Advanced One-Pot Synthesis of Asymmetric Cy Fluorescein for Commercial DNA Sequencing
Advanced One-Pot Synthesis of Asymmetric Cy Fluorescein for Commercial DNA Sequencing
The landscape of fluorescent labeling for next-generation sequencing (NGS) is undergoing a significant transformation driven by the need for cost-effective and high-purity reagents. Patent CN109320445B introduces a groundbreaking synthesis method for Cy series fluorescein that addresses the longstanding challenges of asymmetric dye production. Traditionally, synthesizing asymmetric cyanine dyes involved cumbersome multi-step processes with low yields and difficult purification requirements. This new technical disclosure outlines a robust one-pot methodology that not only streamlines the reaction pathway but also optimizes the physicochemical properties of the resulting dyes, specifically targeting moderate water solubility which is crucial for efficient bioconjugation. For R&D directors and procurement specialists in the life sciences sector, this represents a pivotal shift towards more scalable and economically viable manufacturing of high-performance fluorescent probes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of asymmetric Cy dyes such as Cy3, Cy5, and their derivatives has been plagued by synthetic inefficiencies that drive up costs and limit supply chain reliability. Conventional routes typically rely on stepwise condensation reactions where intermediates must be isolated and purified, often requiring expensive preparative High-Performance Liquid Chromatography (HPLC). This reliance on HPLC is a major bottleneck; it is time-consuming, requires specialized equipment, and results in significant material loss, leading to overall yields that are often commercially unviable for large-scale applications. Furthermore, traditional methods frequently struggle with selectivity, producing mixtures of symmetric and asymmetric byproducts that are chemically similar and notoriously difficult to separate, thereby compromising the purity required for sensitive DNA sequencing applications.
The Novel Approach
The innovative strategy detailed in the patent fundamentally reengineers the synthesis workflow by converting the final two critical steps into a seamless one-pot reaction. Instead of isolating the key polymethine intermediate, the process generates it in situ and immediately reacts it with a second heterocyclic component. This approach drastically reduces the number of unit operations and eliminates the need for intermediate isolation. By carefully controlling reaction conditions and solvent systems, the method achieves high selectivity for the asymmetric product while allowing for purification via standard silica gel column chromatography rather than preparative HPLC. This shift not only simplifies the operational complexity but also significantly enhances the throughput potential for industrial manufacturing.
This general synthetic route illustrates the convergence of two distinct indole derivatives via a polymethine bridge, highlighting how the one-pot technique bypasses intermediate isolation. The ability to tune the substituents on the indole rings allows for precise control over the emission wavelength and solubility profile, enabling the production of a diverse library of dyes including Cy3.5, Cy5, and Cy7 variants tailored for specific spectral requirements in multiplexed sequencing.
Mechanistic Insights into One-Pot Asymmetric Condensation
The success of this synthesis hinges on a sophisticated understanding of reaction kinetics and nucleophilic susceptibility within the polymethine chain formation. The mechanism involves the initial formation of a quaternary ammonium salt from an indole derivative and a halo-acid, creating a carboxyl-functionalized intermediate. This intermediate is then condensed with a linker precursor, such as N,N-diphenylformamidine or its derivatives, to generate a reactive electrophilic species. The critical mechanistic insight revealed is the order of addition; the intermediate with lower reactivity must be introduced first to form the initial half-dye structure. Only after this species is fully formed is the second, more reactive indole derivative added to complete the conjugation. This sequential addition prevents the self-condensation of the more reactive component, which would otherwise lead to symmetric impurities.
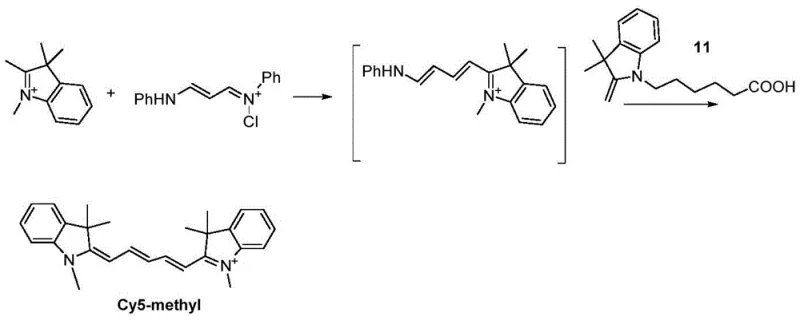
The image above demonstrates the consequence of incorrect addition order, where adding the highly reactive intermediate first leads to the formation of symmetric byproducts like Cy5-methyl instead of the desired asymmetric dye. This mechanistic control is vital for ensuring high purity without resorting to extensive chromatographic separation. By maintaining the reaction in a single vessel, the thermal energy and chemical potential are managed to favor the cross-coupling reaction over homocoupling. This level of control ensures that the final product possesses the intended asymmetric structure with distinct functional groups at each end, which is essential for subsequent conjugation to biomolecules like nucleotides.
