Revolutionizing 2,2'-Dinitrostilbene Production: A Bromine-Free Route for Scalable API Manufacturing
Revolutionizing 2,2'-Dinitrostilbene Production: A Bromine-Free Route for Scalable API Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for critical active pharmaceutical ingredient (API) intermediates. A pivotal advancement in this domain is detailed in patent CN102627565A, which discloses a novel, one-step synthesis method for 2,2'-dinitrostilbene. This compound serves as a vital precursor in the manufacture of Carbamazepine, a widely prescribed anticonvulsant and mood-stabilizing drug. The traditional reliance on multi-step halogenation processes has long plagued supply chains with safety hazards and purity concerns. The innovative approach outlined in this patent utilizes a phase transfer catalysis system to directly condense o-nitrotoluene and o-nitrobenzaldehyde. This breakthrough not only simplifies the operational workflow but also fundamentally alters the impurity profile of the final product, ensuring a bromine-free specification that meets the most rigorous global pharmacopoeia standards.
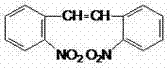
For R&D directors and process chemists, the structural integrity and purity of 2,2'-dinitrostilbene are paramount. As illustrated in the chemical structure, the molecule features a central ethene double bond linking two nitro-substituted phenyl rings. Any deviation in this structure, particularly the presence of halogenated byproducts, can compromise the efficacy and safety of the downstream API. The method described in CN102627565A achieves yields of approximately 80% with purity levels exceeding 97%, demonstrating a significant leap forward in process efficiency. By leveraging readily available starting materials and mild reaction conditions, this technology offers a compelling solution for manufacturers aiming to optimize their production lines for high-value pharmaceutical intermediates while adhering to green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,2'-dinitrostilbene has been fraught with significant technical and safety challenges. The conventional route, as depicted in earlier literature and patent backgrounds, typically initiates with o-nitrotoluene, which undergoes a series of transformations to form 2,2'-dinitrobibenzyl. This intermediate must then be subjected to a hazardous bromination step followed by a debromination elimination to generate the requisite double bond. This multi-step sequence is inherently inefficient, often resulting in overall yields hovering around 65%. More critically, the introduction of elemental bromine or brominating agents creates severe safety liabilities, including the risk of explosion during the debromination phase. Furthermore, the inevitable presence of residual organic bromine impurities poses a formidable purification challenge. These residues are notoriously difficult to eliminate completely and can persist through subsequent synthetic steps, ultimately contaminating the final Carbamazepine product and leading to potential adverse patient reactions.
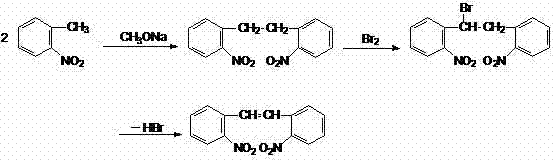
In addition to safety and purity issues, the traditional methodology suffers from substantial environmental drawbacks. The generation of bromine-containing waste streams necessitates complex and costly treatment protocols to meet environmental regulations. Alternative routes, such as the Horner-Wadsworth-Emmons reaction using o-nitrophosphonates (referenced in US4013639A), avoid bromine but introduce their own set of complications. The synthesis of o-nitrophosphonate precursors often involves phosphoric acid and other reagents that contribute to severe water pollution. Moreover, these phosphonate starting materials are not always readily available on the bulk chemical market, creating supply chain bottlenecks. The cumulative effect of these limitations—low yield, high hazard potential, difficult purification, and environmental burden—has created an urgent demand for a more streamlined and sustainable manufacturing process for this key intermediate.
The Novel Approach
The synthesis method disclosed in CN102627565A represents a paradigm shift by collapsing the entire synthetic sequence into a single, direct condensation step. By reacting o-nitrotoluene directly with o-nitrobenzaldehyde in the presence of a phase transfer catalyst and an inorganic strong base, the process bypasses the need for any halogenation or phosphonate chemistry. This one-step approach dramatically shortens the process flow, reducing the number of unit operations and the associated capital expenditure on equipment. The reaction proceeds under mild conditions, typically between 20°C and 40°C, which significantly lowers energy consumption compared to high-temperature alternatives. Crucially, the complete absence of bromine in the reaction scheme guarantees that the final product is free from bromine residues. This intrinsic purity advantage simplifies downstream processing and quality control, providing a robust foundation for the synthesis of high-quality Carbamazepine. The method effectively resolves the trade-off between yield and purity, delivering approximately 80% yield with superior product quality.
Mechanistic Insights into Phase Transfer Catalyzed Condensation
The success of this novel synthesis hinges on the efficient application of phase transfer catalysis (PTC) to drive the condensation reaction between two distinct phases. In this system, the reactants—o-nitrotoluene and o-nitrobenzaldehyde—are dissolved in an organic solvent such as toluene or benzene, while the base (e.g., sodium hydroxide or potassium hydroxide) resides in the aqueous phase. Without a catalyst, the reaction would be kinetically hindered due to the lack of contact between the organic substrates and the hydroxide ions required for deprotonation. The quaternary ammonium salt catalyst, such as tetrabutylammonium bromide, acts as a molecular shuttle. It transports hydroxide ions from the aqueous layer into the organic layer, where they can deprotonate the methyl group of o-nitrotoluene to form a reactive carbanion. This activated species then attacks the carbonyl carbon of o-nitrobenzaldehyde, initiating the condensation cascade that ultimately leads to the formation of the stilbene double bond.
