Scalable Manufacturing of 5-Nitroindole-2-Carboxylic Acid via Optimized Fischer Indole Synthesis
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, particularly those serving antiretroviral therapies like Azidothymidine (AZT). Patent CN100491350C introduces a transformative preparation method for 5-nitroindole-2-carboxylic acid, a pivotal building block in this therapeutic class. This technology addresses historical bottlenecks by utilizing a modified Fischer indole synthesis catalyzed by polyphosphoric acid (PPA) in a benzene-class solvent system. Unlike traditional approaches that rely on hazardous solvent-free conditions or prohibitively expensive reagents, this innovation leverages ethyl pyruvate and p-nitrophenylhydrazine hydrochloride to achieve yields exceeding 70% in the cyclization step. The process is characterized by exceptional thermal control and simplified downstream processing, resulting in a final product purity surpassing 98%. For global procurement teams, this represents a shift towards a more reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
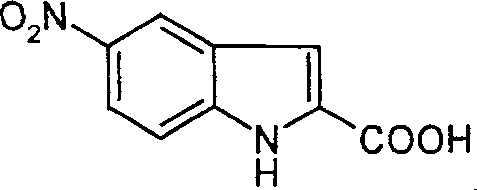
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-nitroindole derivatives has been plagued by significant safety and economic challenges that hinder industrial adoption. One prominent prior art method utilizes the Japp-Klingemann reaction, coupling diazonium salts with methyl acetoacetate; however, this pathway is economically unviable due to the exorbitant cost of methyl acetoacetate and the complexity of handling unstable diazonium intermediates. Another widely cited approach involves a solvent-free Fischer indole cyclization, which presents severe engineering risks. In the absence of a heat-transfer medium, the reaction mixture exhibits poor mixing efficiency, leading to localized hot spots. As the temperature approaches the critical 90°C threshold, a massive, uncontrollable exotherm occurs, posing a direct threat to operator safety and equipment integrity. These factors collectively render conventional methods unsuitable for modern, large-scale commercial production where safety and cost-efficiency are paramount.
The Novel Approach
The patented methodology fundamentally re-engineers the synthesis by introducing a biphasic-like environment using toluene as a reaction solvent and polyphosphoric acid as a dual-function catalyst and dehydrating agent. This strategic modification eliminates the dangerous thermal runaway associated with solvent-free conditions by ensuring uniform heat distribution throughout the reaction mass. Furthermore, the substitution of expensive precursors with ethyl pyruvate drastically reduces the raw material bill of materials. The process operates within a controlled temperature window of 85°C to 115°C, allowing for precise management of the cyclization kinetics. By decoupling the cyclization and hydrolysis steps, the method facilitates easier isolation of the intermediate ethyl ester, thereby minimizing side reactions and impurity formation. This approach not only enhances safety but also streamlines the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into PPA-Catalyzed Fischer Indole Cyclization
The core of this technological advancement lies in the efficient activation of the hydrazone intermediate through Lewis and Brønsted acid catalysis provided by polyphosphoric acid. In the initial condensation phase, p-nitrophenylhydrazine hydrochloride reacts with ethyl pyruvate to form the corresponding hydrazone, a stable precursor that sets the stage for ring closure. Upon introduction of PPA in toluene, the acid protonates the imine nitrogen of the hydrazone, increasing its electrophilicity and facilitating the crucial [3,3]-sigmatropic rearrangement characteristic of the Fischer indole mechanism. The aromatic ring then attacks the activated iminium species, followed by aromatization and the elimination of ammonia to close the indole ring. The presence of the nitro group at the 5-position requires careful electronic management, which the PPA system handles effectively by stabilizing transition states without promoting unwanted reduction or decomposition of the sensitive nitro functionality.
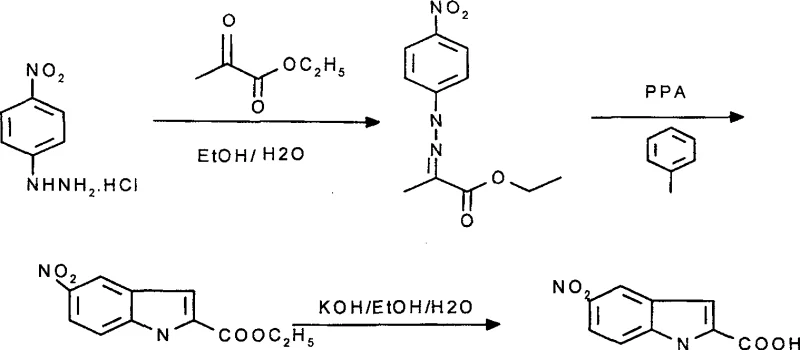
Impurity control is rigorously maintained through the specific selection of reaction parameters and the sequential nature of the synthesis. The use of toluene as a solvent allows for the azeotropic removal of water generated during cyclization, driving the equilibrium towards the desired indole ester product and preventing hydrolysis of the ester group prematurely. Following cyclization, the separate alkaline hydrolysis step using potassium hydroxide ensures complete conversion of the ester to the carboxylic acid without exposing the sensitive indole core to harsh acidic conditions for prolonged periods. The final acidification with hydrochloric acid to pH 1 precipitates the product in a highly pure crystalline form, effectively excluding organic impurities that remain soluble in the aqueous mother liquor. This meticulous control over the reaction environment ensures that the final impurity profile meets the stringent specifications required for API synthesis.
