Advanced Ionic Liquid Catalysis for High-Purity Phenylpropionate Derivatives Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical intermediates, particularly steroidal esters like nandrolone phenylpropionate and testosterone phenylpropionate. Patent CN109678718B introduces a groundbreaking methodology that utilizes imidazolium ionic liquids as catalysts to synthesize phenylpropionate derivatives in a single step. This technology represents a significant departure from conventional multi-step esterification processes, offering a route that is not only chemically elegant but also industrially robust. By employing cinnamaldehyde or its derivatives alongside various alcohol or phenol substrates, this method achieves high conversion rates without the need for additional acidic or basic additives. The implications for large-scale manufacturing are profound, as it addresses long-standing issues regarding waste generation, equipment corrosion, and operational complexity associated with traditional acylation techniques.
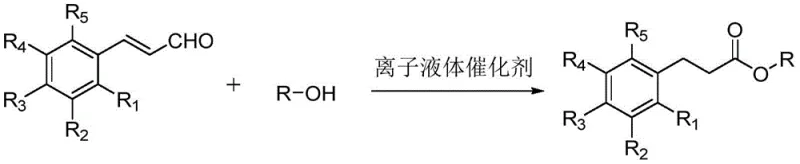
For R&D directors and process chemists, understanding the limitations of the status quo is essential for appreciating the value of this innovation. Conventional synthesis of phenylpropionate esters typically relies on a two-step sequence starting from phenylpropionic acid. The first step involves acylation using reagents such as thionyl chloride to generate phenylpropionyl chloride, an intermediate that is highly unstable and prone to hydrolysis upon exposure to moisture. This necessitates strict anhydrous conditions and specialized handling protocols. Furthermore, the subsequent esterification step generates significant amounts of acidic byproducts, requiring large quantities of alkali for neutralization during workup. These traditional methods are characterized by long production cycles, high energy consumption, and severe equipment corrosion due to the presence of hydrochloric acid and other corrosive species. The cumulative effect is a process that is not only costly but also environmentally burdensome, creating substantial challenges for waste treatment and regulatory compliance in modern chemical plants.
In stark contrast, the novel approach detailed in the patent leverages the unique properties of ionic liquids to facilitate a direct, one-step transformation. By reacting cinnamaldehyde derivatives directly with alcohols or phenols in the presence of a tailored ionic liquid catalyst, the process bypasses the formation of unstable acid chlorides entirely. This eliminates the need for hazardous acylating agents and the subsequent neutralization steps, streamlining the workflow significantly. The reaction conditions are remarkably mild yet effective, operating within a temperature range of 50°C to 150°C, with preferred embodiments functioning optimally between 80°C and 130°C. The simplicity of the post-reaction processing is another major advantage; the target phenylpropionate compounds can be isolated through straightforward extraction, concentration, or crystallization. This reduction in unit operations not only shortens the overall manufacturing timeline but also minimizes the potential for product degradation, ensuring a higher quality final material suitable for sensitive pharmaceutical applications.
Mechanistic Insights into Ionic Liquid Catalyzed Esterification
The core of this technological advancement lies in the specific structure and function of the imidazolium ionic liquid catalysts, represented generally by Formula IV in the patent documentation. These catalysts feature a cationic imidazolium ring with alkyl chains ranging from C1 to C10, paired with anions such as acetate, molybdate, or tungstate. The choice of anion plays a critical role in modulating the catalytic activity and selectivity of the reaction. For instance, tungstate and molybdate anions are known for their ability to activate carbonyl groups and facilitate nucleophilic attacks under mild conditions. The ionic liquid acts as a dual-function medium, providing both the catalytic active sites and a distinct phase that can be easily separated from the organic product. This phase behavior is crucial for the recyclability of the catalyst, as it allows the ionic liquid to remain in the aqueous layer after the organic product is extracted, enabling simple recovery via dehydration and drying for subsequent batches.
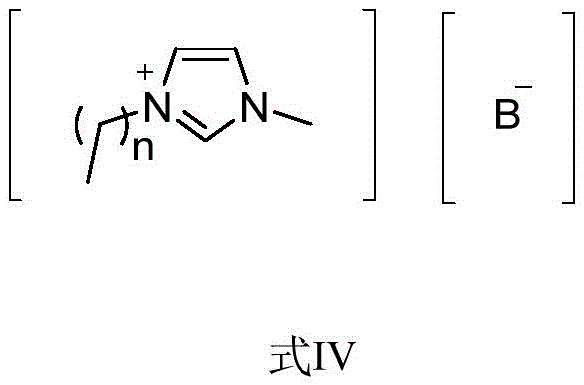
From an impurity control perspective, this mechanism offers superior cleanliness compared to acid-catalyzed esterifications. Traditional methods often suffer from side reactions such as polymerization of the aldehyde or ether formation from the alcohol, especially under harsh acidic conditions. The ionic liquid system, however, promotes the specific addition of the alcohol to the activated cinnamaldehyde derivative with high selectivity. The absence of strong mineral acids prevents the degradation of sensitive functional groups that might be present on complex substrates like steroids or terpenes. Furthermore, the high yield reported in the patent, exceeding 90% in optimized conditions, suggests that side reactions are effectively suppressed. This high selectivity translates directly to a cleaner crude product profile, reducing the burden on downstream purification processes and ensuring that the final API intermediates meet stringent purity specifications required by global regulatory bodies for pharmaceutical use.
