Advanced Solid-Phase Cyclization Strategy for Commercial Carbetocin Production
The pharmaceutical landscape for uterotonic agents has seen a significant shift towards more efficient manufacturing processes, exemplified by the innovations detailed in patent CN102796178A. This document outlines a robust solid-phase synthesis method for Carbetocin, a long-acting oxytocin analogue critical for preventing postpartum hemorrhage. Unlike traditional approaches that rely on cumbersome liquid-phase cyclization, this novel technique leverages a unique mercapto protecting group strategy combined with the principles of solid-phase pseudo-dilution. By anchoring the peptide chain to a Rink Amide AM Resin, the process effectively mitigates the risks of intermolecular polymerization while streamlining the purification workflow. For global procurement teams and R&D directors, this represents a pivotal advancement in peptide API manufacturing, offering a pathway to higher purity profiles and reduced environmental impact through minimized solvent usage.
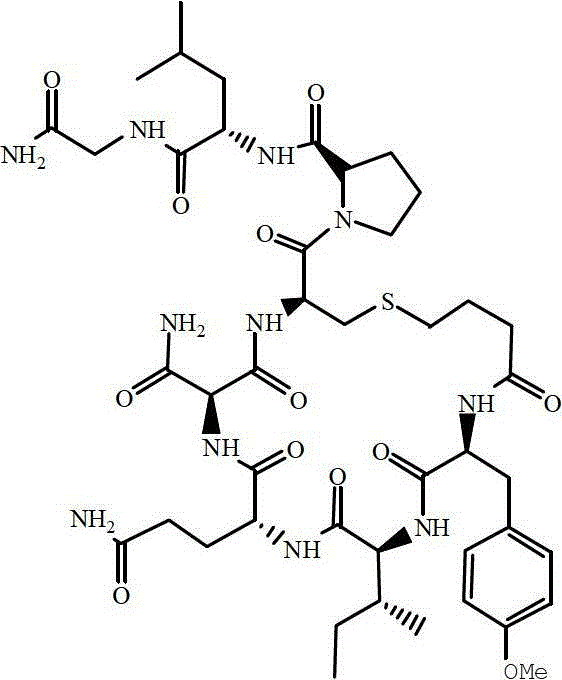
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Carbetocin has been plagued by the inefficiencies inherent in solution-phase cyclization. To successfully form the intramolecular disulfide or thioether bridge without creating intermolecular polymers, conventional methods necessitate extreme high-dilution conditions, often ranging from 10^-4 to 10^-5 mol/L. This requirement forces manufacturers to utilize vast quantities of organic solvents to maintain the necessary concentration gradients, resulting in substantial operational costs related to solvent purchase, recovery, and waste disposal. Furthermore, the reliance on allyl-based protecting groups for the cysteine side chain typically demands the use of expensive palladium catalysts and phosphine ligands for deprotection. These heavy metal reagents not only inflate the bill of materials but also introduce complex downstream purification challenges to ensure residual metal levels meet stringent pharmacopeial limits, creating a bottleneck for scalable production.
The Novel Approach
The methodology presented in CN102796178A fundamentally reengineers this workflow by transitioning the cyclization step onto the solid support. By employing a "pseudo-dilution" effect, the resin beads act as isolated micro-reactors where the growing peptide chains are physically separated from one another. This spatial isolation allows the cyclization to proceed efficiently at much higher effective concentrations compared to solution phase, drastically reducing the volume of solvents required. Additionally, the introduction of a novel cyanoethyl ester protecting group for the carboxyl functionality offers a distinct economic advantage. This group is stable during the standard piperidine-mediated Fmoc deprotection cycles but can be selectively cleaved using tetrabutyl ammonium fluoride (TBAF) under mild conditions. This eliminates the need for costly palladium scavenging steps, simplifying the overall process flow and enhancing the economic viability of large-scale manufacturing.
