Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalysis
Scalable Synthesis of Axial Chiral Indole-Naphthalene Compounds for Advanced Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and environmentally benign synthetic routes to complex chiral scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN110452150B, which discloses a novel preparation method for axial chiral indole-naphthalene compounds. These structures, characterized by their restricted rotation around a single bond leading to stable atropisomers, serve as privileged scaffolds in the design of next-generation organocatalysts and ligands. The patent introduces a robust organocatalytic strategy that constructs the axially chiral indole-naphthalene skeleton in a single step from racemic precursors. This approach represents a paradigm shift from traditional multi-step syntheses, offering a streamlined pathway to high-value intermediates that are critical for the pharmaceutical and fine chemical industries. By leveraging chiral phosphoric acid catalysis, the method achieves exceptional stereocontrol under mild conditions, addressing long-standing challenges in the synthesis of sterically hindered biaryl systems.
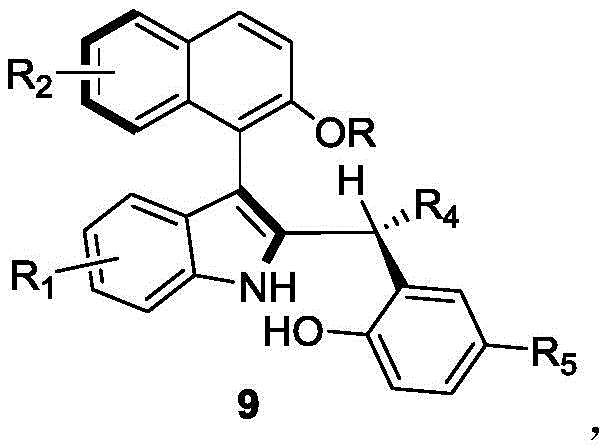
For procurement managers and supply chain directors, the implications of this technology are profound. The ability to access these complex chiral building blocks through a simplified, one-pot process translates directly into reduced manufacturing lead times and lower operational expenditures. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such proprietary synthetic routes allows us to offer clients not just a product, but a strategic advantage in their own drug development pipelines. The versatility of the substrate scope, accommodating various electron-donating and electron-withdrawing groups, ensures that a diverse library of derivatives can be generated to meet specific structure-activity relationship (SAR) requirements without necessitating entirely new process development efforts for each analog.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral indole-naphthalene compounds has been fraught with significant technical hurdles that impede large-scale adoption. Conventional strategies often rely on transition metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Ullmann-type couplings, to forge the critical carbon-carbon bond between the indole and naphthalene moieties. While effective in some contexts, these methods frequently suffer from poor atom economy and require the use of expensive palladium or copper catalysts along with specialized ligands. Furthermore, controlling the axial chirality in these coupling reactions often demands rigorous temperature control, sometimes necessitating cryogenic conditions to prevent racemization, which drastically increases energy consumption and equipment costs. Another persistent issue is the removal of trace metal residues, a critical quality attribute for pharmaceutical intermediates that adds complex purification steps and generates substantial hazardous waste. Additionally, many existing literature methods are limited in substrate scope, failing to accommodate bulky substituents that are often necessary for enhancing the catalytic activity of the final product.
