Revolutionizing Heterocyclic Acetylation: A Deep Dive into Green Catalytic Manufacturing and Commercial Scalability
Revolutionizing Heterocyclic Acetylation: A Deep Dive into Green Catalytic Manufacturing and Commercial Scalability
The landscape of fine chemical manufacturing is undergoing a profound transformation driven by the urgent need for greener, more efficient synthetic pathways that align with stringent environmental regulations and cost-efficiency mandates. A pivotal advancement in this domain is documented in patent CN101928268A, which discloses a sophisticated method for synthesizing heterocyclic acetylized compounds through the composite catalysis of ionic liquids and phosphoric acid. This technology represents a significant leap forward for the production of critical pharmaceutical intermediates, such as 2-acetylfuran and 2-acetylthiophene, by replacing harsh traditional catalysts with a mild, highly selective, and environmentally benign system. For R&D directors and procurement strategists alike, understanding the nuances of this ionic liquid-phosphoric acid synergy is essential for optimizing supply chains and reducing the total cost of ownership in complex organic synthesis. The ability to achieve high conversion rates under mild thermal conditions without the reliance on volatile organic solvents positions this methodology as a cornerstone for sustainable industrial chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acetylation of heterocyclic compounds like furan and thiophene has been plagued by significant technical challenges that compromise both yield and operational safety. Traditional methodologies often rely on strong Lewis acids such as aluminum chloride, zinc chloride, or corrosive mineral acids, which necessitate harsh reaction conditions including elevated temperatures and the use of large volumes of toxic organic solvents to manage exotherms and solubility. These aggressive conditions frequently trigger undesirable side reactions, including polymerization, ring-opening, and condensation, leading to a complex impurity profile that requires extensive and costly downstream purification efforts. Furthermore, the stoichiometric consumption of certain metal salt catalysts generates substantial quantities of hazardous waste, creating a heavy burden on waste treatment facilities and increasing the overall environmental footprint of the manufacturing process. The economic implications are severe, as the loss of valuable heterocyclic substrates to side reactions and the high cost of solvent recovery erode profit margins and destabilize supply continuity for key API intermediates.
The Novel Approach
In stark contrast to these legacy methods, the innovative approach detailed in the patent utilizes a composite catalyst system comprising the ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim]PF6) and phosphoric acid to drive the acetylation reaction with exceptional efficiency. This dual-catalyst strategy creates a unique microenvironment that activates the acetic anhydride effectively at significantly lower temperatures, typically ranging from 30 to 60 degrees Celsius, thereby preserving the structural integrity of the sensitive heterocyclic rings. By eliminating the need for extraneous organic solvents during the reaction phase, this method not only simplifies the reactor setup but also drastically reduces the volume of volatile organic compounds (VOCs) emitted, aligning perfectly with modern green chemistry principles. The result is a streamlined process that delivers superior selectivity, minimizing the formation of by-products and ensuring that the final crude product possesses a purity profile that facilitates easier isolation and crystallization. 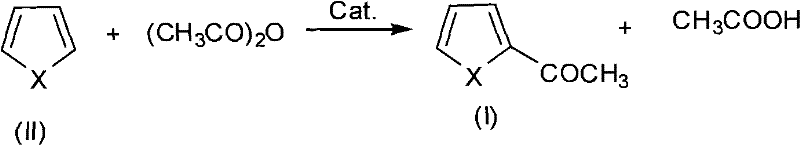
Mechanistic Insights into Ionic Liquid-Phosphoric Acid Composite Catalysis
The mechanistic superiority of this system lies in the synergistic interaction between the Brønsted acidity of the phosphoric acid and the unique solvation properties of the ionic liquid. The phosphoric acid acts as the primary proton donor to activate the carbonyl oxygen of the acetic anhydride, making it more susceptible to nucleophilic attack by the electron-rich heterocyclic ring. Simultaneously, the ionic liquid [Bmim]PF6 serves as a stabilizing medium that likely solvates the transition state and the resulting acetate anion, preventing the accumulation of free protons that could otherwise lead to acid-catalyzed degradation of the heterocycle. This cooperative effect allows the reaction to proceed with high kinetic energy at mild temperatures, effectively bypassing the activation energy barriers that typically require heat in conventional acid-catalyzed systems. The non-coordinating nature of the hexafluorophosphate anion further ensures that the catalyst remains active without being poisoned by the reaction products, sustaining high turnover numbers throughout the batch cycle.
From an impurity control perspective, this mechanism is particularly advantageous because it suppresses the electrophilic aromatic substitution pathways that lead to poly-acetylation or resinous condensation products. In traditional Friedel-Crafts type acetylations using metal halides, the generation of hydrogen halide by-products can catalyze the polymerization of the furan or thiophene ring, leading to dark, tarry residues that are difficult to separate. The phosphate-based system, however, generates acetic acid as the sole by-product, which is benign and easily removed during the aqueous workup. This clean reaction profile means that the crude product contains significantly fewer high-boiling impurities, reducing the load on the distillation columns and extending the campaign life of the purification equipment. For quality assurance teams, this translates to a more consistent specification of the final intermediate, with gas chromatography analysis routinely showing purity levels exceeding 99%, which is critical for downstream coupling reactions in API synthesis.
