercial manufacturing of complex fluorinated intermediates.
Mechanistic Insights into Copper Photocatalytic Radical Cyclization
The success of this synthesis hinges on the unique electronic properties of the copper-based photosensitizers, specifically the Xantphos-derived complexes labeled as formulas (III) and (IV). These catalysts are designed to absorb blue light efficiently, promoting an electron from the ground state to an excited state capable of engaging in single-electron transfer (SET) processes. Upon irradiation, the excited copper complex interacts with the alpha-trifluoromethyl-styrene substrate, facilitating the homolytic cleavage or activation required to generate a carbon-centered radical species. This radical then undergoes an intramolecular addition to the adjacent aromatic ring, initiating the cyclization cascade that forms the central six-membered ring of the dihydrophenanthrene skeleton.
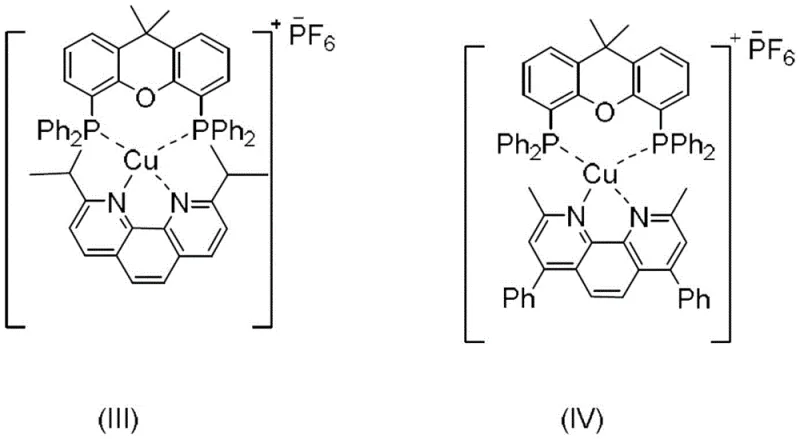
The regeneration of the catalyst and the termination of the radical chain are carefully managed by the alkaline substance present in the reaction mixture, such as potassium phosphate or carbonate. These bases play a dual role: they neutralize any acidic byproducts generated during the radical propagation steps and help maintain the catalytic cycle by stabilizing the copper species in its active oxidation state. The choice of solvent, preferably tetrahydrofuran, is also critical as it provides the appropriate polarity to solubilize both the organic substrate and the ionic base while remaining transparent to the blue LED irradiation. This precise orchestration of light, catalyst, and reagents ensures a clean reaction pathway with minimal side reactions, resulting in a high-purity product that requires less intensive downstream purification.
Furthermore, the mechanism allows for the retention of the trifluoromethyl group at the 9-position, a feature that is notoriously difficult to achieve with high regioselectivity using traditional electrophilic fluorination methods. The radical nature of the cyclization ensures that the CF3 group remains intact throughout the process, preserving the valuable physicochemical properties it imparts to the final molecule. This mechanistic understanding is vital for scaling the process, as it highlights the importance of maintaining consistent light intensity and oxygen-free conditions to prevent quenching of the excited catalyst or unwanted oxidation of the radical intermediates.
How to Synthesize 9-Trifluoromethyl-9,10-Dihydrophenanthrene Efficiently
To implement this advanced photocatalytic technology in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding light source calibration and inert atmosphere maintenance. The process begins with the precise weighing of the alpha-trifluoromethyl-styrene substrate, the copper photosensitizer, and the alkaline base, followed by their dissolution in dry tetrahydrofuran. The reaction mixture is then sealed in a vessel equipped with a blue LED array, ensuring that the light penetrates the solution uniformly to maximize photon absorption by the catalyst. Detailed standardized operating procedures for mixing ratios, irradiation times, and workup techniques are essential to reproduce the high yields reported in the patent examples consistently.
- Mix the alpha-trifluoromethyl-styrene substrate with a copper-based photosensitizer (such as Xantphos-derived Cu complexes), an alkaline substance like potassium phosphate, and a solvent such as tetrahydrofuran in a sealed reaction vessel.
- Irradiate the reaction mixture with a 15W Blue LED light source while maintaining a temperature between 15°C and 40°C under an inert gas atmosphere for a duration of 20 to 36 hours to facilitate radical cyclization.
- Upon completion, adsorb the reaction mixture onto column chromatography silica gel, remove the solvent under reduced pressure, and purify the crude product via column chromatography using petroleum ether and ethyl acetate to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of high-temperature heating and toxic solvents translates directly into reduced operational costs and simplified regulatory compliance. By avoiding the use of pyridine and microwave reactors, facilities can lower their expenditure on specialized waste treatment and safety equipment, thereby achieving significant cost reduction in pharmaceutical intermediate manufacturing. The mild reaction conditions also extend the lifespan of reactor vessels and reduce maintenance downtime, contributing to a more reliable and continuous supply chain.
- Cost Reduction in Manufacturing: The transition from thermal to photocatalytic processing removes the need for expensive energy inputs associated with maintaining 160°C temperatures, leading to substantial utility savings. Additionally, the use of relatively inexpensive copper catalysts and common bases like potassium phosphate instead of costly organometallic reagents further drives down the raw material costs. The high atom economy of the reaction minimizes waste generation, reducing the financial burden of waste disposal and environmental fees, which collectively enhances the overall profit margin for the final API.
- Enhanced Supply Chain Reliability: The robustness of this method against varying substrate substituents ensures a stable supply of diverse intermediates without the need for multiple distinct synthetic routes. Since the starting materials are readily available styrene derivatives, sourcing risks are minimized, and lead times can be significantly shortened. The simplicity of the workup procedure, involving standard silica gel chromatography, allows for faster turnaround times from reaction completion to product release, enabling quicker response to market demands and R&D timelines.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but the use of flow chemistry or multi-lamp batch reactors makes this process highly adaptable to commercial volumes. The absence of noxious waste gases and the use of greener solvents align with increasingly stringent environmental regulations, future-proofing the manufacturing process against evolving sustainability mandates. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper photocatalytic technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this novel synthesis route. Understanding these details is crucial for making informed decisions about integrating this method into existing production workflows or R&D pipelines.
Q: What are the primary advantages of this photocatalytic method over traditional thermal cyclization?
A: Unlike traditional methods requiring high temperatures (e.g., 160°C) and toxic solvents like pyridine, this copper photocatalytic approach operates at mild temperatures (15-40°C) using environmentally benign solvents like THF, significantly reducing energy consumption and operational hazards.
Q: What is the substrate scope for this 9-trifluoromethyl-9,10-dihydrophenanthrene synthesis?
A: The method demonstrates excellent adaptability, successfully cyclizing substrates with various substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like chlorine and phenyl rings, ensuring broad applicability for diverse drug scaffolds.
Q: Can the resulting dihydrophenanthrene compounds be further functionalized for drug development?
A: Yes, the synthesized 9-trifluoromethyl-9,10-dihydrophenanthrenes serve as versatile precursors that can be readily oxidized to fully aromatic phenanthrene derivatives using reagents like DDQ, expanding their utility in the synthesis of complex bioactive molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Trifluoromethyl-9,10-Dihydrophenanthrene Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this patented photocatalytic methodology into commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 9-trifluoromethyl-9,10-dihydrophenanthrene delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this green synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. Please contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your drug development timeline with our reliable supply of high-quality fluorinated intermediates.
