Advanced Moguisteine Manufacturing: Enhancing Purity and Scalability via Novel Cyclic Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic feasibility, particularly for active pharmaceutical ingredients (APIs) like Moguisteine, a potent peripheral antitussive agent. Patent CN101883761B introduces a transformative methodology for the synthesis of Moguisteine, specifically addressing the limitations of prior art routes that relied on unstable and impure reagents. This innovation centers on the creation of a novel cyclic intermediate, 2-[(2-methoxyphenoxy)methyl]-1,3-dioxolane, which serves as a stable precursor for the thiazolidine core. By shifting from liquid, hydrolysis-prone acetals to crystalline cyclic acetals, the process fundamentally alters the impurity profile and operational safety of the manufacturing sequence. For R&D directors and procurement specialists, this represents a critical opportunity to secure a reliable Moguisteine intermediate supplier capable of delivering material with superior consistency. The technical breakthrough lies not just in the final yield, but in the inherent stability of the intermediates, which simplifies downstream processing and ensures that the final API meets stringent regulatory standards for impurity control without excessive purification burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Moguisteine has been plagued by the use of ethyl chloroformoacetate and bromoacetaldehyde diethyl acetal, reagents that introduce significant variability and risk into the production line. Ethyl chloroformoacetate is notoriously thermally unstable, requiring careful vacuum distillation for purification, yet even then, its quality rarely exceeds 95% purity. This inherent instability leads to decomposition during storage and reaction, generating unpredictable by-products that complicate the isolation of the final API. Furthermore, the traditional precursor, bromoacetaldehyde diethyl acetal, is susceptible to hydrolysis, releasing free aldehydes and ethanol which react with guaiacol to form stubborn impurities like 2-ethoxyanisole. These side reactions result in dark, polycondensed liquid intermediates that are exceptionally difficult to purify, often necessitating high-vacuum distillation at elevated temperatures. Such harsh conditions not only degrade the product but also pose safety hazards and increase energy consumption, making the conventional route economically inefficient and environmentally burdensome for large-scale commercial operations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes 2-halomethyl-1,3-dioxolane derivatives to generate a cyclic acetal intermediate that exhibits exceptional physical stability. Unlike its linear counterparts, this cyclic structure resists hydrolysis, preventing the formation of free aldehydes and the subsequent polycondensation by-products that darken the reaction mixture. The result is an off-white crystalline solid with a purity exceeding 99%, which can be isolated through simple filtration and washing rather than energy-intensive distillation. Additionally, the final acylation step replaces the problematic ethyl chloroformoacetate with monoethyl malonate or its alkali metal salts, such as potassium monoethyl malonate. This substitution eliminates the need for handling corrosive and unstable acid chlorides, allowing the reaction to proceed under milder conditions using standard condensing agents like DCC. This strategic shift in reagent selection drastically simplifies the workflow, reduces raw material costs, and ensures a cleaner reaction profile that is far more amenable to industrial scale-up.
Mechanistic Insights into Cyclic Acetal Formation and Amide Condensation
The core of this synthetic advancement lies in the nucleophilic substitution mechanism employed in the first step, where guaiacol reacts with 2-bromomethyl-1,3-dioxolane in the presence of a base like potassium carbonate. The use of a high-boiling solvent, such as 1-methoxy-2-propanol, facilitates the reaction at temperatures between 100°C and 140°C, promoting the formation of the ether linkage while maintaining the integrity of the dioxolane ring. The cyclic nature of the acetal protecting group is crucial; it sterically hinders water attack, thereby suppressing the hydrolysis pathway that plagues linear acetals. This mechanistic stability ensures that the intermediate remains chemically inert until intentionally activated in the subsequent step with cysteamine. The transition from the dioxolane to the thiazolidine ring involves an acid-catalyzed transacetalization and cyclization, where the amino and thiol groups of cysteamine displace the oxygen atoms of the dioxolane. This transformation is highly efficient, yielding the thiazolidine core with minimal racemization or degradation, setting a pristine foundation for the final coupling reaction.