The Novel Approach
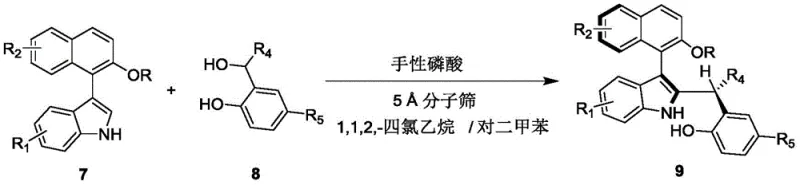
In stark contrast to these legacy methods, the technology disclosed in CN110452150B utilizes a dynamic kinetic resolution strategy mediated by a chiral phosphoric acid organocatalyst. This innovative approach enables the direct asymmetric addition of an indole derivative to a naphthol-based electrophile, constructing the axially chiral backbone with high fidelity in a single operational step. The reaction proceeds efficiently in a mixed solvent system of 1,1,2,2-tetrachloroethane and p-xylene at ambient temperatures ranging from 20 to 30°C, eliminating the need for energy-intensive heating or cooling cycles. The use of 5 Angstrom molecular sieves as an additive further drives the reaction equilibrium towards product formation by sequestering water, a byproduct of the condensation. This methodology not only simplifies the reaction setup but also dramatically improves the environmental profile of the synthesis by avoiding heavy metals. The result is a process that delivers high yields and excellent enantioselectivity (er values up to 99:1), making it an ideal candidate for cost reduction in chiral catalyst manufacturing and the production of high-purity OLED material precursors or pharmaceutical intermediates.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The success of this synthetic route lies in the sophisticated mechanism of dual activation provided by the chiral phosphoric acid catalyst. These organocatalysts, typically derived from BINOL or SPINOL backbones, function as Brønsted acids that simultaneously activate both the electrophilic and nucleophilic partners through a well-defined hydrogen-bonding network. In this specific transformation, the acidic proton of the catalyst likely activates the hydroxyl group of the naphthol derivative (compound 8), facilitating its departure or conversion into a reactive quinone methide-like intermediate. Concurrently, the basic phosphoryl oxygen interacts with the indole nitrogen or the incoming nucleophile, organizing the transition state within a rigid chiral pocket. This precise spatial arrangement forces the reaction to proceed through a specific trajectory, thereby inducing the formation of one atropisomer over the other with high selectivity. The steric bulk of the substituents on the catalyst backbone (such as the 9-anthracenyl groups in catalyst 6) plays a crucial role in shielding one face of the reacting species, ensuring that the newly formed axis of chirality is established with minimal erosion of optical purity.
From an impurity control perspective, this mechanism offers distinct advantages over radical or metal-mediated pathways. Since the reaction does not involve free radical intermediates or oxidative addition/reductive elimination cycles, the formation of homocoupling byproducts or regioisomers is significantly suppressed. The primary impurities are likely to be unreacted starting materials or diastereomers resulting from minor competing pathways, both of which are readily removed via standard silica gel chromatography. The high diastereoselectivity (dr > 95:5) observed across a wide range of substrates indicates that the chiral information is effectively transferred from the catalyst to the product, minimizing the burden on downstream purification processes. This level of control is essential for producing high-purity pharmaceutical intermediates where strict regulatory limits on impurities must be met. Furthermore, the stability of the axially chiral product under the reaction conditions prevents thermal racemization, ensuring that the optical integrity established during the bond-forming event is preserved throughout the workup and isolation stages.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves mixing the indole precursor (compound 7) and the naphthol derivative (compound 8) in a specific ratio, typically 1:1.2, to ensure complete consumption of the limiting reagent. The reaction is heterogeneous due to the presence of molecular sieves, which requires efficient stirring to maintain mass transfer. Monitoring the reaction progress via Thin Layer Chromatography (TLC) allows for precise determination of the endpoint, preventing over-reaction or degradation. Once complete, the simple filtration step removes the solid desiccant, and the solvent can be recovered and recycled, further enhancing the green chemistry metrics of the process. For detailed operational parameters and safety considerations, please refer to the standardized guide below.
- Prepare the reaction mixture by combining compound 7 and compound 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene (1: 4 v/v) with 5 Angstrom molecular sieves.
- Add the chiral phosphoric acid catalyst (compound 6, 10 mol%) to the mixture and stir at 25°C for approximately 12 hours until TLC indicates completion.
- Filter the mixture to remove molecular sieves, concentrate the filtrate, and purify the crude product via silica gel column chromatography using petroleum ether/ethyl acetate (10: 1).
