Scaling Green Electrochemical Oxidation for High-Purity Bispyrazole Pharmaceutical Intermediates
Scaling Green Electrochemical Oxidation for High-Purity Bispyrazole Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are increasingly prioritizing green manufacturing technologies that minimize environmental impact while maximizing process efficiency. A pivotal advancement in this domain is detailed in patent CN101838816A, which discloses a novel electrochemical preparation method for 5,5'-dihydroxy-4,4'-bispyrazole compounds. These compounds are critical scaffolds in medicinal chemistry, known for their potent ability to scavenge reactive oxygen species (ROS) and free radicals, making them valuable candidates for treating conditions ranging from cerebral ischemia to inflammatory diseases. The patented technology replaces hazardous chemical oxidants with electricity, utilizing simple alcohol solvents and alkali metal halides to drive the oxidative coupling of 5-pyrazolones. This shift represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials through sustainable processes.
For R&D directors and process chemists, the implications of this electrochemical route are profound. Traditional synthetic pathways often rely on stoichiometric amounts of toxic reagents like selenium dioxide or unstable peroxides, which complicate purification and generate substantial hazardous waste. In contrast, the method described in CN101838816A operates under mild conditions, typically between -10°C and 40°C, and utilizes inexpensive, commercially available electrode materials. By leveraging constant current electrolysis, the process achieves precise control over the oxidation state, minimizing side reactions and enhancing the purity profile of the final product. This level of control is essential for producing high-purity bispyrazole compounds that meet the stringent regulatory standards required for active pharmaceutical ingredient (API) manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5,5'-dihydroxy-4,4'-bispyrazoles has been fraught with significant technical and safety challenges that hinder large-scale production. Prior art methods, such as those described by Obara et al., typically employ selenium dioxide as a catalyst alongside a three-fold excess of 30% hydrogen peroxide as the oxidant. This approach is inherently dangerous; hydrogen peroxide is unstable and can lead to explosive decomposition if not handled with extreme care, while selenium dioxide is highly toxic and poses severe environmental disposal issues. Furthermore, these reactions often require an inert atmosphere, such as nitrogen protection, to prevent unwanted side reactions, adding complexity and cost to the operational setup. Yields from these conventional oxidative coupling reactions are notoriously poor, frequently falling below 50%, and in some cases dropping as low as 18% when using triazine oxides. Such low efficiency not only drives up the cost of goods sold but also creates bottlenecks in the supply chain, making it difficult to secure consistent volumes of material for clinical or commercial needs.
The Novel Approach
The electrochemical methodology introduced in the patent offers a transformative solution to these longstanding industry pain points by fundamentally reimagining the oxidation mechanism. Instead of relying on chemical oxidants, this process uses electrons as the primary oxidizing agent, effectively turning the anode into a clean, reusable reagent. The reaction is conducted in a simple single-chamber electrolytic cell using common alcohol solvents like ethanol or methanol, which are far cheaper and safer than the specialized solvents often required for sensitive chemical oxidations. As illustrated in the reaction scheme below, the 5-pyrazolone substrate undergoes anodic oxidation to form radical intermediates that subsequently couple to form the desired bispyrazole structure. This eliminates the need for toxic selenium catalysts and unstable peroxides entirely, resulting in a much cleaner reaction profile.
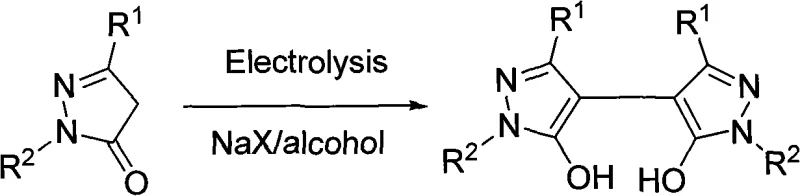
Moreover, the operational simplicity of this novel approach cannot be overstated. The process does not require rigorous exclusion of air or moisture, nor does it demand complex pressure vessels. By simply adjusting the current density to a range of 3-8 mA/cm² and controlling the temperature, manufacturers can achieve yields significantly higher than traditional methods, with optimized examples reaching up to 74%. This robustness makes the technology ideal for the commercial scale-up of complex heterocyclic intermediates, allowing producers to transition seamlessly from laboratory benchtop experiments to multi-ton industrial production without the need for extensive re-engineering of the process infrastructure.
Mechanistic Insights into Electrochemical Oxidative Coupling
Understanding the mechanistic underpinnings of this electrochemical transformation is crucial for optimizing process parameters and ensuring batch-to-batch consistency. The reaction initiates at the anode surface, where the 5-pyrazolone substrate undergoes a single-electron transfer (SET) oxidation. This generates a radical cation intermediate, which is resonance-stabilized by the adjacent nitrogen atoms and the carbonyl group within the pyrazolone ring. The presence of the alkali metal halide supporting electrolyte, such as sodium bromide, plays a dual role: it enhances the conductivity of the alcohol solvent and may participate in a mediated electron transfer process, potentially forming transient halogen species that facilitate the oxidation of the substrate. Following the initial oxidation, the radical species rapidly dimerize at the 4-position of the pyrazole ring, driven by the high electron density at this site, to form the carbon-carbon bond characteristic of the 4,4'-bispyrazole skeleton.