How to Synthesize Asymmetric Cy Fluorescein Efficiently
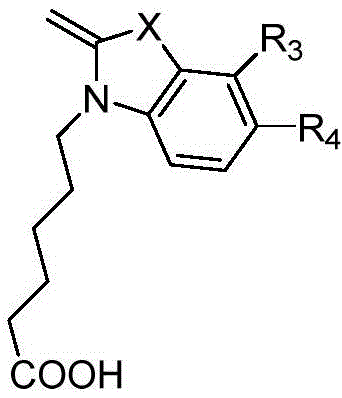
Implementing this synthesis protocol requires precise adherence to the stoichiometric ratios and temperature profiles outlined in the patent data. The process begins with the quaternization of the indole nucleus, followed by the in situ generation of the polymethine chain. The key to success lies in the direct addition of the second coupling partner without workup, leveraging the reactivity of the intermediate species. Detailed standardized synthesis steps for specific variants like Cy3.5-C and Cy5-C are provided in the technical documentation below, ensuring reproducibility across different batch sizes.
- React indole derivatives with halo-acids to form quaternary ammonium intermediates carrying a carboxyl group.
- Condense the carboxyl-bearing intermediate with a polymethine linker precursor in acetic anhydride to form a reactive intermediate in situ.
- Directly add a second indole derivative with different substituents to complete the asymmetric coupling without isolating the intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot synthesis method offers tangible benefits that extend beyond mere technical novelty. The elimination of preparative HPLC purification steps represents a massive reduction in operational expenditure (OPEX) and capital expenditure (CAPEX). HPLC solvents are expensive, and the columns have limited lifespans; replacing this with silica gel chromatography significantly lowers the cost of goods sold (COGS). Furthermore, the simplified workflow reduces the turnaround time for production batches, enhancing the agility of the supply chain to respond to fluctuating market demands for sequencing reagents. The robustness of the reaction conditions also implies a lower risk of batch failure, ensuring a more consistent and reliable supply of critical raw materials.
- Cost Reduction in Manufacturing: The primary driver for cost reduction is the removal of high-cost purification technologies. By avoiding preparative HPLC, manufacturers save significantly on solvent consumption, column media, and instrument maintenance. Additionally, the higher overall yield achieved through the one-pot method means less raw material is wasted on byproducts. The use of conventional solvents like acetonitrile and acetic acid, which are readily available and inexpensive, further contributes to a leaner cost structure compared to methods requiring exotic reagents or complex solvent exchanges.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup reduces the dependency on specialized equipment and highly skilled operators for complex purification tasks. This democratization of the manufacturing process allows for easier scaling across multiple production sites, mitigating the risk of supply disruptions. The use of stable, commercially available starting materials ensures that raw material sourcing remains secure, preventing bottlenecks that often plague the supply chains of specialty fine chemicals. This reliability is crucial for maintaining the continuous production schedules required by the high-volume DNA sequencing industry.
- Scalability and Environmental Compliance: Scaling a one-pot reaction is inherently more straightforward than scaling a multi-step process with multiple isolation points. Fewer unit operations mean less waste generation and lower energy consumption for heating and cooling cycles. The reduction in solvent usage, particularly the avoidance of large volumes of HPLC-grade solvents, aligns with green chemistry principles and simplifies waste treatment protocols. This environmental efficiency not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations governing chemical manufacturing facilities.
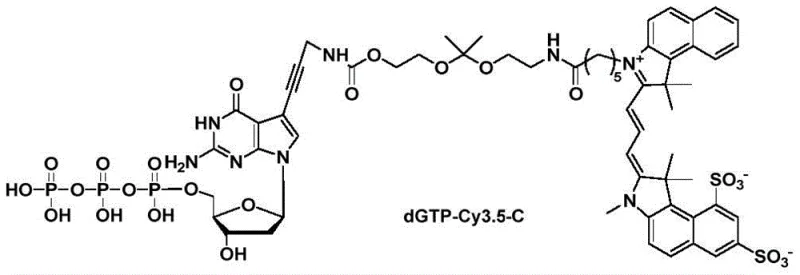
The application data highlights how dyes with moderate water solubility, such as Cy3.5-C synthesized via this method, improve the efficiency of downstream nucleotide conjugation. Unlike highly hydrophobic dyes that suffer from poor reaction rates in aqueous buffers, or highly hydrophilic dyes that are difficult to purify, these optimized intermediates strike a balance that maximizes both synthetic yield and ease of handling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this new method for industrial production.
Q: Why is the addition order critical in asymmetric Cy dye synthesis?
A: Adding the less reactive intermediate first ensures the formation of the desired asymmetric product. Reversing the order leads to symmetric byproducts like Cy5-methyl due to self-condensation.
Q: How does this method improve purification compared to traditional routes?
A: Traditional methods often require preparative HPLC. This one-pot method allows for purification using standard silica gel column chromatography, significantly lowering processing costs.
Q: What is the advantage of moderate water solubility in these dyes?
A: Moderate solubility balances reaction efficiency in aqueous buffers with ease of purification, avoiding the extreme hydrophobicity or hydrophilicity that complicates downstream nucleotide conjugation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cy Series Fluorescein Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorescent dyes play in advancing genomic research and diagnostic capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cy series fluorescein meets the exacting standards required for DNA sequencing applications. Our commitment to technical excellence allows us to deliver products that consistently perform in sensitive biological assays.
We invite you to collaborate with us to leverage these advanced synthesis capabilities for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall reagent costs.
