Revolutionizing OLED Performance with Novel Diazadibenzofuran Carbazole Derivatives for Commercial Scale-Up
The rapid evolution of organic light-emitting diode (OLED) technology demands continuous innovation in host materials to achieve higher efficiency and extended operational lifetimes. Patent CN109790173B introduces a groundbreaking class of diazadibenzofuran or diazadibenzothiophene derivatives substituted by carbazole structures, specifically engineered for use in advanced electronic devices. These compounds address critical limitations in existing phosphorescent OLED systems by optimizing charge transport and exciton confinement within the emissive layer. The strategic integration of carbazole moieties with diaza-heterocyclic cores creates a robust molecular architecture that balances electron and hole mobility, thereby reducing driving voltage and enhancing overall power efficiency. For industry leaders seeking a reliable OLED host materials supplier, understanding the structural nuances of these derivatives is essential for next-generation display manufacturing. The patent details extensive synthetic pathways and device performance data, validating the commercial viability of these materials for large-scale production.
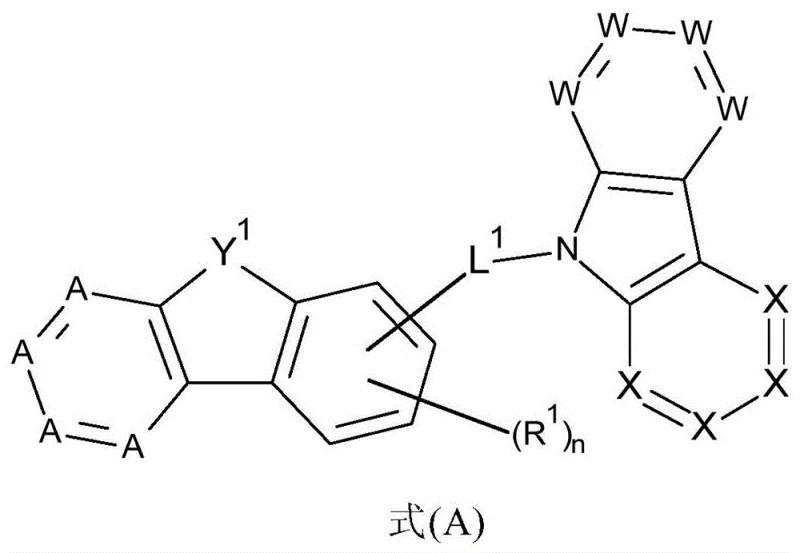
Conventional host materials often struggle with inadequate triplet energy levels or poor thermal stability, leading to efficiency roll-off and premature device failure in high-brightness applications. Traditional carbazole-based hosts, while widely used, may lack the necessary electron-transporting capabilities required for balanced charge injection in phosphorescent systems. Furthermore, many existing compounds suffer from low glass transition temperatures, which can cause morphological instability during prolonged operation or vacuum deposition processes. These limitations necessitate frequent recalibration of manufacturing lines and increase the total cost of ownership for display producers. In contrast, the novel approach described in the patent leverages the electron-deficient nature of diazadibenzofuran and diazadibenzothiophene units to complement the hole-transporting character of carbazoles. This synergistic design results in bipolar transport characteristics that are crucial for maximizing exciton formation within the emission zone. By systematically varying the substitution patterns on the aromatic rings, chemists can fine-tune the HOMO and LUMO energy levels to match specific dopant requirements, ensuring efficient energy transfer without back-transfer losses.
The novel approach utilizes sophisticated coupling reactions to assemble these complex architectures with high precision and yield. The patent outlines methods involving palladium-catalyzed cross-couplings, such as Suzuki or Buchwald-Hartwig reactions, which are well-established in industrial organic synthesis yet optimized here for these specific steric environments. This ensures that the resulting materials possess the high purity necessary for electronic applications, typically exceeding 99% after sublimation. The structural flexibility allows for the incorporation of various aryl or heteroaryl groups, enabling the customization of solubility profiles for solution-processing techniques like inkjet printing. This adaptability is a significant advantage for manufacturers looking to transition from vacuum thermal evaporation to more cost-effective solution-based manufacturing methods. Consequently, these materials offer a pathway to cost reduction in electronic chemical manufacturing by simplifying processing requirements while maintaining superior device performance metrics.
