Optimizing Non-Ionic Contrast Agent Production with Advanced Triiodobenzene Chemistry
Optimizing Non-Ionic Contrast Agent Production with Advanced Triiodobenzene Chemistry
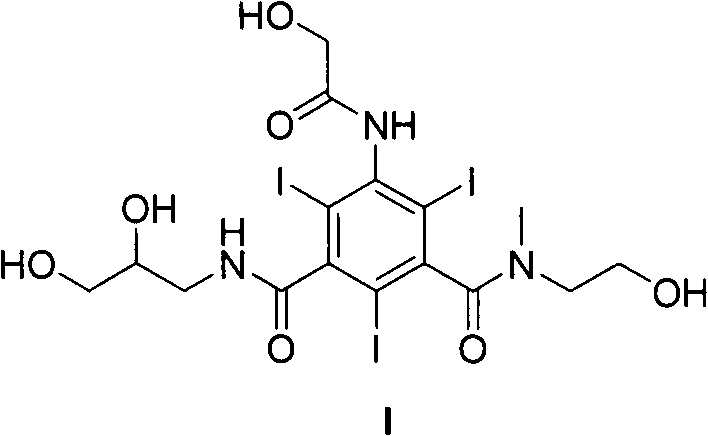
The landscape of diagnostic imaging is constantly evolving, driven by the urgent need for safer and more efficient contrast media that minimize patient discomfort while maximizing image quality. Patent CN102126986B introduces a groundbreaking triiodobenzene compound, specifically 5-(2-hydroxyacetamido)-N-methyl-N-(2-hydroxyethyl)-N'-(2,3-dihydroxy-n-propyl)-2,4,6-triiodoisophthalamide, which represents a significant leap forward in non-ionic X-ray contrast agent technology. This innovative molecule addresses critical limitations found in existing clinical monomers like Iohexol and Iomeprol, primarily by achieving a superior balance between iodine content and physicochemical properties. By strategically reducing the number of hydroxyl groups and optimizing the side-chain asymmetry, this new compound delivers markedly lower osmotic pressure and viscosity, which are paramount factors for renal safety and injection ease. For pharmaceutical manufacturers and CDMOs, mastering the synthesis of this advanced intermediate opens doors to producing next-generation imaging agents that meet the rigorous demands of modern radiology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional non-ionic contrast agents, while a vast improvement over their ionic predecessors, still suffer from inherent physicochemical drawbacks that limit their clinical utility and manufacturing efficiency. Most commercially available monomers, such as Iohexol, possess a high number of hydroxyl groups—typically six per molecule—which significantly increases the solution's viscosity and osmotic pressure relative to blood plasma. This high viscosity necessitates the use of larger gauge needles or heating of the contrast media prior to injection, complicating clinical workflows and increasing patient anxiety. Furthermore, the high osmotic load can induce adverse physiological responses, including endothelial damage and renal toxicity, particularly in vulnerable patient populations. From a manufacturing perspective, the synthesis of these highly hydroxylated molecules often involves complex protection and deprotection strategies that generate substantial chemical waste and require extensive purification steps to remove toxic byproducts, thereby inflating production costs and extending lead times for reliable contrast agent intermediate supplier networks.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues through a clever molecular design that reduces the hydroxyl count to just four while maintaining high water solubility and iodine density. By incorporating a 2-hydroxyacetamido group and optimizing the N-substitution patterns on the isophthalamide core, the new compound achieves a molecular weight of 747, which is notably lower than the 821 of Iohexol. This structural refinement results in a viscosity reduction of approximately 33.3% at room temperature compared to standard agents, allowing for smoother administration without the need for pre-heating. The synthesis route is equally innovative, utilizing a streamlined four-step process that avoids the use of expensive transition metal catalysts or hazardous reagents. This method not only enhances the purity profile by minimizing side reactions but also aligns perfectly with green chemistry principles, offering a sustainable pathway for cost reduction in pharmaceutical imaging manufacturing that appeals to both procurement teams and environmental compliance officers.