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this organocatalytic platform offers compelling economic and logistical benefits. The shift from precious metal catalysis to organic small-molecule catalysis fundamentally alters the cost structure of the manufacturing process. By eliminating palladium or other transition metals, the process avoids the volatility associated with precious metal markets and removes the necessity for expensive metal scavenging resins or complex aqueous workups designed to reduce metal content to ppm levels. This simplification of the downstream processing train results in substantial cost savings and a reduced environmental footprint, aligning with modern sustainability goals. Moreover, the use of commodity chemicals as starting materials ensures a stable and resilient supply chain, mitigating the risk of shortages that often plague specialized reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts serves as a primary driver for cost optimization. Traditional methods often incur hidden costs related to catalyst recovery, metal testing, and waste disposal of heavy metal-contaminated streams. By utilizing a chiral phosphoric acid, which is used in catalytic amounts and is metal-free, the process significantly reduces raw material costs and waste treatment expenses. Additionally, the mild reaction temperature (25°C) means that energy consumption for heating or cooling is negligible compared to processes requiring reflux or cryogenic conditions. The high atom economy of the addition reaction ensures that the majority of the mass of the starting materials ends up in the final product, minimizing waste generation and maximizing yield efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more predictable and reliable supply chain. The reagents involved, such as substituted indoles and naphthols, are commercially available or easily synthesized from bulk petrochemical feedstocks. This reduces dependency on single-source suppliers for exotic catalysts. The operational simplicity of the reaction—requiring only standard glassware or stainless steel reactors without specialized pressure or temperature controls—means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging demand. The short reaction time (approximately 12 hours) allows for high throughput, enabling manufacturers to respond quickly to market needs and reduce inventory holding times.
- Scalability and Environmental Compliance: Scaling this process from grams to tons presents minimal technical risk. The absence of exothermic hazards associated with strong oxidants or unstable organometallic reagents makes the process inherently safer for large-scale operations. The solvent system, while requiring appropriate handling, is manageable within standard industrial hygiene frameworks. Furthermore, the reduction in hazardous waste, particularly heavy metal sludge, simplifies regulatory compliance and lowers the cost of environmental permits. This 'green' profile is increasingly becoming a prerequisite for supplying major pharmaceutical companies who are under pressure to reduce the carbon footprint of their supply chains. The ability to produce high-value chiral intermediates with such a favorable environmental profile positions this technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axial chiral compounds. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is crucial for R&D teams evaluating this route for their specific projects and for procurement officers assessing the feasibility of long-term supply agreements. We encourage potential partners to review these insights to fully appreciate the strategic value of this synthetic methodology.
Q: What are the key advantages of this organocatalytic method over traditional metal-catalyzed coupling?
A: This method utilizes a chiral phosphoric acid organocatalyst, eliminating the need for expensive and toxic transition metals. This significantly simplifies downstream purification, reduces heavy metal residue risks in API intermediates, and lowers overall production costs by avoiding complex metal scavenging steps.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the reaction operates under mild conditions (20-30°C) using economically available solvents and reagents. The simple workup procedure involving filtration and standard column chromatography makes it highly suitable for commercial scale-up from kilogram to multi-ton quantities without requiring specialized cryogenic equipment.
Q: What is the optical purity achievable with this method?
A: The patent data demonstrates excellent enantioselectivity, with enantiomeric ratios (er) reaching up to 99:1 (e.g., product 12 derived from 9aa). The dynamic kinetic resolution strategy effectively converts racemic starting materials into high-purity axially chiral products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
The emergence of efficient organocatalytic routes for complex chiral scaffolds marks a new era in fine chemical synthesis, and NINGBO INNO PHARMCHEM is at the forefront of translating these academic breakthroughs into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory can be reliably reproduced on an industrial scale. We understand that the transition from milligram to ton-scale requires more than just copying a recipe; it demands a deep understanding of process engineering, thermodynamics, and impurity profiling. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess required for high-performance catalytic applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of specific derivatives or a full-scale supply of the core scaffold, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall cost of goods sold. Let us be your partner in navigating the complexities of chiral chemical manufacturing.