How to Synthesize 2-Acetylfuran Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of addition and temperature management to maximize the benefits of the composite catalyst. The process begins with the preparation of the catalyst mixture, followed by the controlled addition of acetic anhydride and the heterocyclic substrate to manage the exotherm and ensure uniform dispersion. Maintaining the reaction temperature within the optimal window of 40 to 50 degrees Celsius is crucial for balancing reaction rate and selectivity, while the molar ratio of acetic anhydride to substrate is kept tight between 1.1:1 and 1.3:1 to minimize raw material waste. The detailed standardized operating procedures, including specific stirring rates, quenching protocols, and distillation parameters required to replicate the high yields reported in the patent, are outlined in the technical guide below.
- Prepare the composite catalyst system by mixing phosphoric acid (or polyphosphoric acid) and the ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim]PF6) in a reaction vessel at room temperature with stirring.
- Add acetic anhydride to the catalyst mixture and disperse uniformly, then slowly dropwise add the heterocyclic substrate (such as furan or thiophene) while maintaining the temperature between 15 to 30 degrees Celsius.
- Gradually raise the reaction temperature to 30 to 60 degrees Celsius and maintain insulation for 5 to 15 hours to complete the acetylation, followed by standard extraction and distillation purification steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-catalyzed process offers transformative advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure and risk profile of producing heterocyclic intermediates. The elimination of expensive and hazardous Lewis acid catalysts, combined with the removal of bulk organic solvents from the reaction step, results in a drastic simplification of the raw material bill of materials and a significant reduction in waste disposal costs. This leaner manufacturing approach enhances the overall resilience of the supply chain by reducing dependency on volatile solvent markets and minimizing the logistical complexities associated with handling and storing large quantities of flammable liquids. Furthermore, the mild reaction conditions reduce the energy intensity of the process, lowering utility costs and allowing for the use of standard glass-lined or stainless steel reactors without the need for specialized corrosion-resistant alloys required by hydrochloric acid-generating processes.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the substantial reduction in raw material consumption and waste treatment expenses. By utilizing a catalytic amount of phosphoric acid and recoverable ionic liquid instead of stoichiometric amounts of metal salts, the process eliminates the cost of purchasing expensive Lewis acids and the subsequent cost of neutralizing and disposing of metal-containing sludge. Additionally, the high selectivity of the reaction minimizes the loss of the heterocyclic substrate to side products, ensuring that a greater proportion of the input material is converted into saleable product, which directly improves the gross margin per kilogram. The simplified workup procedure, which avoids complex solvent exchanges and extensive washing steps, further reduces labor hours and utility consumption, contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly de-risks the supply of critical intermediates by removing bottlenecks associated with harsh reaction conditions and difficult purifications. Because the process operates at near-ambient pressures and moderate temperatures, it is less prone to thermal runaways or equipment failures that can cause unplanned downtime and shipment delays. The use of readily available and stable reagents like acetic anhydride and phosphoric acid ensures that raw material sourcing is secure and not subject to the supply constraints often faced with specialized organometallic catalysts. This stability allows for more accurate production planning and inventory management, enabling suppliers to meet just-in-time delivery requirements with greater confidence and consistency.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more straightforward due to the absence of highly corrosive by-products and the reduced heat of reaction. The environmental profile of the process is exemplary, as it avoids the generation of chlorinated waste streams and minimizes VOC emissions, facilitating easier compliance with increasingly strict environmental regulations such as REACH and local EPA standards. This 'green' credential not only mitigates regulatory risk but also enhances the brand value of the final pharmaceutical products by supporting the sustainability goals of end-user clients. The ability to recycle the ionic liquid catalyst further amplifies these benefits, creating a circular economy model within the manufacturing plant that reduces long-term operational costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid composite catalysis technology, providing clarity on its operational feasibility and strategic value. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a realistic view of what manufacturers can expect when transitioning to this advanced synthetic route. Understanding these details is vital for technical teams evaluating the fit of this technology within their existing infrastructure and for commercial teams negotiating supply agreements based on these improved manufacturing capabilities.
Q: How does the ionic liquid composite catalyst improve yield compared to traditional Lewis acids?
A: The composite system of [Bmim]PF6 and phosphoric acid significantly enhances catalytic activity at lower temperatures, effectively suppressing side reactions like condensation and polymerization that are common with traditional Lewis acids such as aluminum chloride or zinc chloride, thereby achieving yields exceeding 90%.
Q: Is the use of toxic organic solvents required for this acetylation process?
A: No, one of the primary advantages of this patented method is that the reaction itself proceeds without the need for additional organic solvents as a reaction medium, which drastically reduces environmental pollution and simplifies the post-reaction workup process.
Q: What is the molar ratio of acetic anhydride to heterocyclic substrate for optimal results?
A: The patent specifies a molar ratio of acetic anhydride to the heterocyclic compound between 1.1:1 and 1.3:1, which is sufficient to drive the reaction to completion while avoiding the excessive waste and recovery costs associated with using large excesses of raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetylfuran Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies like the one described in CN101928268A requires a partner with deep technical expertise and a proven track record in process optimization. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this ionic liquid system are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of heterocyclic acetylized compound meets the exacting standards required for pharmaceutical applications, providing our clients with the confidence they need to advance their drug development pipelines. We are committed to leveraging this green chemistry innovation to deliver superior value, combining technical excellence with operational reliability.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic impact of switching to this catalytic system for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and sustainability in your manufacturing operations.
