Advanced Indolocarbazole Derivatives for High-Efficiency OLED Manufacturing and Commercial Scale-Up
The rapid evolution of the organic electronics sector demands materials that transcend the limitations of current generation technologies, particularly in the realm of organic electroluminescent devices (OLEDs). Patent CN103459391A introduces a groundbreaking class of indolocarbazole and indenocarbazole derivatives, specifically defined by Formula (I) and Formula (II), which address critical bottlenecks in device longevity and operational voltage. These compounds represent a significant leap forward in the design of matrix materials and charge transport layers, offering a robust solution for next-generation display and lighting applications. By integrating fused aromatic systems with specific bridging groups, the invention achieves a balance of high thermal stability and efficient charge carrier mobility that was previously difficult to attain with conventional biphenyl-carbazole architectures. For procurement specialists and R&D directors seeking a reliable OLED material supplier, understanding the structural nuances of these derivatives is essential for securing a competitive advantage in electronic chemical manufacturing.
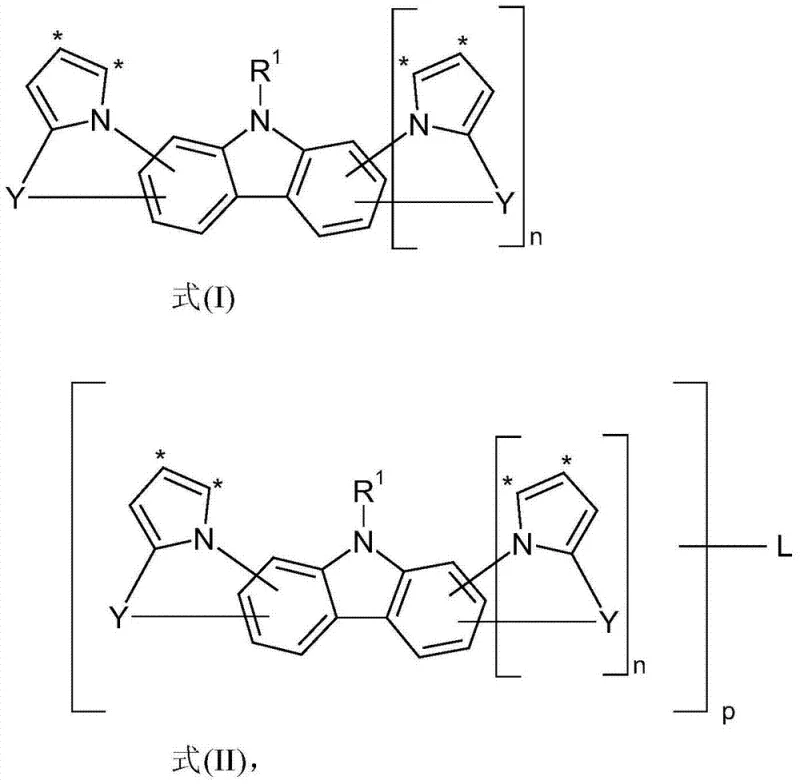
The development of these materials is driven by the urgent need to overcome the performance ceilings of existing host materials. Traditional matrix materials, such as bis(carbazolyl)biphenyl (CBP) or various metal complexes like BAlq, have served the industry well but suffer from inherent drawbacks including limited thermal stability, susceptibility to hydrolysis, and relatively high operating voltages that impact power efficiency. In particular, blue-emitting devices have historically struggled with short operational lifetimes due to the high energy excitons involved, which accelerate material degradation. The compounds disclosed in this patent are engineered to mitigate these failure modes by introducing rigid, fused ring systems that resist morphological changes and chemical decomposition under electrical stress. This structural innovation translates directly into enhanced device reliability, making them an ideal candidate for high-purity OLED material applications where consistency is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional approaches to OLED host material synthesis often rely on simple linkage strategies, such as single bonds between carbazole units or the use of ketone and phosphine oxide moieties that, while effective for electron transport, introduce polarity issues and stability concerns. Metal complexes, although capable of good efficiency, frequently exhibit sensitivity to moisture and require complex purification processes to remove residual metal catalysts, which can act as quenching sites in the final device. Furthermore, the glass transition temperatures of many standard hosts are insufficient for the rigorous thermal demands of vacuum deposition processes, leading to crystallization within the thin film and subsequent device failure. These limitations necessitate a shift towards all-organic, fused-ring architectures that offer intrinsic stability without the baggage of metal coordination chemistry or labile functional groups.