Mechanistic Insights into Acylation and Hydrolysis Pathways
The core of this synthesis lies in a highly efficient acylation strategy that builds the complex side chains onto the triiodinated benzene ring with exceptional regioselectivity. The process begins with the reaction of 3-amino-5-(2,3-diacetoxypropylcarbamoyl)-2,4,6-triiodobenzoic acid with alpha-Acetoxyacetyl chloride in a non-protonic solvent like THF. This step is critical as it installs the 5-position substituent that differentiates this molecule from prior art, creating a steric environment that discourages polymerization and ensures high yields. The subsequent conversion of the carboxylic acid to an acyl chloride using reagents such as triphosgene or oxalyl chloride is performed under mild conditions, preserving the sensitive iodine-carbon bonds on the aromatic ring. This careful control of reaction conditions prevents dehalogenation, a common pitfall in polyiodinated compound synthesis, ensuring that the final product retains the high iodine content of 51.0% necessary for effective X-ray attenuation. The mechanistic precision here allows for the production of high-purity triiodobenzene compound intermediates with minimal impurity profiles, reducing the burden on downstream purification units.
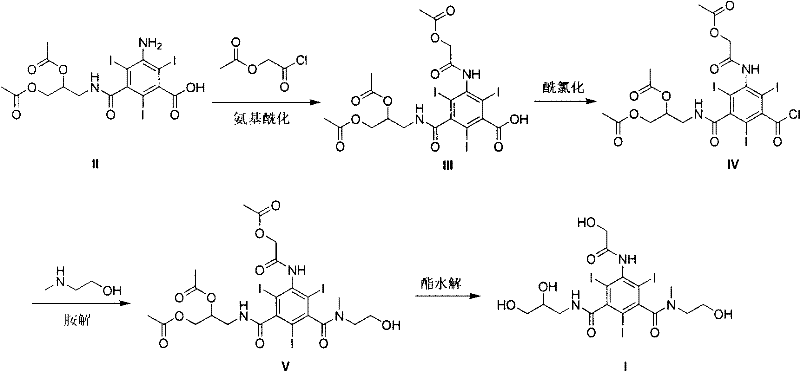
The final transformation involves a selective ester hydrolysis that reveals the free hydroxyl groups essential for water solubility without compromising the amide linkages. Using sodium methoxide in methanol at controlled low temperatures, the acetyl protecting groups are cleanly removed to yield the target molecule. This step is mechanistically favorable because the amide bonds are significantly more resistant to base-catalyzed hydrolysis than the ester groups under these specific conditions. This chemoselectivity is a major advantage for commercial scale-up of complex contrast agents, as it eliminates the need for chromatographic purification, allowing the product to be isolated via simple crystallization or resin treatment. The result is a robust process that consistently delivers material with a purity exceeding 98%, meeting the stringent quality standards required for injectable pharmaceutical products. This level of control over the reaction mechanism ensures batch-to-batch consistency, a key metric for supply chain heads managing global distribution networks.
How to Synthesize 5-(2-hydroxyacetamido)-N-methyl-N-(2-hydroxyethyl)-N'-(2,3-dihydroxy-n-propyl)-2,4,6-triiodoisophthalamide Efficiently
The synthesis of this advanced contrast agent intermediate follows a logical, four-step sequence designed for maximum yield and operational safety in an industrial setting. The process begins with the acylation of the amino-acid precursor, followed by activation of the carboxyl group, amidation with N-methylethanolamine, and concludes with a global deprotection step. Each stage has been optimized to utilize common organic solvents and reagents, minimizing the need for specialized equipment or hazardous handling protocols. The detailed standardized synthesis steps below outline the specific molar ratios, temperature ranges, and reaction times required to replicate the high yields reported in the patent literature, providing a clear roadmap for process chemists aiming to implement this technology.
- Perform amino acylation of 3-amino-5-(2,3-diacetoxypropylcarbamoyl)-2,4,6-triiodobenzoic acid with alpha-Acetoxyacetyl chloride in THF.
