Advanced Synthesis of Low-Osmotic Triiodo-Benzene Contrast Agent Intermediates for Commercial Scale
Introduction to Next-Generation Contrast Agent Technology
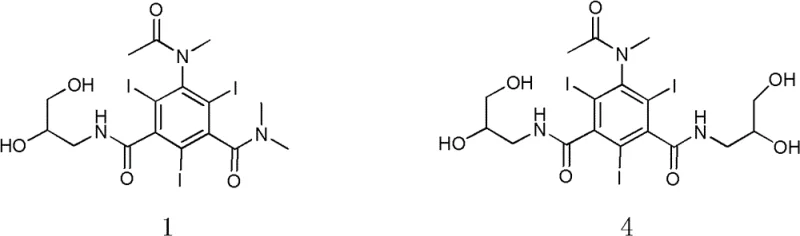
The field of diagnostic imaging has long sought a balance between radiopacity and biocompatibility, a challenge addressed comprehensively in patent CN102516116B. This pivotal intellectual property introduces a novel non-ionic monomer X-ray contrast agent, specifically N,N-dimethyl-N'-(2,3-dihydroxy-n-propyl)-5-(N-methylacetamido)-2,4,6-triiodo-1,3-isophthalamide, referred to herein as Compound 1. Unlike earlier generations of contrast media that suffered from high osmotic pressure leading to significant physiological stress, this new compound represents a strategic evolution in molecular design. By modifying the side chains of known triiodo-benzene structures, the inventors have achieved a physicochemical profile that closely mimics human plasma osmolarity while maintaining the high iodine content necessary for effective X-ray absorption. For R&D directors and procurement specialists in the pharmaceutical sector, this patent outlines a robust pathway to producing safer diagnostic agents that mitigate the risks of erythrocyte damage and hemato-encephalic barrier infringement associated with hypertonic solutions.
The significance of this technology extends beyond mere clinical efficacy; it offers a tangible opportunity for cost reduction in contrast agent manufacturing through streamlined purification processes. Traditional methods often rely on complex chromatographic separations to remove toxic byproducts and inorganic salts, which drives up production costs and limits scalability. In contrast, the methodology described in CN102516116B leverages accessible macroporous adsorption resins and ion-exchange technologies to achieve high-purity standards (>98%) efficiently. This approach not only simplifies the downstream processing but also enhances the environmental profile of the synthesis by reducing solvent waste. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is crucial for partners looking to secure a competitive edge in the global market for diagnostic imaging supplies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of X-ray contrast agents has been plagued by a trade-off between viscosity and osmotic pressure. First-generation high-osmotic agents, such as Urogranoic acid, exhibited osmotic pressures 5 to 8 times that of human blood, causing severe side effects including cardiac insufficiency and vascular pain. While second-generation non-ionic monomers like Iopromide and Ioversol reduced toxicity, they still maintained osmotic pressures approximately twice that of plasma (around 600 mOsm/kg H2O). Furthermore, third-generation non-ionic dimers, while iso-osmotic, suffered from excessively high viscosity, complicating injection procedures and requiring heated injectors. These limitations underscore the urgent need for a molecule that combines the low viscosity of monomers with the low osmotic pressure of dimers, a gap that conventional synthetic routes have struggled to fill without compromising iodine content or solubility.
The Novel Approach
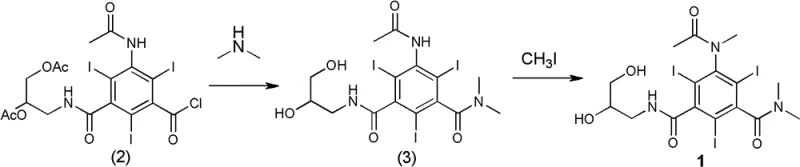
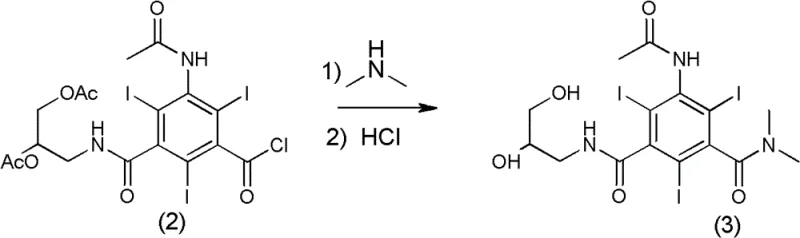
The innovative strategy presented in this patent circumvents these historical bottlenecks by introducing a specific N-methylation on the acetamido group of the triiodo-benzene core. This subtle structural modification, transforming the acetamido group into an N-methylacetamido group while retaining the dihydroxypropyl side chain, results in a dramatic reduction in osmotic pressure to 336 mOsm/kg H2O. This value is remarkably close to the physiological baseline of 320 mOsm/kg H2O, significantly enhancing patient tolerance. The synthesis route is equally ingenious, utilizing a two-step sequence that avoids harsh conditions and expensive catalysts. The process begins with the aminolysis of a triiodo-benzoyl chloride derivative, followed by a selective N-methylation using methyl iodide. This streamlined approach ensures high yield and purity while minimizing the formation of difficult-to-remove impurities.
Mechanistic Insights into N-Methylation and Resin Purification
The core chemical transformation in this process involves a nucleophilic substitution followed by an alkylation, both of which are optimized for industrial feasibility. In the first step, the acid chloride functionality of the precursor reacts with dimethylamine in an aqueous medium. This reaction is exothermic and requires careful temperature control between -15°C and 55°C to prevent hydrolysis of the sensitive triiodo-benzene ring. The presence of the three iodine atoms provides the necessary radiopacity but also adds steric bulk, which influences the reaction kinetics. The subsequent N-methylation step employs methyl iodide and a base such as sodium hydroxide in ethylene glycol monomethyl ether. This solvent system is critical as it solubilizes the polar intermediate while allowing the methylation to proceed smoothly at room temperature. The mechanism likely proceeds via the deprotonation of the acetamido nitrogen to form a nucleophilic anion, which then attacks the methyl iodide to form the stable N-methyl bond.