The Novel Approach
The novel approach detailed in the patent leverages the synthesis of indolo[1,2-a]indole and related fused skeletons, creating a rigid planar or semi-planar structure that significantly elevates the glass transition temperature and thermal decomposition point. By fusing the indole unit directly onto the carbazole or benzene core, the molecular freedom of rotation is restricted, which suppresses non-radiative decay pathways and enhances the overall quantum efficiency of the device. This structural rigidity also facilitates high-purity sublimation, a critical step in OLED manufacturing, as the compounds can be vaporized without breaking down. The versatility of the synthesis allows for the attachment of various aryl and heteroaryl groups via robust carbon-nitrogen bonds, enabling fine-tuning of the HOMO-LUMO levels to match specific dopant requirements. This methodology not only improves performance metrics but also streamlines the supply chain by utilizing scalable coupling reactions.
Mechanistic Insights into Palladium-Catalyzed Cyclization and Ullmann Coupling
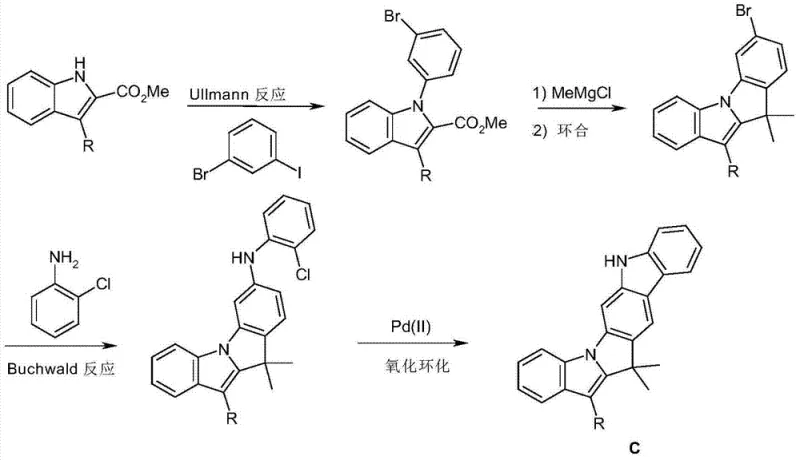
The synthesis of these advanced materials relies on a sophisticated sequence of organometallic transformations, primarily centering on Ullmann coupling and palladium-catalyzed oxidative cyclization. The process begins with the coupling of an indole derivative with a halogenated benzene precursor, forming a C-N bond that serves as the foundation for the fused system. This is followed by a Grignard addition to a carboxylate group, generating a tertiary alcohol intermediate that is primed for ring closure. The critical mechanistic step involves an acid-catalyzed cyclization, often using polyphosphoric acid or methanesulfonic acid, which dehydrates the alcohol to form the new carbon-carbon bond, locking the indole and benzene rings together. This cascade creates the core indolo-indole skeleton with high regioselectivity, ensuring that the resulting isomers (Skeleton A and B) can be separated and utilized for specific electronic properties.
Following the formation of the core skeleton, the functionality is expanded through Buchwald-Hartwig amination, where the bromine substituent on the fused ring is displaced by an amine, such as chloroaniline or other aryl amines. This step is crucial for attaching the charge-transporting carbazole units or other functional groups defined as R1 in the general formula. The final oxidative ring closure, catalyzed by palladium species, further rigidifies the structure by forming the second carbazole ring, completing the complex polycyclic aromatic system. This multi-step catalytic cycle ensures that impurities are minimized at each stage, as the conditions are optimized to favor the desired coupling over side reactions. The result is a highly pure intermediate that serves as a versatile building block for a wide range of OLED materials, demonstrating the power of modern catalytic methods in fine chemical synthesis.
