Advanced Synthesis of Hydroxy Benzene Sulfonate Esters for Commercial Scale-up
Advanced Synthesis of Hydroxy Benzene Sulfonate Esters for Commercial Scale-up
The pharmaceutical industry faces increasing regulatory scrutiny regarding genotoxic impurities, particularly sulfonate esters which can alkylate DNA and pose significant safety risks. Patent CN110981763A introduces a breakthrough preparation method for hydroxy benzene sulfonate compounds that addresses these safety concerns while optimizing production efficiency. This novel approach utilizes tripropyl orthoformate to react with 2,5-dihydroxy benzene sulfonic acid, creating a robust pathway for generating high-purity intermediates essential for modern drug synthesis. By shifting away from traditional alkylating agents, this technology offers a safer, greener alternative that aligns with strict ICH Q3C guidelines for residual solvents and impurities. For R&D directors and procurement leaders, understanding this shift is critical for securing reliable supply chains that prioritize both safety and cost-effectiveness in the manufacturing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of sulfonate esters typically relies on the reaction between sulfonic acids or sulfonyl chlorides and alcohols, often requiring harsh conditions and generating significant waste. As illustrated in general reaction schemes, the direct esterification using sulfonyl chlorides can lead to the formation of corrosive byproducts like hydrogen chloride, necessitating complex neutralization and purification steps. 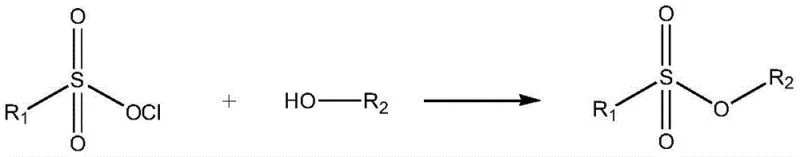 . These conventional routes frequently suffer from low yields and difficult separation processes, especially when dealing with sensitive hydroxy-substituted benzene rings that may undergo side reactions. Furthermore, the use of alkyl halides in alternative pathways introduces the risk of genotoxic impurities, requiring extensive and costly analytical monitoring to ensure patient safety. The operational complexity and environmental burden of these legacy methods make them increasingly unsustainable for large-scale commercial production in a regulated environment.
. These conventional routes frequently suffer from low yields and difficult separation processes, especially when dealing with sensitive hydroxy-substituted benzene rings that may undergo side reactions. Furthermore, the use of alkyl halides in alternative pathways introduces the risk of genotoxic impurities, requiring extensive and costly analytical monitoring to ensure patient safety. The operational complexity and environmental burden of these legacy methods make them increasingly unsustainable for large-scale commercial production in a regulated environment.
The Novel Approach
In contrast, the method disclosed in CN110981763A employs tripropyl orthoformate as a key reagent, fundamentally changing the reaction mechanism to avoid hazardous byproducts. This orthoformate-mediated esterification proceeds under milder conditions, significantly reducing the thermal stress on the substrate and minimizing the formation of degradation impurities. The process eliminates the need for corrosive sulfonyl chlorides, thereby simplifying the post-reaction workup and reducing the consumption of neutralizing agents. By streamlining the synthetic route, this approach not only enhances the overall yield but also improves the purity profile of the final hydroxy benzene sulfonate ester. For supply chain managers, this translates to a more predictable production timeline and reduced dependency on specialized waste treatment infrastructure, offering a clear competitive advantage in terms of operational efficiency and regulatory compliance.
Mechanistic Insights into Orthoformate-Mediated Esterification
The core innovation lies in the nucleophilic attack of the sulfonic acid group on the orthoformate carbon, facilitated by the specific electronic environment of the 2,5-dihydroxy benzene scaffold. 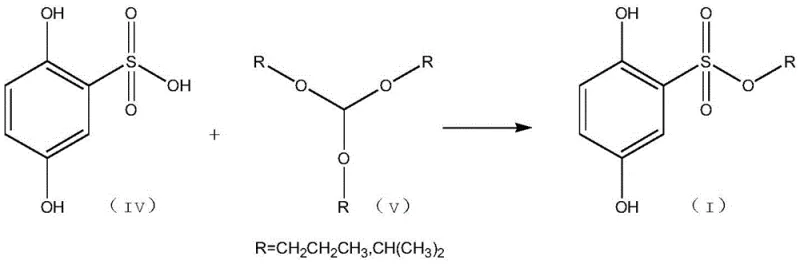 . This mechanism avoids the high-energy intermediates associated with sulfonyl chloride activation, leading to a smoother energy profile and fewer side reactions. The presence of the hydroxyl groups on the benzene ring is carefully managed through solvent selection, such as dichloromethane or ethyl acetate, which stabilizes the transition state without participating in unwanted etherification. Detailed analysis of the reaction kinetics suggests that the orthoformate acts as both a reactant and a dehydrating agent, driving the equilibrium towards the ester product without the need for external water scavengers. This intrinsic driving force is crucial for achieving high conversion rates, ensuring that the residual starting materials are minimized to levels that simplify downstream purification.
