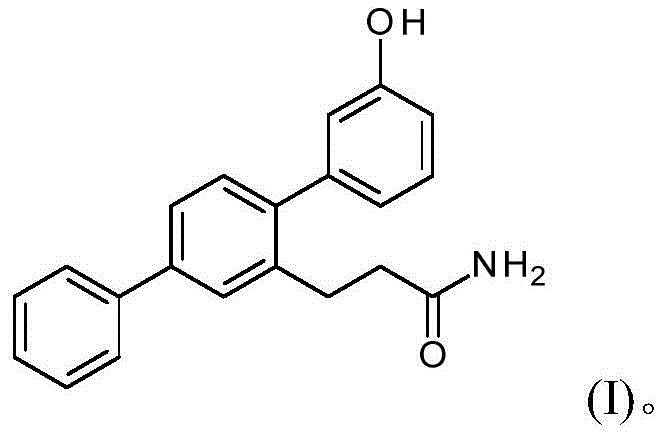
Advanced Synthesis of Terphenyl Compound VB0004 for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive molecules that balance high purity with manufacturability. Patent CN112457280A discloses a groundbreaking methodology for the synthesis of terphenyl compounds, specifically focusing on the potent hypotensive and anti-fibrotic agent known as VB0004. This compound, chemically defined as 2'-[3-hydroxy-(1,1':4',1''-terphenyl)]propionamide, represents a significant therapeutic candidate for treating fibrosis and hypertension. The disclosed invention provides a comprehensive, multi-step pathway that transforms readily available starting materials into high-value intermediates and finally the target API precursor. Unlike prior art methods which often struggled with purification bottlenecks and low overall yields, this novel approach leverages strategic functional group interconversions, including sequential Suzuki-Miyaura couplings and precise amidation techniques. For R&D directors and procurement specialists, understanding this pathway is critical as it offers a viable solution for cost reduction in API manufacturing while ensuring the supply of high-purity terphenyl compounds necessary for clinical and commercial success.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex terphenyl structures like VB0004 has been plagued by inefficiencies inherent in traditional cross-coupling strategies. Prior art, such as that disclosed in WO2015/039173, often relied on linear sequences that accumulated impurities at every stage, necessitating rigorous and yield-depleting purification protocols. A major bottleneck in conventional routes involves the difficulty of introducing the third aromatic ring onto a pre-functionalized biphenyl system without causing degradation of sensitive functional groups like phenols or carboxylic acids. Furthermore, traditional methods frequently employed stoichiometric amounts of expensive catalysts or harsh reaction conditions that led to significant by-product formation, such as ester contaminants during the final amidation step. These inefficiencies not only drove up the cost of goods but also extended the lead time for high-purity pharmaceutical intermediates, creating supply chain vulnerabilities for drug developers aiming for rapid clinical progression.
The Novel Approach
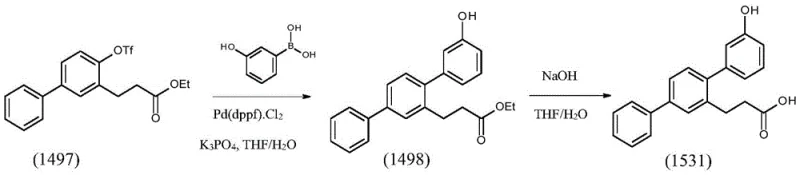
The methodology presented in CN112457280A revolutionizes this landscape by introducing a convergent strategy centered around a dihydrocoumarin scaffold. As illustrated in the overall reaction scheme below, the process initiates with the regioselective bromination of dihydrocoumarin, followed by a ring-opening Suzuki coupling to establish the first biaryl bond. This clever design allows for the simultaneous construction of the carbon skeleton and the installation of the propionic acid side chain. Subsequent steps involve protecting group manipulation via esterification and triflation, enabling a second, highly efficient Suzuki coupling to install the final hydroxyphenyl ring. This modular approach decouples the formation of the terphenyl core from the final amide functionality, allowing each step to be optimized independently for maximum yield and purity.
By utilizing this stepwise assembly, the novel approach effectively mitigates the formation of difficult-to-remove impurities and simplifies the isolation of intermediates. The ability to isolate stable intermediates like the triflate (1497) and the terphenyl ester (1498) provides crucial quality control checkpoints, ensuring that only material meeting strict specifications proceeds to the final stages. This level of process control is essential for the commercial scale-up of complex intermediates, offering manufacturers a reliable roadmap from kilogram to tonne-scale production.
Mechanistic Insights into Suzuki-Miyaura Coupling and Functional Group Manipulation
The heart of this synthetic strategy lies in the dual application of the Suzuki-Miyaura cross-coupling reaction, a palladium-catalyzed process renowned for its tolerance of various functional groups. The first coupling event transforms the 6-bromodihydrocoumarin (1490) into the open-ring acid (1495). Mechanistically, this involves the oxidative addition of the Pd(0) catalyst into the carbon-bromine bond, followed by transmetallation with phenylboronic acid in the presence of a base like KOH. The subsequent reductive elimination forms the new carbon-carbon bond while the basic conditions facilitate the hydrolysis of the lactone ring, elegantly delivering the carboxylic acid functionality in a single pot. This tandem transformation is a masterclass in atom economy, reducing the step count and minimizing waste generation compared to stepwise ring opening and coupling.