Impurity control is a critical aspect of this mechanism, particularly regarding the prevention of over-oxidation. In chemical oxidation methods, the strong oxidizing power of reagents like hydrogen peroxide can lead to the degradation of the pyrazole ring or the formation of N-oxides, creating difficult-to-remove impurities that compromise the safety profile of the final API. In the electrochemical system, the oxidation potential is strictly controlled by the applied current density. By maintaining the current within the optimal window of 4-7 mA/cm², the system provides just enough energy to drive the coupling reaction without pushing the potential high enough to degrade the product. Additionally, the use of an iron cathode facilitates the reduction of protons generated during the anodic oxidation, maintaining charge balance and preventing the accumulation of acidic byproducts that could catalyze hydrolysis of the sensitive pyrazolone moieties. This inherent self-regulation of the electrochemical environment contributes to the superior purity observed in the final product.
How to Synthesize 5,5'-Dihydroxy-4,4'-Bispyrazole Efficiently
Implementing this electrochemical synthesis requires careful attention to cell configuration and electrolyte selection to maximize efficiency. The patent outlines a straightforward protocol where the 5-pyrazolone starting material is dissolved in a lower alcohol, with ethanol being the preferred solvent due to its balance of solubility and cost. A supporting electrolyte, typically sodium bromide, is added to ensure sufficient ionic conductivity. The reaction is then initiated by applying a constant current across metal electrodes, with carbon or platinum serving as the anode and iron as the cathode. The process is monitored by the quantity of electricity passed, targeting approximately 1.0 Faraday per mole of substrate to ensure complete conversion while minimizing energy waste. Detailed standardized synthesis steps for specific derivatives are provided in the technical guide below.
- Dissolve the 5-pyrazolone substrate in an alcohol solvent such as ethanol or methanol within a single-chamber electrolytic cell.
- Add an alkali metal halide supporting electrolyte, preferably sodium bromide, and install metal electrodes (carbon/platinum anode and iron cathode).
- Apply a constant current density of 3-8 mA/cm² at temperatures between -10°C and 40°C until 0.2-2 Faradays per mole of electricity have passed.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates directly into tangible economic and logistical benefits. The primary driver of value is the drastic simplification of the raw material portfolio. By eliminating the need for selenium dioxide and high-concentration hydrogen peroxide, companies can reduce their dependency on hazardous material suppliers and avoid the associated regulatory compliance costs, such as special storage permits and hazardous waste disposal fees. The substitution of these expensive and dangerous reagents with electricity and common salts like sodium bromide results in cost reduction in pharmaceutical intermediate manufacturing that improves the overall margin profile of the final drug product. Furthermore, the mild reaction conditions mean that standard glass-lined or stainless steel reactors can be adapted for electrochemical use with minimal modification, protecting capital expenditure budgets.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling when analyzing the total cost of ownership. Traditional methods incur hidden costs related to the neutralization and disposal of selenium-containing waste streams, which are classified as hazardous and require specialized treatment facilities. By switching to an electrochemical method where electrons act as the reagent, these waste treatment costs are virtually eliminated. Additionally, the higher yields reported in the patent (up to 74% compared to <50% for prior art) mean that less raw material is required to produce the same amount of product, directly lowering the variable cost per kilogram. The use of inexpensive electrode materials like carbon rods and iron sheets further ensures that operating expenses remain low, as these components do not require frequent replacement or precious metal recovery processes.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the accessibility of the required inputs. Alcohol solvents and alkali metal halides are commodity chemicals produced in massive global volumes, ensuring that supply disruptions are highly unlikely even during market volatility. In contrast, specialized oxidants like 3,6-diphenyl-1,2,4-triazine-4-oxide are niche products with limited suppliers, creating a single point of failure risk. Moreover, the operational simplicity of the electrochemical process reduces the reliance on highly skilled labor for complex handling procedures, such as managing nitrogen blankets or handling explosive peroxides. This ease of operation allows for more flexible production scheduling and reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling electrochemical processes is inherently more straightforward than scaling batch chemical oxidations because the reaction rate is controlled by current rather than mixing or heat transfer limitations alone. The patent demonstrates that the reaction works effectively in a simple single-chamber cell, a configuration that is easily modularized for larger production capacities. From an environmental perspective, this method aligns perfectly with modern ESG (Environmental, Social, and Governance) goals. The absence of toxic heavy metals and the reduction of organic waste make the process cleaner and easier to permit in regions with strict environmental regulations. This "green" credential not only future-proofs the supply chain against tightening regulations but also enhances the brand reputation of the downstream pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of bispyrazole derivatives. These answers are derived directly from the experimental data and technical specifications outlined in patent CN101838816A, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these details is key to making informed decisions about process adoption and supplier qualification.
Q: What are the safety advantages of this electrochemical method over traditional oxidation?
A: Unlike conventional methods requiring toxic selenium dioxide or unstable hydrogen peroxide which pose explosion risks, this electrochemical route uses electrons as the oxidant, eliminating hazardous chemical waste and improving operational safety.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly highlights scalability due to the use of simple single-chamber cells, inexpensive electrode materials like carbon and iron, and mild reaction conditions that do not require complex nitrogen protection systems.
Q: What yields can be expected compared to prior art methods?
A: While prior art methods using selenium dioxide often yielded less than 50%, this electrochemical approach has demonstrated improved yields ranging from 51% to 74% depending on the specific substrate and optimization of current density.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,5'-Dihydroxy-4,4'-Bispyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of critical pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101838816A are fully realized in practical, industrial settings. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify the absence of heavy metal contaminants and oxidative byproducts. Our facility is equipped to handle electrochemical processes safely and efficiently, bridging the gap between innovative academic research and reliable commercial supply.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for these valuable antioxidant intermediates. By leveraging our expertise in green chemistry and process intensification, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our electrochemical capabilities can enhance the quality and reliability of your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