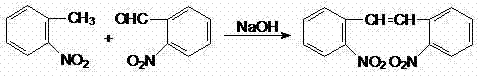
Beyond merely accelerating the reaction rate, the PTC mechanism plays a vital role in impurity control and selectivity. The mild basic conditions facilitated by the phase transfer agent prevent the degradation of sensitive functional groups that might occur under harsher thermal or acidic conditions. The careful control of the base concentration (maintained between 40% and 60%) and the dropwise addition protocol ensure that the reaction exotherm is managed effectively, preventing runaway reactions that could lead to byproduct formation. Furthermore, the choice of solvent and catalyst loading allows for fine-tuning of the reaction kinetics. For instance, optimizing the molar ratio of the phase transfer catalyst to the aldehyde between 0.075:1.0 and 0.15:1.0 ensures maximum turnover without excessive catalyst residue in the final product. This mechanistic precision results in a clean reaction profile, minimizing the formation of polymeric byproducts or over-condensed species, thereby contributing to the high purity (>97%) observed in the isolated solid product.
How to Synthesize 2,2'-Dinitrostilbene Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of a homogeneous mixture of the organic substrates and the catalyst in a suitable solvent. Temperature control is critical during the addition of the base to maintain the reaction within the optimal window of 30°C to 35°C. Following the reaction period, a straightforward workup involving aqueous washing and solvent removal yields the crude product, which can be further purified by precipitation with methanol. The detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are outlined below.
- Mix o-nitrotoluene, o-nitrobenzaldehyde, and a phase transfer catalyst (e.g., tetrabutylammonium bromide) in an organic solvent like toluene.
- Maintain temperature at 30-35°C and slowly add aqueous inorganic strong base (e.g., 50% NaOH) while stirring for 5-12 hours.
- Wash the reaction mixture to neutral pH, remove solvent under reduced pressure, and precipitate the product using methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this bromine-free synthesis route offers transformative economic and logistical benefits. The primary driver of value is the drastic simplification of the manufacturing process. By eliminating the bromination and debromination steps, manufacturers can significantly reduce the consumption of hazardous reagents and the associated costs of handling, storage, and disposal. The removal of these steps also shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. Furthermore, the use of commodity chemicals like o-nitrotoluene and o-nitrobenzaldehyde as starting materials ensures a stable and reliable supply base, mitigating the risks associated with sourcing specialized or scarce reagents like o-nitrophosphonates.
- Cost Reduction in Manufacturing: The streamlined one-step process inherently lowers operational expenditures by reducing energy consumption, labor hours, and solvent usage. The elimination of the bromination step removes the need for expensive corrosion-resistant equipment and complex waste treatment systems required for halogenated effluents. Additionally, the higher yield of approximately 80% compared to the traditional 65% means that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS). The absence of bromine residues also reduces the burden on quality control laboratories, as extensive testing for halogen impurities is no longer necessary, further driving down overhead costs.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals rather than specialized intermediates enhances the resilience of the supply chain. The traditional reliance on bromine introduces volatility, as bromine markets can be subject to regulatory restrictions and supply disruptions. By shifting to a base-catalyzed condensation using common inorganic bases like sodium hydroxide, the process becomes less susceptible to such external shocks. This stability ensures consistent production schedules and reliable delivery timelines for downstream API manufacturers, fostering stronger long-term partnerships between intermediate suppliers and pharmaceutical companies.
- Scalability and Environmental Compliance: The environmental profile of this new method aligns perfectly with modern sustainability goals and regulatory frameworks. The reduction in hazardous waste generation simplifies compliance with environmental protection laws, reducing the risk of fines and operational shutdowns. The process is inherently safer, removing the explosion risks associated with debromination, which lowers insurance premiums and improves workplace safety metrics. From a scalability perspective, the simplicity of the reaction setup allows for easy translation from kilogram-scale batches to multi-ton annual production capacities without the need for complex engineering modifications, facilitating rapid capacity expansion to meet growing global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,2'-dinitrostilbene using this advanced synthesis method. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and product quality attributes.
Q: Why is the bromine-free synthesis route critical for Carbamazepine production?
A: Traditional methods introduce bromine residues that are difficult to remove completely. These residues can carry over into the final Carbamazepine API, causing side effects and failing stringent international quality standards. The new method eliminates this risk entirely.
Q: How does the phase transfer catalyst improve the reaction efficiency?
A: The quaternary ammonium salt facilitates the transport of hydroxide ions from the aqueous phase into the organic phase where the reactants reside. This significantly accelerates the condensation rate at mild temperatures (30-35°C), avoiding the need for harsh conditions.
Q: What are the scalability advantages of this one-step process?
A: By reducing the synthesis from multiple steps (including hazardous bromination) to a single condensation step, the process drastically reduces equipment requirements, processing time, and waste generation, making it ideal for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Dinitrostilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this bromine-free synthesis are fully realized at an industrial level. We are committed to delivering high-purity 2,2'-dinitrostilbene that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch is free from critical impurities, providing a solid foundation for your Carbamazepine manufacturing processes.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out to request specific COA data and route feasibility assessments to verify how our advanced synthesis method can enhance your production efficiency. Let us help you secure a reliable, high-quality supply of this critical intermediate while advancing your sustainability goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
