Advanced Arylimidazole Quaternary Salts for Scalable Chiral Boronic Acid Manufacturing
Advanced Arylimidazole Quaternary Salts for Scalable Chiral Boronic Acid Manufacturing
The pharmaceutical industry's relentless pursuit of potent protease inhibitors has placed alpha-amino boronic acids at the forefront of medicinal chemistry, exemplified by the clinical success of Bortezomib. However, the efficient and stereoselective synthesis of these critical pharmacophores has historically been hindered by complex catalytic requirements and limited substrate scope. Patent CN103483267A introduces a groundbreaking solution through the development of diverse arylimidazole quaternary ammonium salts, represented by the general formula (L-n'), which serve as highly effective ligands for copper-catalyzed asymmetric borylation. This technology addresses the critical bottlenecks in producing chiral building blocks by eliminating the need for stringent inert atmosphere conditions while simultaneously expanding the range of compatible substrates to include challenging aromatic imines. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift towards more robust, cost-effective, and scalable manufacturing processes for next-generation therapeutic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the state-of-the-art for asymmetric synthesis of alpha-amino boronic acids relied heavily on methods developed by researchers such as Ellman, which utilized chiral tert-butyl sulfonamides and specific NHC-Cu complexes. While these methods offered high stereoselectivity for aliphatic substrates, they suffered from severe practical limitations that impeded industrial adoption. Specifically, the catalysts required were often air-sensitive N-heterocyclic carbene copper complexes that could only be prepared and stored within a glove box, necessitating expensive infrastructure and specialized operator training. Furthermore, the substrate scope was notoriously narrow; while aliphatic imines reacted well, aromatic imines typically resulted in poor yields ranging from 52% to 61%, or failed to produce the target product entirely. This lack of versatility forced process chemists to develop separate, inefficient routes for different classes of drug candidates, thereby inflating development timelines and material costs.

The Novel Approach
The methodology disclosed in the present patent fundamentally overcomes these barriers by introducing a new class of arylimidazole quaternary ammonium salts that function as robust, air-stable ligands. Unlike the fragile complexes of the past, these salts can be synthesized via a straightforward one-pot method without the need for a glove box, drastically reducing operational overhead. The chemical versatility of this approach is evident in its ability to accommodate a wide array of substituents on the imidazole ring, including various alkyl, aryl, and heteroaryl groups, allowing for fine-tuning of the steric and electronic environment around the catalytic center. Most critically for process scalability, this novel system delivers consistent high yields of 78% to 94% even for difficult aromatic imine substrates, ensuring that valuable raw materials are converted efficiently into high-value intermediates with an optical purity dr exceeding 98:2.
Mechanistic Insights into Copper-Catalyzed Asymmetric Borylation
The core utility of these arylimidazole quaternary ammonium salts lies in their ability to coordinate with univalent copper ions to form active catalytic species capable of enantioselective borylation. In the reaction mechanism, the copper-ligand complex activates the diboron reagent, facilitating the nucleophilic addition of the boron moiety to the imine carbon. The bulky aryl groups on the imidazole nitrogen atoms create a defined chiral pocket that directs the approach of the substrate, ensuring high stereocontrol. This precise spatial arrangement is crucial for generating the specific enantiomers required for biological activity in serine protease inhibitors. The stability of the quaternary ammonium structure ensures that the catalyst maintains its integrity throughout the reaction cycle, preventing premature decomposition that often plagues softer ligand systems.
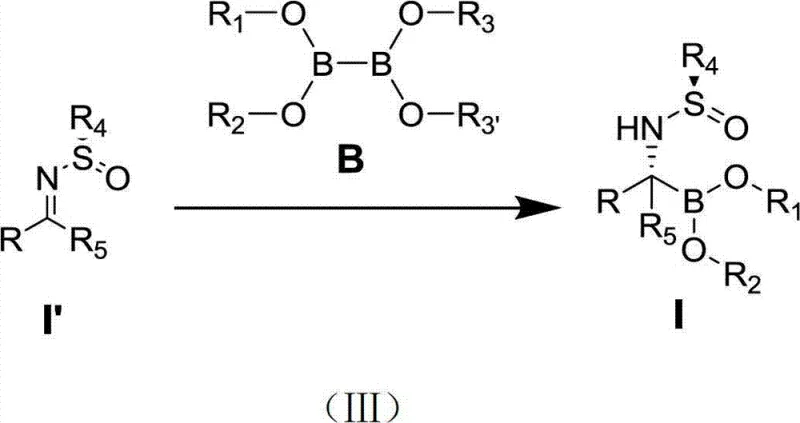
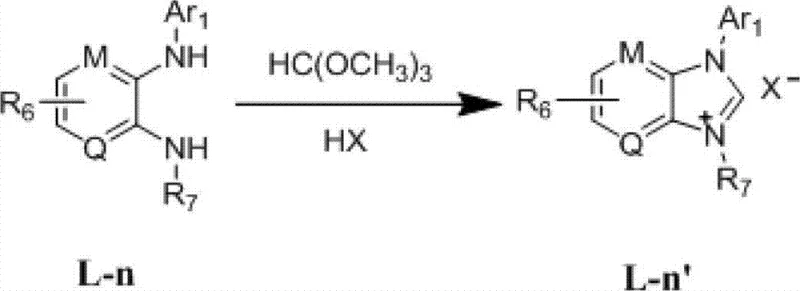
Beyond the primary catalytic cycle, the synthesis of the ligand itself offers deep insights into process robustness. The preparation involves a Buchwald-Hartwig amination followed by an acid-catalyzed cyclization, both of which are well-understood transformations in organic synthesis. The initial coupling of o-phenylenediamine with aryl bromides using palladium catalysis allows for the modular introduction of diverse aryl groups, which is key to tuning the catalyst's performance. Subsequent cyclization with trimethyl orthoformate under acidic conditions proceeds with high efficiency, yielding the final quaternary salt in 89% to 95% yield. This high-yielding precursor synthesis ensures that the ligand itself is not a bottleneck in the supply chain, providing a reliable source of high-purity catalyst for downstream manufacturing.