How to Synthesize 5-Nitroindole-2-Carboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing high-quality 5-nitroindole-2-carboxylic acid suitable for industrial application. The process begins with the formation of the hydrazone intermediate under mild aqueous-ethanolic conditions, followed by the critical cyclization step in toluene with PPA. The final stage involves saponification and acid precipitation to isolate the target acid. This structured approach minimizes variability and maximizes yield, addressing the common pain points of batch-to-batch inconsistency found in older methods. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Condense p-nitrophenylhydrazine hydrochloride with ethyl pyruvate in an ethanol-water mixture at 20-60°C to form the hydrazone intermediate.
- Perform Fischer indole cyclization using polyphosphoric acid (PPA) in toluene solvent at 85-115°C to generate the ethyl ester.
- Hydrolyze the ester using potassium hydroxide followed by acidification with hydrochloric acid to isolate the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond simple unit cost savings. The transition from solvent-free or diazonium-based chemistries to this PPA-catalyzed toluene process mitigates significant operational risks, ensuring uninterrupted production schedules. By utilizing commodity chemicals like ethyl pyruvate and p-nitrophenylhydrazine hydrochloride, the supply chain becomes less vulnerable to the volatility associated with specialized, high-cost reagents. Furthermore, the enhanced safety profile reduces the need for expensive engineering controls and insurance premiums, indirectly lowering the total cost of ownership. This stability is crucial for maintaining long-term contracts with API manufacturers who demand absolute reliability in their raw material streams.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of raw materials and the simplification of the reaction workflow. By eliminating the need for expensive methyl acetoacetate and avoiding the complex handling of diazonium salts, the direct material costs are substantially lowered. Additionally, the improved yield in the cyclization step, which exceeds literature values by a significant margin, means that less raw material is wasted per kilogram of final product. The ability to operate at atmospheric pressure with standard glass-lined or stainless steel reactors further reduces capital expenditure requirements compared to processes needing specialized high-pressure or cryogenic equipment.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production is not bottlenecked by the scarcity of niche reagents. Ethyl pyruvate and polyphosphoric acid are standard industrial commodities with robust global supply networks, minimizing the risk of stockouts. The robustness of the reaction conditions, specifically the tolerance for slight variations in temperature within the 85°C to 115°C range, allows for flexible manufacturing scheduling. This flexibility enables suppliers to respond more rapidly to fluctuating market demands, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream API production lines remain fully operational.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the use of toluene as a recyclable solvent and the absence of heavy metal catalysts simplify waste treatment protocols. The process generates fewer hazardous byproducts compared to diazonium routes, aligning with increasingly strict environmental regulations. The exothermic nature of the reaction is well-managed by the solvent system, allowing for safe scale-up from pilot plant quantities to multi-ton commercial production without the fear of thermal runaway. This scalability ensures that the supply can grow in tandem with the market demand for antiretroviral medications, providing a secure foundation for long-term business partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-nitroindole-2-carboxylic acid using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: Why is the PPA-catalyzed method superior to solvent-free Fischer indole synthesis?
A: The solvent-free method described in older literature suffers from poor mixing and dangerous exothermic spikes near 90°C, making temperature control difficult. The patented PPA/toluene method ensures uniform heat distribution and safer reaction kinetics.
Q: What is the expected purity of the final 5-nitroindole-2-carboxylic acid?
A: Using the optimized hydrolysis and acidification steps outlined in patent CN100491350C, the final product achieves a purity of greater than 98% as measured by HPLC, suitable for sensitive pharmaceutical applications.
Q: How does this route impact the cost of AZT intermediate production?
A: By replacing expensive methyl acetoacetate with readily available ethyl pyruvate and eliminating complex purification steps associated with diazonium salts, this route significantly lowers raw material costs and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitroindole-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications depends on the reliability and quality of every intermediate in the value chain. Our technical team has thoroughly analyzed the PPA-catalyzed route described in CN100491350C and validated its potential for large-scale implementation. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-nitroindole-2-carboxylic acid meets the exacting standards required for AZT synthesis.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in process chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can drive efficiency and security in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
