Industrial Scale Synthesis of Novel Sweet Taste Inhibitors for Advanced Food Formulations
Industrial Scale Synthesis of Novel Sweet Taste Inhibitors for Advanced Food Formulations
The global food industry faces a persistent challenge in balancing consumer demand for sweetness with the negative sensory attributes of high sugar loads, often described as 'greasy' or 'cloying'. Addressing this requires sophisticated flavor modifiers that can block sweet receptors without introducing off-notes. Patent CN112876354B introduces a breakthrough class of 2-(4-methoxyphenoxy)propionic acid derivatives designed specifically to inhibit sweetness while minimizing the sour aftertaste associated with traditional inhibitors. This technology represents a significant leap forward for formulators seeking clean-label solutions that maintain texture and mouthfeel without excessive sucrose. The structural versatility of these compounds allows for fine-tuning sensory profiles, making them invaluable assets for modern low-sugar product development.
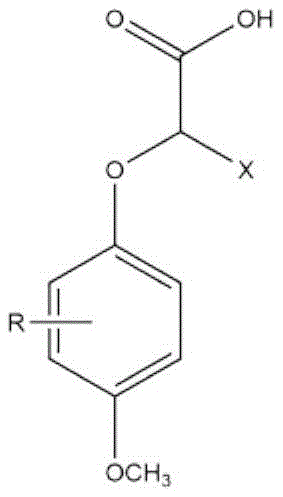
These novel compounds function by interacting with sweet taste receptors to modulate signal transduction, effectively lowering the perceived intensity of sugars like sucrose and fructose. Unlike previous iterations which often suffered from poor solubility or intense acidity, the derivatives disclosed in this patent feature specific substitutions on the phenoxy ring and the propionic acid chain. The general structure allows R to represent C1-C4 alkyl or alkoxy groups, nitro groups, or halogens, while X can be hydrogen or small alkyl chains. This modular chemical architecture enables manufacturers to optimize the balance between inhibition efficacy and sensory neutrality, providing a robust toolkit for the reliable food additive supplier looking to diversify their portfolio with high-performance ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(4-methoxyphenoxy)propionic acid and its analogues has been plagued by harsh reaction conditions that compromise both safety and product quality. Prior art methods, such as those cited in the background of the patent, often require reaction temperatures as high as 135°C to drive the etherification process to completion. Such extreme thermal stress not only increases energy consumption significantly but also promotes side reactions that generate difficult-to-remove impurities. Furthermore, existing protocols frequently result in oily reaction mixtures that are notoriously difficult to purify, leading to low yields and inconsistent batch-to-batch quality. The reliance on aqueous or anhydrous ethanol systems in older patents often fails to fully dissolve the starting phenolic materials, creating heterogeneous reaction environments that hinder kinetics and reduce overall process efficiency.
The Novel Approach
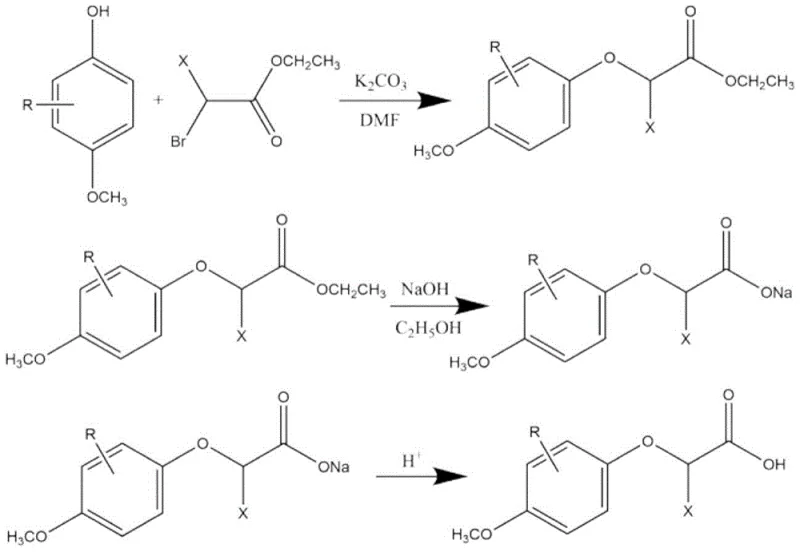
The industrial production method disclosed in CN112876354B overcomes these historical bottlenecks through a refined two-step synthetic strategy utilizing dimethylformamide (DMF) as a superior polar aprotic solvent. By shifting the reaction medium to DMF, the process ensures complete solubility of the p-methoxy substituted phenols and ethyl 2-bromoalkanoates, facilitating a homogeneous reaction environment that proceeds efficiently at much milder temperatures between 50°C and 80°C. This reduction in thermal load drastically minimizes the formation of thermal degradation byproducts, directly contributing to the high purity specifications of the final product. The subsequent workup involves a straightforward liquid-liquid extraction and recrystallization protocol that converts what was previously an intractable oil into a filterable white solid, streamlining the isolation process and enhancing cost reduction in food additive manufacturing.