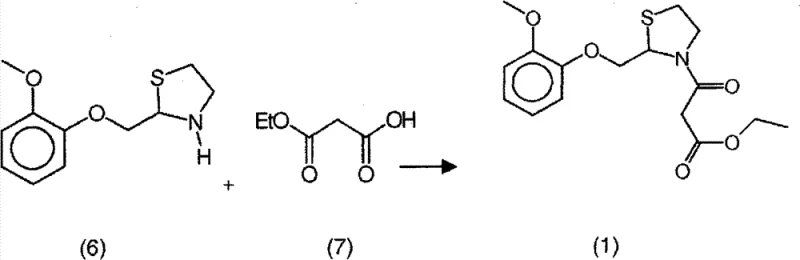
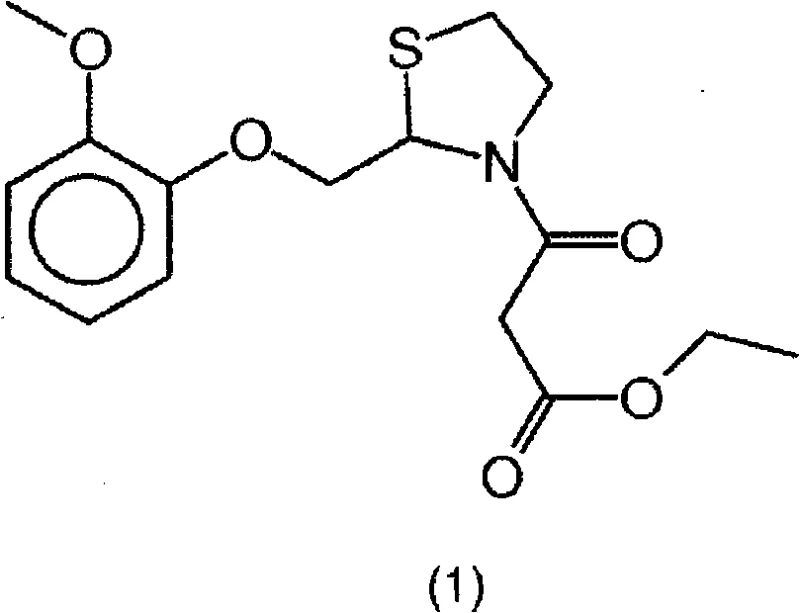
The final coupling stage employs a carbodiimide-mediated condensation mechanism, typically using N,N'-dicyclohexylcarbodiimide (DCC) to activate the carboxylic acid of monoethyl malonate. This activation forms an O-acylisourea intermediate, which is then attacked by the nitrogen of the thiazolidine ring to form the desired amide bond. The inclusion of catalysts like 1-hydroxybenzotriazole hydrate can further accelerate this process and suppress side reactions. Crucially, the by-product of this condensation, dicyclohexylurea (DCU), is insoluble in many organic solvents and can be easily removed by filtration, acting as a built-in purification step. This contrasts sharply with the soluble salts and decomposition products generated by acid chloride methods. The mechanistic elegance of using monoethyl malonate salts ensures that the beta-keto ester functionality is preserved without the risk of decarboxylation or chlorination side reactions. Consequently, the final Moguisteine product emerges with an HPLC purity often greater than 99.5%, demonstrating how precise control over reaction mechanisms directly translates to superior product quality and reduced downstream processing requirements.
How to Synthesize Moguisteine Efficiently
The synthesis of Moguisteine via this patented route involves a streamlined three-step sequence designed for maximum efficiency and purity. The process begins with the alkylation of guaiacol using a cyclic bromo-acetal in a polar aprotic or high-boiling alcoholic solvent, followed by the cyclization with cysteamine to form the thiazolidine ring. The final step couples this core with a malonate derivative using standard peptide coupling conditions. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and compliance with GMP standards.
- React guaiacol with 2-bromomethyl-1,3-dioxolane in a high-boiling solvent like 1-methoxy-2-propanol to form the stable cyclic intermediate (4).
