Advanced Pd(0)-Catalyzed Synthesis of Optically Active Beta-Aryl-2(5H)-Furanones for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand high-purity chiral building blocks to ensure the safety and efficacy of final active ingredients. Patent CN1106393C introduces a groundbreaking methodology for the synthesis of optically active beta-aryl substituted-2(5H)-furanones, a class of five-membered unsaturated lactones known for significant biological activities including antibacterial, antifungal, and antitumor properties. This technology addresses the critical challenge of accessing these complex structures with high stereocontrol without relying on expensive or difficult-to-obtain chiral starting materials. By leveraging a palladium-catalyzed coupling strategy, the process enables the construction of the chiral center directly from readily available 2,3-allenoic acid salts and aryl iodides. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this route represents a substantial advancement in process chemistry, offering a robust pathway to generate high-value intermediates with exceptional optical purity.
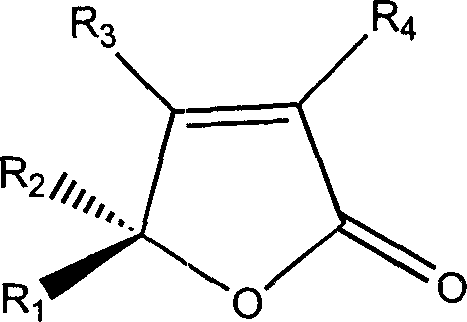
Five-membered ring unsaturated lactones are ubiquitous structural motifs in natural products, yet their asymmetric synthesis has historically been fraught with difficulties regarding regioselectivity and enantiocontrol. Conventional methods often rely on the resolution of racemic mixtures, which inherently limits the maximum theoretical yield to 50% and generates significant waste, or they require complex chiral pool starting materials that drive up costs and extend lead times. Furthermore, traditional approaches may involve harsh reaction conditions that compromise functional group tolerance, leading to impurity profiles that are difficult to manage during scale-up. The limitations of these legacy processes create bottlenecks in the supply chain, making it challenging to secure consistent quantities of high-quality material for clinical and commercial applications. Consequently, there is a pressing need for a catalytic asymmetric synthesis that can deliver these targets efficiently while minimizing environmental impact and operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active polyaryl or alkyl substituted 2(5H)-furanones has depended heavily on the use of chiral substrates that are neither commercially abundant nor cost-effective. Literature precedents, such as those cited in the patent background, describe routes starting from difficult-to-obtain chiral precursors, which necessitates multi-step sequences to install the necessary functionality before the final cyclization can occur. These lengthy synthetic pathways not only accumulate impurities at each stage but also result in poor overall atom economy, driving up the cost of goods sold (COGS) significantly. Additionally, the separation of enantiomers from racemic mixtures via chiral chromatography or diastereomeric crystallization is often labor-intensive and requires specialized equipment that is not always available in standard manufacturing facilities. The reliance on stoichiometric amounts of chiral auxiliaries in older methods further exacerbates waste generation, creating disposal challenges that conflict with modern green chemistry principles and regulatory expectations for sustainable manufacturing practices.
The Novel Approach
In stark contrast, the methodology disclosed in CN1106393C utilizes a direct Pd(0)-catalyzed coupling reaction that constructs the chiral furanone core in a single operational step from simple precursors. This novel approach employs a salt formed between 2,3-allenoic acid and a chiral organic base, such as cinchonidine, cinchonine, or alpha-methylbenzylamine, which acts as both a reactant and a source of chirality. The reaction proceeds under remarkably mild conditions, typically between 0 and 25 degrees Celsius, in common non-polar solvents like toluene, benzene, or cyclohexane. This eliminates the need for cryogenic temperatures or high-pressure equipment, thereby simplifying the engineering requirements for commercial production. The ability to use inexpensive, naturally derived chiral bases that can be recovered and recycled post-reaction provides a distinct economic advantage, drastically reducing the raw material burden compared to stoichiometric chiral reagents.
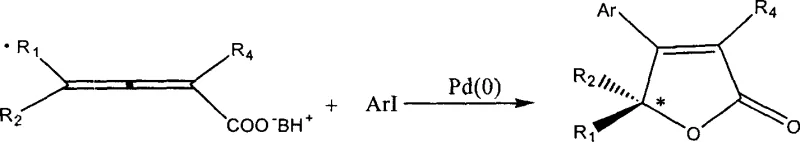
Mechanistic Insights into Pd(0)-Catalyzed Asymmetric Cyclization
The core of this transformative synthesis lies in the sophisticated interplay between the palladium catalyst and the chiral ion-pair substrate. The mechanism initiates with the oxidative addition of the aryl iodide to the zero-valent palladium species, generating an aryl-palladium(II) intermediate. This electrophilic complex then undergoes a coordinated insertion into the allene system of the chiral salt. The chirality of the organic base influences the spatial arrangement of the transition state, effectively directing the facial selectivity of the nucleophilic attack by the carboxylate oxygen onto the activated pi-system. This intramolecular cyclization step is crucial for establishing the stereocenter at the beta-position of the resulting lactone ring. Following cyclization, a reductive elimination step releases the optically active beta-aryl-2(5H)-furanone product and regenerates the active Pd(0) catalyst, allowing the cycle to continue. The precise control over the ligand environment and the nature of the chiral counter-ion ensures that the reaction proceeds with high enantioselectivity, consistently delivering ee values in the range of 91% to 97%.