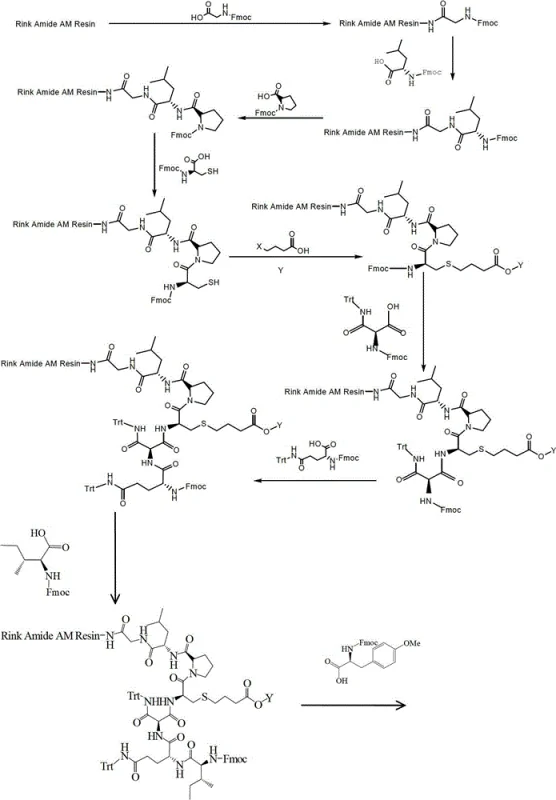
Mechanistic Insights into Solid-Phase Pseudo-Dilution Cyclization
The core mechanistic advantage of this process lies in the thermodynamic and kinetic control afforded by the solid-phase environment. In solution, the probability of two free peptide chains colliding to form a dimer is significant unless the concentration is kept infinitesimally low. However, when the C-terminus of the linear peptide is covalently bound to the insoluble Rink Amide AM Resin, the local concentration of reactive groups is effectively managed by the loading capacity of the resin (optimized at 0.1 to 0.5 mmol/g in this patent). When the protecting groups are removed to expose the reactive thiol and carboxyl moieties, the intramolecular reaction is favored because the reactive ends of the same chain are tethered in close proximity, while the diffusion of separate chains is restricted by the polymer matrix. This phenomenon, known as pseudo-dilution, ensures that the cyclization yield is maximized while suppressing the formation of oligomeric impurities that typically plague liquid-phase syntheses.
Furthermore, the chemical stability of the intermediates plays a crucial role in the final purity profile. The use of the cyanoethyl group (-COOCH2CH2CN) provides orthogonal protection that withstands the repetitive basic treatments required for Fmoc removal (20-25% piperidine in DMF). The subsequent deprotection using TBAF in THF/DMF is highly specific, cleaving the ester to reveal the free carboxylic acid necessary for the lactam ring formation without affecting other sensitive side-chain functionalities protected by Trt groups. This precise control over reactivity ensures that the cyclization proceeds cleanly, leading to the reported purity levels exceeding 99.3% after preparative HPLC. The avoidance of harsh acidic or metallic conditions during the key cyclization precursor steps preserves the integrity of the peptide backbone, minimizing racemization and degradation by-products.
How to Synthesize Carbetocin Efficiently
The synthesis protocol described in the patent offers a standardized pathway for producing high-quality Carbetocin suitable for clinical applications. The process begins with the swelling of Rink Amide AM Resin and the sequential coupling of Fmoc-protected amino acids, starting from the C-terminus. Critical to the success of this route is the mid-synthesis alkylation step where a halo-acid derivative is introduced to the cysteine sulfur, followed immediately by the cyanoethyl protection. This sequence sets the stage for the final on-resin cyclization. While the general workflow is outlined here, precise control of reaction times, temperatures (0-5°C for activation, 10-30°C for coupling), and washing protocols is essential to maintain the structural fidelity of the nonapeptide. For detailed operational parameters and stoichiometry, please refer to the technical guide below.