- Convert intermediate (4) to (R,S)-2-[(2-methoxyphenoxy)methyl]-1,3-thiazolidine (6) using cysteamine hydrochloride in an acidic alcohol-water mixture.
- Couple the thiazolidine (6) with monoethyl malonate or its potassium salt using DCC as a condensing agent to yield high-purity Moguisteine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits that extend beyond mere technical superiority. The elimination of ethyl chloroformoacetate, a reagent known for its high cost and short shelf-life, immediately reduces raw material expenditure and inventory management complexity. Furthermore, the ability to isolate intermediates as stable crystalline solids rather than unstable oils significantly enhances supply chain reliability. Solid intermediates are easier to transport, store, and quality control, reducing the risk of degradation during logistics and ensuring a consistent feedstock for final API production. This stability also mitigates the risk of batch failures caused by reagent decomposition, leading to more predictable production schedules and improved on-time delivery performance for downstream pharmaceutical customers seeking a reliable Moguisteine intermediate supplier.
- Cost Reduction in Manufacturing: The replacement of expensive, unstable acid chlorides with commercially available monoethyl malonate salts results in significant cost savings. The process eliminates the need for specialized equipment required for handling corrosive gases and high-vacuum distillation of thermally sensitive liquids. Additionally, the high purity of the intermediates reduces the volume of solvents and adsorbents needed for purification, lowering waste disposal costs and improving the overall atom economy of the synthesis. These factors combine to create a more lean and cost-effective manufacturing model that enhances profit margins without compromising quality.
- Enhanced Supply Chain Reliability: By utilizing reagents that are stable and widely available, the process minimizes dependencies on niche chemical suppliers who may face production disruptions. The robustness of the cyclic intermediate allows for stockpiling without significant degradation, providing a buffer against market volatility. This resilience is critical for maintaining continuous production lines and meeting the rigorous delivery timelines demanded by global pharmaceutical clients. The simplified purification steps also reduce the lead time for batch release, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The avoidance of high-vacuum distillation and the use of milder reaction conditions make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduction in hazardous waste generation, particularly the elimination of chlorinated by-products and acidic effluents associated with acid chloride hydrolysis, aligns with increasingly strict environmental regulations. This green chemistry approach not only reduces the environmental footprint but also lowers the regulatory burden associated with waste treatment, facilitating smoother audits and approvals for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and quality outcomes of the new method.
Q: Why is the new cyclic intermediate superior to the traditional acetal?
A: The traditional linear acetal (bromoacetaldehyde diethyl acetal) is prone to hydrolysis, generating free aldehydes and polycondensation by-products that result in black liquids requiring difficult high-vacuum distillation. The novel cyclic intermediate (2-[(2-methoxyphenoxy)methyl]-1,3-dioxolane) is a stable crystalline solid with greater than 99% purity that can be isolated via simple filtration, eliminating complex purification steps.
Q: How does this process improve supply chain reliability for Moguisteine?
A: By replacing the thermally unstable and expensive ethyl chloroformoacetate with commercially available monoethyl malonate salts, the process removes a critical bottleneck. The ability to isolate solid intermediates with high purity ensures consistent quality and reduces the risk of batch failure associated with distilling unstable liquid reagents.
Q: What are the purity specifications achievable with this method?
A: The patent data indicates that Moguisteine produced via this route achieves HPLC purity greater than 99%, often exceeding 99.5% without the need for subsequent recrystallization steps, which significantly streamlines the final isolation process compared to prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moguisteine Supplier
The technological advancements described in patent CN101883761B underscore the potential for producing Moguisteine with unprecedented purity and efficiency. As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that these laboratory innovations are successfully translated into industrial reality. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the high-quality standards required for pharmaceutical intermediates. We understand the critical nature of supply continuity and quality consistency in the API sector, and our infrastructure is designed to support the complex chemistry involved in thiazolidine synthesis and amide coupling.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-value chemical solutions.