How to Synthesize Indolocarbazole Derivatives Efficiently
The production of these high-performance OLED materials requires precise control over reaction conditions and stoichiometry to ensure the formation of the correct isomeric skeleton. The patent outlines a clear pathway starting from commercially available indole esters and dihalobenzenes, proceeding through distinct intermediates that can be purified via recrystallization before the final cyclization steps. This modular approach allows manufacturers to isolate key intermediates, verify their purity, and then proceed to the final coupling stages with confidence. The detailed synthesis protocol emphasizes the importance of anhydrous conditions during the Grignard step and the careful selection of ligands for the palladium catalysts to maximize yield and minimize metal residue. For those looking to implement this technology, the following guide summarizes the critical operational phases required to achieve commercial-grade quality.
- Perform Ullmann coupling between indole-2-carboxylate and bromo-iodobenzene to form the initial bridged intermediate.
- React the intermediate with methylmagnesium chloride followed by acid-catalyzed ring closure to form the indolo-indole skeleton.
- Execute Buchwald coupling with chloroaniline and subsequent palladium-catalyzed oxidative cyclization to finalize the carbazole core.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of these indolocarbazole derivatives offers substantial strategic benefits for supply chain managers and procurement officers focused on cost reduction in electronic chemical manufacturing. The synthetic route avoids the use of expensive and sensitive metal complexes that require specialized handling and disposal protocols, thereby simplifying the waste management process and reducing overall operational costs. The thermal stability of the final products means that they can withstand the rigors of large-scale vacuum sublimation without significant degradation, leading to higher recovery rates and less material waste during the purification phase. This robustness translates into a more predictable and reliable supply of high-purity materials, reducing the risk of production delays caused by batch failures or inconsistent quality.
- Cost Reduction in Manufacturing: The elimination of hydrolytically unstable metal ligands and the use of standard organometallic coupling reagents significantly lower the raw material costs associated with catalyst systems. Furthermore, the ability to purify these compounds via simple recrystallization and sublimation reduces the need for complex chromatographic separations, which are often the most expensive part of fine chemical processing. This streamlined workflow results in a lower cost of goods sold, allowing for more competitive pricing in the global OLED material market without compromising on performance specifications.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as indole carboxylates and halogenated benzenes, are commodity chemicals with established global supply chains, minimizing the risk of raw material shortages. The robustness of the chemical bonds formed during the synthesis ensures that the intermediates have a long shelf life, allowing for strategic stockpiling and buffer inventory management. This stability is crucial for maintaining continuous production lines in the fast-paced display industry, where downtime can be extremely costly.
- Scalability and Environmental Compliance: The reactions described, including Ullmann and Buchwald couplings, are well-understood industrial processes that can be easily scaled from kilogram to tonne quantities using standard reactor equipment. The absence of heavy metal residues in the final organic product simplifies environmental compliance and reduces the burden on effluent treatment plants. This aligns with increasingly stringent global regulations on hazardous substances in electronics, positioning these materials as a sustainable choice for future-proofing manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel indolocarbazole compounds in OLED device fabrication. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for making informed decisions about material selection and process integration.
Q: How do these indolocarbazole derivatives improve OLED lifetime compared to traditional CBP?
A: The fused ring structure provides higher thermal stability and glass transition temperatures, allowing sublimation without decomposition and resulting in more stable emitting layers with reduced degradation over time.
Q: Are these compounds suitable for both fluorescent and phosphorescent OLEDs?
A: Yes, depending on the substitution pattern (R1 groups), these compounds can function effectively as matrix materials for phosphorescent dopants or as hole-transport materials for fluorescent devices.
Q: What is the primary advantage of the synthetic route described in the patent?
A: The route utilizes robust organometallic couplings like Ullmann and Buchwald reactions, which are scalable and allow for precise control over the isomeric purity of the final skeleton A and B structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolocarbazole Derivatives Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for navigating the complexities of commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials, guaranteeing that every batch performs consistently in your devices. We understand the critical nature of supply continuity in the display sector and are committed to delivering high-purity OLED materials that meet the highest industry standards.
We invite you to engage with our technical procurement team to discuss how these advanced indolocarbazole derivatives can optimize your product lineup. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to this novel chemistry can reduce your overall manufacturing expenses while enhancing device performance. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique application requirements. Let us help you secure a competitive edge in the electronic materials market with our proven expertise and dedication to quality.