How to Synthesize Phenylpropionate Derivatives Efficiently
Implementing this synthesis route requires careful attention to the preparation of the ionic liquid catalyst and the optimization of reaction parameters. The catalyst is typically prepared by exchanging the halide anion of an imidazolium salt with hydroxide using a strong basic ion exchange resin, followed by neutralization with the desired acid (acetic, tungstic, or molybdic). Once the catalyst is ready, the reaction proceeds by mixing the cinnamaldehyde substrate and the alcohol or phenol in a suitable solvent such as toluene or 1,4-dioxane. The molar ratio of reactants is flexible, typically ranging from 1:1 to 5:1, allowing for process optimization based on the cost and availability of raw materials. The detailed standardized synthesis steps, including specific stoichiometric ratios and workup procedures for various derivatives like n-butyl phenylpropionate or nandrolone phenylpropionate, are outlined below to guide process development teams in replicating these high-yield results.
- Prepare the imidazolium ionic liquid catalyst (e.g., [Bmim]2[WO4]) via anion exchange and acid neutralization.
- React cinnamaldehyde derivatives with alcohol or phenol substrates in a solvent like toluene or dioxane at 50-150°C.
- Isolate the product via simple extraction and crystallization, allowing for catalyst recovery from the aqueous phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid catalyzed process offers tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of corrosive reagents like thionyl chloride fundamentally alters the cost structure of manufacturing phenylpropionate derivatives. By removing the need for specialized corrosion-resistant equipment and the extensive safety measures required for handling volatile acylating agents, capital expenditure and operational maintenance costs are significantly reduced. Additionally, the simplification of the workup procedure means that less solvent and fewer auxiliary chemicals are consumed per kilogram of product. This reduction in material intensity directly contributes to a lower cost of goods sold (COGS), making the supply of these critical intermediates more economically resilient against fluctuations in raw material pricing. The ability to produce high-purity material with fewer processing steps also enhances the overall throughput of the manufacturing facility.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive and hazardous acylating agents. Traditional routes require stoichiometric amounts of reagents like thionyl chloride, which are not only costly to purchase but also expensive to dispose of safely due to the generation of acidic waste streams. By shifting to a catalytic system that utilizes stable cinnamaldehyde feedstocks, the process eliminates these variable costs entirely. Furthermore, the recyclability of the ionic liquid catalyst means that the catalyst cost is amortized over multiple batches, leading to substantial long-term savings. The simplified purification process also reduces energy consumption associated with distillation and drying, further enhancing the economic viability of large-scale production runs without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on hazardous raw materials that are subject to strict transportation and storage regulations. Thionyl chloride, for example, is a controlled substance in many jurisdictions due to its potential use in illicit drug manufacture and its inherent danger. By replacing such reagents with benign cinnamaldehyde derivatives and recoverable ionic liquids, the supply chain becomes more robust and less susceptible to regulatory disruptions. The stability of the raw materials ensures that inventory can be held for longer periods without degradation, allowing for better demand planning and stock management. This reliability is crucial for meeting the just-in-time delivery requirements of major pharmaceutical clients who depend on consistent availability of key intermediates for their own API synthesis schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, particularly regarding waste generation and emissions. This ionic liquid method is inherently greener, generating minimal three wastes (waste water, gas, and residue) compared to traditional methods. The absence of acidic off-gases and the ability to recycle the aqueous catalyst phase significantly reduce the environmental footprint of the manufacturing process. This aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. The ease of scaling is further supported by the homogeneous nature of the reaction mixture, which ensures consistent heat and mass transfer even in large reactors. This predictability reduces the risk of batch failures during scale-up, ensuring a steady supply of high-quality product to the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is vital for technical teams evaluating the feasibility of adopting this technology for their specific production needs. The responses cover aspects ranging from catalyst recovery to substrate scope, ensuring that all stakeholders have the necessary information to make informed decisions about integrating this green chemistry solution into their existing manufacturing portfolios.
Q: How does this ionic liquid method improve upon traditional acylation processes?
A: Traditional methods require hazardous acylating agents like thionyl chloride and extensive neutralization steps. This novel approach uses stable cinnamaldehyde and eliminates corrosive reagents, significantly reducing equipment corrosion and waste generation.
Q: What is the expected yield and purity profile for these derivatives?
A: According to patent data, the conversion rate and product yield can both exceed 90%. The process allows for high-purity isolation through simple extraction and crystallization without complex chromatographic purification.
Q: Is the ionic liquid catalyst recyclable for industrial scale-up?
A: Yes, the catalyst resides in the aqueous phase after extraction. It can be recovered by removing water and drying, enabling multiple reuse cycles which drastically lowers catalyst consumption costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylpropionate Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN109678718B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that consistency is key in the pharmaceutical supply chain, and our facilities are designed to maintain the highest standards of GMP compliance while leveraging green chemistry principles to minimize environmental impact.
We invite you to collaborate with us to explore how this efficient synthesis route can benefit your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this ionic liquid catalyzed method for your supply chain. Please contact our technical procurement team today to request specific COA data for our phenylpropionate derivatives and to discuss route feasibility assessments tailored to your volume needs. Together, we can drive efficiency and sustainability in the manufacturing of critical anabolic steroid intermediates and fine chemicals.