From an impurity control perspective, this mechanism offers significant advantages due to the high specificity of the catalytic cycle. Because the reaction is driven by the formation of a specific organometallic intermediate, side reactions such as polymerization of the allene or homocoupling of the aryl iodide are minimized when optimal catalyst loading (5-10 mol%) and temperature controls are maintained. The use of non-polar solvents further aids in suppressing polar side reactions and facilitates the isolation of the product. The patent data indicates that the product is often obtained as a single major isomer, which simplifies downstream purification processes like column chromatography or crystallization. For quality assurance teams, this means a cleaner crude profile and a more robust process capable of meeting stringent purity specifications required for pharmaceutical intermediates. The stability of the chiral base salt under the reaction conditions also prevents racemization, ensuring that the optical integrity of the starting material is faithfully transferred to the final product.
How to Synthesize Optically Active Beta-Aryl-2(5H)-Furanones Efficiently
Implementing this synthesis requires careful attention to the preparation of the allenoic acid salt and the selection of the appropriate palladium source. The process begins by forming the salt between the 2,3-allenoic acid derivative and the chosen chiral amine, which is then dissolved in a dry, degassed non-polar solvent. To this solution, the aryl iodide is added, followed by the introduction of the catalyst system, which may include phosphine ligands to enhance activity and selectivity. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving the salt of 2,3-allenoic acid with a chiral organic base and an aryl iodide in a non-polar solvent such as toluene or benzene.
- Add the Pd(0) catalyst, such as Pd2(dba)3·CHCl3 or Pd(PPh3)4, to the mixture under inert atmosphere conditions at low temperatures ranging from 0 to 25 degrees Celsius.
- Stir the reaction for 24 to 72 hours to ensure complete conversion, followed by purification via column chromatography to isolate the optically active product with high ee values.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible improvements in cost structure and supply reliability. The shift from multi-step, resolution-based processes to a direct catalytic asymmetric synthesis fundamentally alters the economics of producing these high-value intermediates. By eliminating the need for expensive chiral pool starting materials and reducing the number of unit operations, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized low-temperature cooling infrastructure, further lowering capital expenditure requirements. Moreover, the recyclability of the chiral organic base creates a closed-loop material flow that minimizes waste disposal costs and reduces the dependency on volatile raw material markets.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the efficient use of catalysts and the recovery of chiral auxiliaries. Unlike traditional methods that consume stoichiometric amounts of expensive chiral reagents, this catalytic route uses sub-stoichiometric amounts of palladium and allows for the regeneration of the chiral base. This drastically lowers the variable cost per kilogram of the final product. Additionally, the high yields reported, ranging from 55% to 90%, mean that less raw material is wasted, improving the overall mass balance of the process. The simplicity of the workup, often requiring only column chromatography or simple crystallization, reduces labor hours and solvent consumption, contributing to a leaner and more cost-effective production workflow that enhances profit margins for downstream users.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity of complex chiral starting materials. This method mitigates that risk by utilizing readily available aryl iodides and allenoic acids, which are commodity chemicals with stable supply chains. The robustness of the reaction conditions, operating effectively at temperatures between 0 and 25 degrees Celsius, ensures that production is less susceptible to fluctuations in utility availability or equipment failures. The ability to scale this reaction from gram to kilogram scales without significant re-optimization provides confidence to supply chain planners that volume demands can be met consistently. Furthermore, the use of common solvents like toluene and hexane ensures that solvent supply risks are minimized, preventing production delays caused by specialty solvent shortages.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The absence of hazardous reagents and the use of mild temperatures reduce the thermal load on the reactor, lowering the risk of runaway reactions. From an environmental standpoint, the high atom economy and the potential for solvent recycling align with green chemistry metrics, facilitating easier regulatory approval and permitting. The reduction in waste generation, particularly the avoidance of large quantities of unwanted enantiomers from resolution processes, simplifies waste treatment protocols. This makes the process not only easier to scale to commercial tonnage but also more sustainable, helping companies meet their corporate social responsibility goals and adhere to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the typical yields and enantiomeric excess values for this synthesis?
A: According to the patent data, this method achieves yields ranging from 55% to 90% with enantiomeric excess (ee) values between 91% and 97%, demonstrating high efficiency and stereoselectivity.
Q: Can the chiral organic base be recovered after the reaction?
A: Yes, one of the key economic advantages of this process is that the chiral organic base used to form the initial salt can be recycled and reused, significantly reducing raw material costs.
Q: What catalysts are suitable for this transformation?
A: The process utilizes Pd(0) catalysts such as Pd2(dba)3·CHCl3, Pd(PPh3)4, or allyl palladium chloride, typically employed at loading levels between 5 mol% and 10 mol%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aryl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN1106393C for developing next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of optically active beta-aryl-2(5H)-furanone meets the highest international standards. Our infrastructure is designed to handle complex catalytic reactions safely and efficiently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for these critical intermediates. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us help you accelerate your development timelines and reduce your overall manufacturing costs through our proven capabilities in asymmetric synthesis and process optimization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