Beyond the reaction chemistry, the purification mechanism is a standout feature of this technology, offering significant advantages for supply chain reliability. Instead of relying on traditional recrystallization or silica gel chromatography, the process utilizes a tandem resin treatment strategy. The reaction mixture is passed through LX18 macroporous adsorption resin, which selectively adsorbs the organic product while allowing inorganic salts and highly polar impurities to pass through. This is followed by treatment with a mixed bed of weak anionic (D301) and strong cationic (732) resins. This dual-resin system effectively scavenges residual iodide ions, sodium cations, and other ionic byproducts generated during the neutralization and methylation steps. This rigorous purification protocol ensures that the final active pharmaceutical ingredient (API) intermediate meets stringent quality specifications without the need for solvent-intensive washing procedures, thereby reducing the overall environmental footprint and operational cost.
How to Synthesize N,N-dimethyl-N'-(2,3-dihydroxy-n-propyl)-5-(N-methylacetamido)-2,4,6-triiodo-1,3-isophthalamide Efficiently
Executing this synthesis requires precise control over reaction parameters and a disciplined approach to workup procedures to ensure maximum recovery and purity. The process is divided into two distinct stages: the formation of the intermediate amide and the final N-methylation. Operators must adhere to strict temperature profiles, particularly during the addition of dimethylamine, to manage the exotherm and prevent side reactions. The use of aqueous dimethylamine simplifies handling compared to gaseous reagents, enhancing safety in a commercial plant setting. Following the reaction, the pH adjustment to neutrality is critical before loading onto the resin columns, as extreme pH levels can degrade the resin capacity or the product itself. The detailed standardized synthesis steps below outline the specific conditions and reagent ratios required to replicate the high yields reported in the patent literature.
- React 3-acetamido-5-(2,3-diacetoxypropylcarbamoyl)-2,4,6-triiodobenzoyl chloride with dimethylamine aqueous solution at -15 to 55°C, followed by acidification and resin purification to obtain the intermediate amide.
- Perform N-methylation of the intermediate using methyl iodide and sodium hydroxide in ethylene glycol monomethyl ether, followed by ion-exchange resin treatment to isolate the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits. The primary advantage lies in the substantial cost savings derived from the purification strategy. By replacing expensive chromatographic media with regenerable macroporous and ion-exchange resins, the cost of goods sold (COGS) is significantly reduced. Resins can be reused multiple times after regeneration, whereas silica gel is typically single-use. Furthermore, the elimination of transition metal catalysts means there is no need for costly and time-consuming heavy metal scavenging steps, which are often a bottleneck in API manufacturing. This simplification of the downstream process translates directly into shorter batch cycles and higher throughput, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The process utilizes commodity chemicals such as dimethylamine, methyl iodide, and sodium hydroxide, which are readily available in the global market at stable prices. The avoidance of precious metal catalysts or exotic reagents eliminates supply chain volatility associated with rare materials. Additionally, the high atom economy of the methylation step and the efficient resin-based purification minimize raw material waste, leading to a leaner and more cost-effective production model that maximizes margin potential for large-scale contracts.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent quality and yield, which is paramount for maintaining uninterrupted supply to downstream formulation partners. The use of water and common organic solvents like tetrahydrofuran and ethylene glycol monomethyl ether reduces dependency on specialized solvent supply chains. Moreover, the mild reaction conditions (-15°C to 55°C) do not require extreme cryogenic or high-pressure equipment, reducing the risk of equipment failure and maintenance downtime. This operational stability guarantees reliable delivery schedules for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales in the patent examples. The aqueous workup and resin filtration steps generate less hazardous waste compared to traditional liquid-liquid extractions involving large volumes of halogenated solvents. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The ability to scale from 100 kgs to 100 MT annual commercial production without fundamental process changes makes this technology an ideal candidate for long-term strategic sourcing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this low-osmotic contrast agent intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing product portfolios. The answers highlight the specific advantages in terms of physiological safety, purification efficiency, and regulatory compliance that distinguish this compound from legacy alternatives.
Q: How does Compound 1 improve patient safety compared to existing contrast agents?
A: Compound 1 exhibits significantly lower osmotic pressure (336 mOsm/kg H2O) compared to traditional non-ionic monomers like Iopromide (610 mOsm/kg H2O), bringing it closer to human plasma osmotic pressure and reducing risks of renal toxicity and hemodynamic disturbance.
Q: What purification method ensures high purity without expensive chromatography?
A: The process utilizes a combination of macroporous adsorption resin (LX18) and mixed ion-exchange resins (D301 and 732) to effectively remove inorganic salts and impurities, achieving over 98% purity without the need for costly silica gel column chromatography.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the synthesis relies on commodity reagents like dimethylamine and methyl iodide under mild conditions (-15 to 55°C), and the workup involves simple filtration and evaporation steps, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-dimethyl-N'-(2,3-dihydroxy-n-propyl)-5-(N-methylacetamido)-2,4,6-triiodo-1,3-isophthalamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation diagnostic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in resin-based purification and complex organic synthesis allows us to optimize the production of Compound 1, offering you a partner who understands both the chemistry and the commercial imperatives of the pharmaceutical market.
We invite you to collaborate with us to leverage this advanced technology for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs. Let us help you bring safer, more effective contrast agents to market faster.