- Convert the resulting carboxylic acid intermediate into an acyl chloride using triphosgene or oxalyl chloride in chloroform.
- Execute aminolysis with N-methylethanolamine followed by ester hydrolysis using sodium methoxide to yield the final triiodobenzene compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple unit cost savings. The streamlined nature of the four-step synthesis significantly reduces the overall processing time and resource consumption compared to traditional methods that require more extensive protection group manipulation. By eliminating the need for transition metal catalysts, the process removes the costly and time-consuming heavy metal scavenging steps that are often bottlenecks in API manufacturing. This simplification translates directly into substantial cost savings and a more agile production schedule, allowing manufacturers to respond more quickly to market demand fluctuations. Furthermore, the use of readily available starting materials ensures a stable supply chain, mitigating the risks associated with sourcing exotic or single-source reagents that can disrupt production timelines.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the reduction in purification steps drastically lower the operational expenditure associated with producing high-purity radiopharmaceuticals. The high atom economy of the acylation and aminolysis steps ensures that raw materials are converted efficiently into the desired product, minimizing waste disposal costs. Additionally, the ability to isolate the final product through crystallization rather than chromatography significantly reduces solvent consumption and processing time, leading to a leaner manufacturing footprint. These efficiencies collectively contribute to a more competitive pricing structure for the final contrast agent, enhancing market positioning without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as THF, chloroform, and sodium methoxide ensures that the supply chain is resilient against geopolitical or market-specific disruptions. Unlike processes that depend on specialized ligands or rare earth metals, this synthesis can be executed in multiple geographic locations with standard chemical infrastructure. This flexibility allows for diversified manufacturing strategies, reducing the risk of single-point failures and ensuring continuous availability of critical imaging agents. For supply chain heads, this means reduced lead time for high-purity radiopharmaceuticals and the ability to maintain robust inventory levels even during periods of high global demand.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-atmospheric pressures, making the process inherently safer and easier to scale from pilot plant to full commercial production. The absence of toxic heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations regarding pharmaceutical effluent. The high yields achieved in each step reduce the volume of chemical waste generated per kilogram of product, supporting sustainability goals and reducing the environmental tax burden. This scalability ensures that the technology can meet growing global healthcare needs while maintaining a responsible environmental stewardship profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triiodobenzene synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the feasibility, safety, and performance characteristics of the new compound. Understanding these details is crucial for stakeholders evaluating the potential integration of this intermediate into their existing product portfolios or supply chains.
Q: How does this triiodobenzene compound compare to Iohexol in terms of viscosity?
A: The novel compound exhibits significantly lower viscosity due to a reduced hydroxyl count (4 hydroxyls versus 6 in Iohexol) and a lower molecular weight, facilitating easier injection and improved patient tolerance.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes readily available starting materials such as 3-amino-5-(2,3-diacetoxypropylcarbamoyl)-2,4,6-triiodobenzoic acid, alpha-Acetoxyacetyl chloride, and N-methylethanolamine, ensuring stable supply chains.
Q: Is the final deprotection step scalable for industrial production?
A: Yes, the final hydrolysis using sodium methoxide in methanol is a robust reaction that can be easily scaled from kilogram to metric ton quantities without requiring exotic catalysts or extreme pressure conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triiodobenzene Compound Supplier
The development of low-viscosity, low-osmolarity contrast agents represents a significant opportunity for innovation in the diagnostic imaging sector, and NINGBO INNO PHARMCHEM is uniquely positioned to support this advancement. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to market-ready product is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of triiodobenzene intermediate meets the highest international standards for safety and efficacy. We understand the critical nature of supply continuity in the healthcare industry and are committed to delivering consistent quality that supports your regulatory filings and commercial launch timelines.
We invite you to collaborate with us to optimize your contrast agent manufacturing strategy and achieve significant operational efficiencies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to deep process expertise and a reliable supply network that can accelerate your time to market. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you engineer the next generation of superior diagnostic imaging solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