Mechanistic Insights into Diazadibenzofuran-Carbazole Coupling and Device Physics
The core mechanism behind the superior performance of these compounds lies in their ability to facilitate balanced charge transport and confine triplet excitons effectively. The diazadibenzofuran core acts as an electron-transporting unit due to the electronegative nitrogen atoms within the heterocyclic ring, while the carbazole substituent serves as an efficient hole-transporting segment. When integrated into an OLED stack, this bipolar nature ensures that the recombination zone is centered within the emissive layer, preventing exciton quenching at the electrode interfaces. The patent specifies that the linkage between these units can be direct or via a bridging group, allowing for modulation of conjugation length and steric hindrance. This control is vital for managing the glass transition temperature and morphological stability of the thin film. Moreover, the high triplet energy of the diazadibenzofuran moiety prevents energy back-transfer from the phosphorescent dopant to the host, a common failure mode in green and blue phosphorescent OLEDs. This ensures that the energy harvested from electrical input is efficiently converted into light output rather than lost as heat.
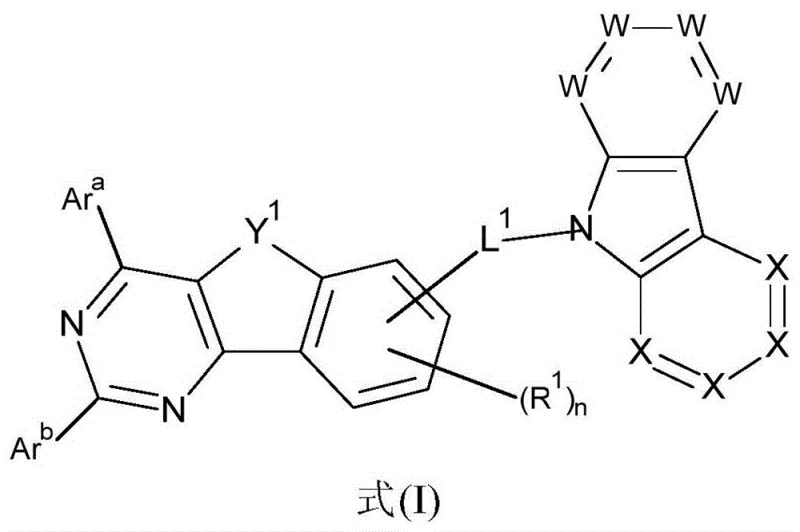
Impurity control is another critical aspect of the mechanistic design, directly impacting the operational lifetime of the final device. The synthetic routes described emphasize rigorous purification steps, including recrystallization and gradient sublimation under high vacuum, to remove trace metal catalysts and organic byproducts. Residual palladium or halogenated impurities can act as quenching sites or degradation initiators under electrical stress, significantly shortening device life. The patent highlights that compounds prepared via these methods exhibit excellent thermal stability, with decomposition temperatures well above the processing conditions required for vacuum deposition. Furthermore, the specific substitution patterns, such as the use of bulky aryl groups, inhibit close packing of molecules in the solid state, which reduces the likelihood of excimer formation that can lead to red-shifted emission and efficiency loss. This level of molecular engineering ensures that the high-purity OLED host materials delivered to production lines maintain consistent batch-to-batch performance, a key requirement for supply chain reliability.
How to Synthesize Diazadibenzofuran Carbazole Derivatives Efficiently
Synthesizing these advanced materials requires a systematic approach that balances reaction efficiency with product purity to meet the stringent standards of the optoelectronics industry. The process begins with the preparation of the halogenated diazadibenzofuran precursor, followed by sequential coupling steps to introduce the carbazole and other aryl substituents. Careful control of reaction parameters such as temperature, catalyst loading, and base selection is essential to minimize side reactions and maximize yield. The patent provides detailed examples where yields of over 80% are achieved for key intermediates, demonstrating the scalability of the chemistry. Following synthesis, the crude material undergoes multiple purification stages to ensure that electronic-grade specifications are met. Detailed standardized synthesis steps see the guide below.
- Prepare the diazadibenzofuran or diazadibenzothiophene core structure through cyclization and halogenation steps.