How to Synthesize Arylimidazole Quaternary Ammonium Salt Efficiently
The preparation of these high-performance ligands is designed for industrial feasibility, utilizing commercially available starting materials and standard reaction conditions. The process begins with the palladium-catalyzed coupling of substituted o-phenylenediamines with aryl halides, followed by a cyclization step that constructs the imidazolium core. Detailed standardized synthetic steps for specific embodiments, including precise stoichiometry and workup procedures, are provided in the guide below to ensure reproducibility across different manufacturing sites.
- Perform Buchwald-Hartwig amination of o-phenylenediamine with aryl bromides using Pd2(dba)3 and BINAP in toluene at 110°C overnight to form the diamine precursor.
- React the resulting diamine precursor with trimethyl orthoformate in the presence of hydrochloric and formic acid at 60-100°C for 1-6 hours.
- Isolate the final quaternary ammonium salt product via recrystallization, achieving yields of 89-95% without requiring inert atmosphere techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel catalyst system offers substantial strategic advantages rooted in operational simplicity and material efficiency. By eliminating the dependency on glove box technology and air-sensitive reagents, manufacturing facilities can utilize existing standard reactor trains without requiring capital-intensive upgrades to inert atmosphere handling systems. This reduction in infrastructure requirements directly translates to lower fixed costs and faster technology transfer times from lab to plant. Furthermore, the broad substrate scope means that a single catalyst platform can potentially service multiple drug programs, reducing the need to qualify and stockpile diverse sets of specialized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive and sensitive NHC-Cu complexes, which require specialized storage and handling, leads to significant savings in reagent costs. Additionally, the high yields observed across both aliphatic and aromatic substrates minimize raw material waste, ensuring that expensive boron reagents and imine precursors are utilized with maximum efficiency. The simplified workup procedures, often involving simple filtration and recrystallization rather than complex chromatographic separations, further reduce solvent consumption and processing time, driving down the overall cost of goods sold for these critical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as o-phenylenediamine, aryl bromides, and trimethyl orthoformate ensures a stable and resilient supply chain. Unlike proprietary chiral auxiliaries that may have single-source suppliers, the precursors for these arylimidazole salts are widely available from multiple global vendors. This diversification mitigates the risk of supply disruptions and provides procurement teams with greater leverage in negotiations. The robustness of the synthesis also means that production schedules are less likely to be impacted by environmental fluctuations or minor variations in raw material quality.
- Scalability and Environmental Compliance: The reaction conditions, typically operating at moderate temperatures between 60°C and 100°C, are easily scalable from kilogram to multi-ton batches using standard stainless steel reactors. The absence of pyrophoric reagents and the use of common organic solvents simplify waste management and regulatory compliance. High atom economy and reduced solvent usage align with green chemistry principles, helping manufacturers meet increasingly stringent environmental standards while maintaining high throughput capabilities for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this catalyst technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the operational parameters and expected outcomes for process development teams.
Q: Does this catalyst synthesis require a glove box?
A: No, a key advantage of the method disclosed in CN103483267A is that the preparation of the arylimidazole quaternary ammonium salts does not require a glove box, significantly simplifying operational complexity compared to prior art NHC-Cu complexes.
Q: What is the optical purity of the resulting alpha-amino boronic acids?
A: When used in copper-catalyzed asymmetric borylation, these catalysts facilitate the production of target products with high optical purity, specifically achieving a diastereomeric ratio (dr) of greater than 98:2.
Q: Can this method handle aromatic imine substrates effectively?
A: Yes, unlike previous methods which showed poor yields for arylamino boronic acids, this novel catalyst system expands the substrate scope to include diverse aromatic imines with yields ranging from 78% to 94%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylimidazole Quaternary Ammonium Salt Supplier
As the demand for chiral boronic acid intermediates continues to grow with the development of new protease inhibitors, securing a reliable supply of high-quality catalysts is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of arylimidazole quaternary ammonium salt performs consistently in your asymmetric borylation processes. We understand the critical nature of these materials in your synthesis of API intermediates and are committed to delivering the reliability and quality your projects demand.
We invite you to engage with our technical team to discuss how this innovative catalyst technology can be integrated into your specific manufacturing workflows. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this robust system. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your target molecules, and let us partner with you to accelerate your path to commercialization.