- Sequentially couple Fmoc-protected amino acids (Gly, Leu, Pro, Cys) onto Rink Amide AM Resin, followed by alkylation with halo-acids and cyanoethyl protection.
- Continue coupling the remaining Fmoc-amino acids (Asn, Gln, Ile, Tyr) to build the linear peptide chain on the solid support.
- Perform on-resin deprotection using TBAF to reveal the thiol and carboxyl groups, followed by solid-phase cyclization and final cleavage.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this solid-phase synthesis route translates directly into tangible operational efficiencies and risk mitigation. The primary driver of cost reduction is the elimination of precious metal catalysts. By replacing the traditional palladium/triphenylphosphine deprotection system with a fluoride-based mechanism, manufacturers can significantly lower the raw material costs associated with each batch. Moreover, the simplified workflow reduces the number of unit operations required, which in turn lowers labor costs and equipment occupancy time. The reduction in solvent volume, driven by the avoidance of high-dilution conditions, further contributes to a leaner cost structure by decreasing both the purchase price of solvents and the expenses related to their disposal and environmental compliance.
- Cost Reduction in Manufacturing: The economic model of this synthesis is bolstered by the substitution of expensive reagents with more commodity-grade chemicals. The removal of palladium catalysts not only saves on direct material costs but also eliminates the need for specialized metal scavenger resins and the associated validation testing for heavy metals. Additionally, the higher yield and purity achieved through solid-phase cyclization mean that less starting material is wasted on failed batches or extensive purification runs. This efficiency gain allows for a more predictable cost of goods sold (COGS), enabling more competitive pricing strategies in the global oxytocin analogue market.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents required for this method, such as TBAF and standard Fmoc-amino acids, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the solid-phase process also means that production timelines are more consistent, as there are fewer variables related to solvent recovery and complex work-up procedures that can delay batch release. This reliability is crucial for maintaining continuous supply to pharmaceutical partners who require just-in-time delivery of API intermediates to meet their own formulation schedules.
- Scalability and Environmental Compliance: The transition from high-dilution liquid phase to solid phase inherently supports better scalability. Managing thousands of liters of solvent for a single kilogram of product is logistically challenging and environmentally taxing. By condensing the reaction volume, this method aligns with green chemistry principles, reducing the facility's carbon footprint and hazardous waste generation. This environmental advantage is increasingly becoming a prerequisite for vendor qualification by major multinational pharmaceutical companies, making this synthesis route a strategic asset for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase Carbetocin synthesis. These insights are derived directly from the experimental data and claims within patent CN102796178A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process transfer and vendor selection.
Q: How does this solid-phase method improve upon traditional liquid-phase cyclization?
A: Traditional methods require extreme high-dilution conditions (10^-4 to 10^-5 M) to prevent polymerization, generating massive solvent waste. This patented solid-phase approach utilizes the 'pseudo-dilution' principle on the resin bead, effectively isolating peptide chains to prevent intermolecular reactions without needing vast solvent volumes.
Q: What is the advantage of the cyanoethyl protecting group used in this process?
A: The cyanoethyl group (-COOCH2CH2CN) is stable under alkaline conditions used for Fmoc removal but can be selectively removed using Tetrabutyl ammonium fluoride (TBAF). This avoids the use of expensive and toxic Palladium catalysts typically required for removing allyl-based protecting groups, significantly lowering raw material costs.
Q: What purity levels can be achieved with this synthesis route?
A: According to the patent embodiment data, this method achieves a crude peptide purity of over 99.3% with single impurities below 0.3% after purification. The solid-phase cyclization minimizes the formation of polymeric by-products common in solution-phase methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this solid-phase cyclization method are fully realized in a GMP environment. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify that every batch meets stringent purity specifications, including the low impurity profiles demonstrated in the patent literature. Our commitment to technical excellence ensures that we can deliver Carbetocin intermediates and APIs that are consistent, safe, and ready for formulation.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this solid-phase method. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term commercial goals with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