- Execute palladium-catalyzed coupling reactions to attach carbazole units to the core framework.
- Purify the final compound via recrystallization and sublimation to achieve electronic-grade purity exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of these novel host materials presents significant opportunities for optimizing production costs and ensuring material availability. The synthetic routes described rely on commercially available starting materials and robust catalytic systems, which mitigates the risk of supply bottlenecks associated with exotic reagents. This accessibility translates into enhanced supply chain reliability, as manufacturers can source raw materials from multiple vendors without compromising on quality. Additionally, the high yields reported in the patent examples suggest that waste generation is minimized, aligning with environmental compliance goals and reducing disposal costs. The ability to process these materials from solution further opens up avenues for reducing capital expenditure on vacuum deposition equipment, offering a flexible manufacturing strategy that can adapt to market demands. These factors collectively contribute to substantial cost savings and a more resilient supply network for electronic device producers.
- Cost Reduction in Manufacturing: The elimination of complex multi-step syntheses involving unstable intermediates streamlines the production process, leading to lower operational expenditures. By utilizing efficient coupling reactions that proceed with high conversion rates, manufacturers can reduce solvent consumption and energy usage per kilogram of product. The high thermal stability of the final compounds also means less material is lost to degradation during the deposition process, improving overall material utilization rates. Furthermore, the potential for solution processing allows for the use of additive manufacturing techniques, which are inherently less wasteful than subtractive methods. These efficiencies drive down the unit cost of the host material, making high-performance OLED displays more economically viable for a broader range of consumer applications.
- Enhanced Supply Chain Reliability: The reliance on standard organic synthesis techniques ensures that production can be scaled up rapidly to meet surges in demand without requiring specialized infrastructure. The chemical stability of the intermediates allows for safer storage and transportation, reducing the logistical complexities associated with hazardous materials. This robustness ensures that delivery schedules remain consistent, even in the face of global supply chain disruptions. Manufacturers can maintain lower inventory levels while still guaranteeing continuity of supply, freeing up working capital for other strategic investments. The versatility of the chemical platform also means that alternative derivatives can be quickly developed if specific raw materials become scarce, providing a built-in contingency plan for procurement teams.
- Scalability and Environmental Compliance: The synthetic methods are designed to be scalable from gram to ton quantities without significant re-optimization, facilitating a smooth transition from R&D to commercial production. The use of recyclable solvents and the minimization of heavy metal waste align with increasingly strict environmental regulations across global markets. This compliance reduces the regulatory burden on manufacturing sites and enhances the corporate sustainability profile of the end-product. Efficient purification via sublimation ensures that the final product meets the highest purity standards without generating excessive chemical waste. These attributes make the technology attractive for long-term investment, ensuring that production facilities remain compliant and operational for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these diazadibenzofuran carbazole derivatives in OLED manufacturing. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about material selection and process integration. For further technical support, our team is available to provide customized analysis.
Q: What are the primary advantages of these diazadibenzofuran carbazoles in OLEDs?
A: These compounds significantly improve device lifetime and efficiency while lowering operating voltage compared to conventional host materials, as demonstrated in patent examples.
Q: Can these materials be processed from solution for printing applications?
A: Yes, specific derivatives with suitable alkyl or aryl substituents exhibit enhanced solubility in standard organic solvents, enabling solution processing techniques like inkjet printing.
Q: What purity levels are required for commercial OLED production?
A: For high-performance electronic devices, purification via sublimation is recommended to achieve purity levels greater than 99%, ensuring minimal impurity-induced degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazadibenzofuran Carbazole Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise to translate these complex patent methodologies into reliable commercial supply chains. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from laboratory validation to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications required for electronic grade materials. We understand the critical nature of impurity profiles in OLED materials and employ advanced analytical techniques to guarantee batch consistency. By partnering with us, you gain access to a supply chain that prioritizes quality, speed, and regulatory compliance, allowing you to focus on device innovation while we handle the chemical complexity.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how these advanced materials can enhance your product lineup. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to these high-efficiency hosts. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your production volumes. Let us help you secure a competitive advantage in the rapidly evolving display market with materials that deliver proven performance and reliability.