Mechanistic Insights into Etherification and Saponification
The core of this synthetic route relies on a classic Williamson ether synthesis mechanism, optimized for industrial scalability through precise stoichiometric control. In the first stage, the phenolic hydroxyl group of the substituted phenol is deprotonated by potassium carbonate to form a nucleophilic phenoxide ion. This ion then attacks the electrophilic carbon of the ethyl 2-bromoalkanoate in an SN2 substitution reaction, displacing the bromide leaving group to form the ester intermediate. The use of potassium carbonate as a mild base is critical; it is strong enough to drive the deprotonation but mild enough to prevent unwanted hydrolysis of the ester moiety during the etherification step. The reaction is typically run with a slight excess of the alkylating agent (molar ratio 1:1.1-1.5) to ensure complete consumption of the valuable phenolic starting material, maximizing atom economy.
Following the formation of the ester intermediate, the process transitions to a saponification step to reveal the free carboxylic acid functionality essential for biological activity. The ester is refluxed with sodium hydroxide in ethanol, where the hydroxide ion attacks the carbonyl carbon of the ester group. This nucleophilic acyl substitution cleaves the ethoxy group, generating the sodium salt of the target acid. The subsequent acidification step, adjusting the pH to 1-2 using hydrochloric or sulfuric acid, protonates the carboxylate anion, causing the target 2-(4-methoxyphenoxy)propionic acid derivative to precipitate out of the aqueous solution. This pH-controlled precipitation acts as a primary purification event, separating the acidic product from neutral organic impurities, thereby ensuring the high-purity sweet taste inhibitors required for sensitive food applications.
How to Synthesize 2-(4-methoxyphenoxy)propionic Acid Derivatives Efficiently
Implementing this synthesis at a commercial scale requires strict adherence to the optimized parameters regarding temperature, solvent ratios, and purification sequences to guarantee consistent quality. The process is designed to be robust, tolerating minor variations while maintaining the critical purity threshold of greater than 95%. Operators must focus on the efficient removal of the high-boiling DMF solvent during the workup phase, typically achieved through aqueous extraction with organic solvents like n-heptane or ethyl acetate. The final recrystallization from alcohols such as ethanol or isopropanol is the key determinant of physical appearance and residual solvent levels. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Perform etherification by reacting p-methoxy substituted phenol with ethyl 2-bromoalkanoate and potassium carbonate in dimethylformamide at 50-80°C.
- Conduct saponification by refluxing the resulting ester with sodium hydroxide in ethanol, followed by acidification to pH 1-2.
- Purify the crude solid product through recrystallization using alcohols such as ethanol or methanol to achieve purity greater than 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical performance. The shift to milder reaction conditions fundamentally alters the cost structure of production by reducing energy demands and extending the lifespan of reactor vessels and heating jackets. Furthermore, the transition from difficult-to-handle oily intermediates to crystalline solids simplifies logistics, storage, and handling, reducing the risk of spillage and contamination during transfer operations. This process reliability translates directly into supply chain stability, ensuring that delivery schedules are met without the delays often caused by complex purification struggles or failed batches in traditional high-temperature processes.
- Cost Reduction in Manufacturing: The elimination of extreme high-temperature requirements (dropping from 135°C to under 100°C) results in substantial energy savings per kilogram of product produced. Additionally, the use of common, commodity-grade solvents like DMF and ethanol, combined with the avoidance of expensive chromatographic purification, significantly lowers the raw material and processing costs. The high yield and purity achieved reduce the need for re-processing or blending, further optimizing the overall cost of goods sold and enhancing margin potential for downstream applications.
- Enhanced Supply Chain Reliability: The robustness of the DMF-based solvent system ensures consistent reaction kinetics regardless of minor fluctuations in ambient conditions, leading to predictable cycle times. The ability to isolate the product as a stable solid via filtration rather than complex distillation of oils minimizes equipment downtime for cleaning and maintenance. This operational simplicity allows for faster turnaround between batches, increasing overall plant throughput and ensuring a steady flow of inventory to meet the rigorous demands of global food and beverage manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, extraction, and crystallization that are standard in multi-ton chemical facilities. The waste streams generated are primarily aqueous salts and recoverable organic solvents, which are easier to treat and manage compared to the complex tarry residues often associated with high-thermal-stress reactions. This alignment with greener chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals by reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel sweet taste inhibitors. Understanding the nuances of the synthesis and the specific sensory benefits of the derivatives is crucial for integrating them into new product formulations. The answers provided are derived directly from the experimental data and technical disclosures within the patent literature, ensuring accuracy and relevance for R&D and procurement teams evaluating this technology for potential adoption.
Q: How does this derivative compare to standard 2-(4-methoxyphenoxy)propionic acid?
A: While maintaining similar sweetness inhibition rates around 30%, the novel derivatives described in patent CN112876354B exhibit significantly reduced sour taste stimulation, solving a major sensory limitation of the parent compound.
Q: What are the critical reaction conditions for high yield?
A: The process utilizes dimethylformamide (DMF) as a polar aprotic solvent to ensure solubility of raw materials, with reaction temperatures strictly controlled below 100°C to prevent decomposition and ensure high purity.
Q: Is the purification process scalable for industrial production?
A: Yes, the method avoids complex chromatography, relying instead on standard liquid-liquid extraction and recrystallization, which are highly scalable unit operations suitable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-methoxyphenoxy)propionic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced flavor modifiers play in the next generation of reduced-sugar food products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest industry standards. Our capability to handle complex etherification and hydrolysis chemistries positions us as a strategic partner for companies seeking to secure a stable supply of high-performance sweet taste inhibitors.
We invite you to collaborate with us to optimize your supply chain and enhance your product formulations. By leveraging our manufacturing expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical metrics rather than theoretical projections.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