. This mechanism avoids the high-energy intermediates associated with sulfonyl chloride activation, leading to a smoother energy profile and fewer side reactions. The presence of the hydroxyl groups on the benzene ring is carefully managed through solvent selection, such as dichloromethane or ethyl acetate, which stabilizes the transition state without participating in unwanted etherification. Detailed analysis of the reaction kinetics suggests that the orthoformate acts as both a reactant and a dehydrating agent, driving the equilibrium towards the ester product without the need for external water scavengers. This intrinsic driving force is crucial for achieving high conversion rates, ensuring that the residual starting materials are minimized to levels that simplify downstream purification.
Impurity control is a paramount concern for R&D directors, especially given the genotoxic potential of sulfonate esters. The new method inherently limits the formation of alkyl sulfonates derived from free alcohols, as the orthoformate pathway does not generate free alcohol byproducts in the same manner as direct acid-alcohol condensation. Instead, the byproducts are primarily alcohols derived from the orthoformate itself, which are easier to remove due to their volatility and distinct polarity. The patent data indicates that HPLC analysis of the crude product shows a purity of 96.25%, demonstrating the effectiveness of this mechanistic approach in suppressing side reactions. By controlling the molar ratio of reactants, specifically maintaining a 1:1 to 1:5 ratio of acid to orthoformate, the process ensures complete consumption of the sulfonic acid while preventing excess reagent contamination. This level of control is essential for meeting the ppm-level limits required for genotoxic impurities in final drug substances.
How to Synthesize 2,5-Dihydroxy Benzene Sulfonate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these results in a laboratory or pilot plant setting. The process begins with the dissolution of 2,5-dihydroxy benzene sulfonic acid in a reaction solvent, followed by the addition of tripropyl orthoformate under stirring. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React 2,5-dihydroxy benzene sulfonic acid with tripropyl orthoformate in a suitable solvent like dichloromethane or ethyl acetate.
- Maintain reaction temperature between 60°C and reflux conditions for 6 to 36 hours depending on the specific orthoformate used.
- Purify the resulting ester through solvent evaporation and silica gel column chromatography to achieve high purity standards.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers, the shift to this orthoformate-based methodology offers substantial cost reduction in pharmaceutical intermediate manufacturing by simplifying the raw material portfolio. The elimination of sulfonyl chlorides removes the need for expensive corrosion-resistant equipment and reduces the costs associated with hazardous waste disposal. Additionally, the high yield and purity achieved reduce the material loss during purification, directly improving the overall material balance and lowering the cost of goods sold. The use of common solvents like ethyl acetate and dichloromethane ensures that raw materials are readily available from multiple suppliers, mitigating the risk of supply chain disruptions. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined process eliminates multiple neutralization and washing steps, significantly reducing utility consumption and labor hours per batch. By avoiding the use of specialized catalysts or high-pressure reactors, the capital expenditure required for setting up production lines is drastically lowered. The high selectivity of the reaction minimizes the formation of hard-to-remove impurities, which reduces the solvent volume needed for chromatography or recrystallization. These cumulative efficiencies result in a more economical production process that enhances profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available reagents like tripropyl orthoformate ensures a consistent supply of key inputs regardless of market fluctuations. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality, providing a buffer against supply chain volatility. Furthermore, the simplified purification process shortens the manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to customer demand. This agility is a key differentiator in the competitive landscape of fine chemical intermediates.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as reduced solvent usage and safer reagents, align with increasingly strict environmental regulations. The absence of heavy metal catalysts or toxic halides simplifies the environmental impact assessment and permits easier approval for new production facilities. Scalability is enhanced by the exothermic nature of the reaction being manageable under standard reflux conditions, allowing for safe expansion from kilogram to tonne scale. This ensures that the supply can grow in tandem with the client's clinical or commercial needs without requiring process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and practical application data to provide clarity for decision-makers.
Q: How does this method control genotoxic impurities?
A: The orthoformate route avoids the use of alkyl halides or harsh sulfonyl chlorides, significantly reducing the risk of forming genotoxic alkyl sulfonate byproducts common in conventional methods.
Q: Is this process scalable for industrial production?
A: Yes, the method utilizes common solvents and mild conditions, eliminating the need for specialized high-pressure equipment, which facilitates easy scale-up from laboratory to commercial tonnage.
Q: What is the purity profile of the final ester?
A: The process yields products with high purity, demonstrated by HPLC analysis showing levels above 96%, meeting stringent requirements for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxy Benzene Sulfonate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic technologies to deliver high-value intermediates to the global market. Our technical team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this orthoformate method are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the required 96%+ purity standards and genotoxic impurity limits. Our commitment to quality assurance means that clients can rely on us for consistent supply of critical building blocks for their drug development programs.
We invite procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By leveraging this optimized synthesis route, we can help you reduce lead time for high-purity pharmaceutical intermediates while maintaining full regulatory compliance. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your next campaign. Together, we can build a more efficient and sustainable supply chain for the future of pharmaceutical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