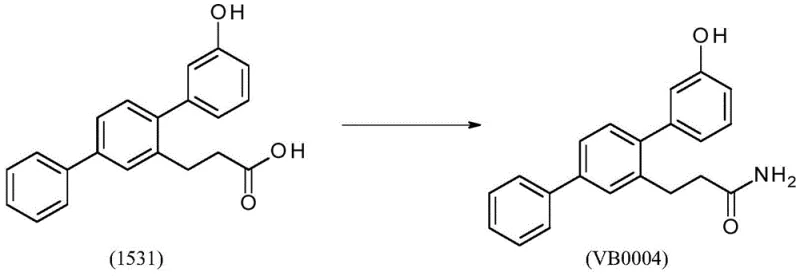
The second critical coupling involves the reaction of the triflate intermediate (1497) with 3-hydroxyphenylboronic acid. Triflates are superior leaving groups compared to halides in many contexts, facilitating rapid oxidative addition even with sterically hindered substrates. The patent details a crucial optimization where the base equivalents (K3PO4) were reduced from four to 1.5. This adjustment is mechanistically significant; excess base can lead to the formation of insoluble inorganic salts that trap product and create stubborn emulsions during work-up. By fine-tuning the base concentration, the process ensures a homogeneous reaction environment that promotes efficient catalytic turnover while simplifying downstream processing. Following the coupling, the ester is hydrolyzed under basic conditions to yield the free acid (1531), setting the stage for the final amidation.
The final conversion of acid 1531 to the amide VB0004 employs carbonyldiimidazole (CDI) as an activating agent. This mechanism proceeds via the formation of an acyl-imidazole intermediate, which is highly reactive towards nucleophilic attack by ammonia. The patent describes a unique protocol involving the bubbling of ammonia gas through the reaction mixture, followed by heating and a secondary methanol-ammonia treatment. This rigorous approach ensures complete conversion of the activated acid to the primary amide, overcoming the common issue of incomplete reaction or ester by-product formation that plagues simpler amidation methods. The result is a product with exceptional purity (>99.9%) and minimal residual palladium (1ppm), demonstrating the efficacy of the mechanistic design.
How to Synthesize VB0004 Efficiently
The synthesis of VB0004 described in patent CN112457280A offers a clear, reproducible pathway for laboratories and manufacturing facilities aiming to produce this valuable terphenyl compound. The process is characterized by its use of commercially available starting materials like dihydrocoumarin and phenylboronic acid, combined with standard organic transformations that are well-understood in process chemistry. Key to the success of this route is the careful control of reaction parameters, particularly temperature and stoichiometry during the coupling and activation steps. The following guide outlines the critical operational phases required to execute this synthesis effectively, ensuring high yields and purity at every stage.
- Brominate dihydrocoumarin to form 6-bromodihydrocoumarin (1490) using bromine in DCM.
- Perform Suzuki coupling with phenylboronic acid to open the lactone ring and form acid 1495.
- Convert acid 1495 to ethyl ester 1496 via Fischer esterification, then triflate to 1497.
- Execute second Suzuki coupling with 3-hydroxyphenylboronic acid to form terphenyl ester 1498.
- Hydrolyze ester 1498 to acid 1531, then activate with CDI and react with ammonia gas to yield VB0004.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational benefits and risk mitigation. The primary advantage lies in the robustness of the supply chain for raw materials; dihydrocoumarin and simple boronic acids are commodity chemicals available from multiple global suppliers, eliminating the dependency on bespoke, single-source precursors. This diversification of the supply base significantly enhances supply security and stabilizes pricing, protecting the project from market volatility. Furthermore, the high yields reported in the patent examples, such as the 74% yield for the initial bromination and coupling sequence, indicate a material-efficient process that maximizes output per unit of input, directly contributing to cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chromatographic purifications by relying on crystallization and precipitation techniques. For instance, the final product VB0004 is isolated by precipitation using toluene as an anti-solvent, a method that is vastly more economical and scalable than column chromatography. Additionally, the optimization of base usage in the Suzuki coupling reduces the consumption of reagents and the volume of waste generated, lowering disposal costs. The high purity achieved (>99%) means less material is lost during final polishing steps, improving the overall mass balance and reducing the effective cost per kilogram of the active ingredient.
- Enhanced Supply Chain Reliability: The stability of key intermediates, such as the triflate (1497), which can be stored at -20°C for months without degradation, provides flexibility in production scheduling. This allows manufacturers to produce and stockpile critical intermediates during periods of low demand or raw material availability, smoothing out production peaks and valleys. The simplified work-up procedures, particularly the reduction of emulsion formation, lead to faster batch cycle times. This efficiency enables facilities to increase throughput without additional capital investment, ensuring that delivery timelines for high-purity pharmaceutical intermediates are consistently met.
- Scalability and Environmental Compliance: The synthetic route avoids the use of highly toxic reagents or extreme conditions, aligning with modern green chemistry principles. The use of standard solvents like ethanol, ethyl acetate, and dichloromethane facilitates solvent recovery and recycling, minimizing the environmental footprint. The ability to scale from gram to kilogram quantities, as demonstrated in the patent examples (e.g., 150g scale for triflation), proves the feasibility of this route for commercial scale-up of complex intermediates. The low residual palladium content (1ppm) achieved without specialized scavengers simplifies regulatory compliance and reduces the burden on quality control laboratories.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of VB0004. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the purity profile of VB0004 synthesized via this novel route?
A: The patented process achieves exceptional purity levels, with UPLC analysis indicating product purity of 99.91%. Furthermore, residual palladium content is tightly controlled to approximately 1ppm, meeting stringent pharmaceutical standards without requiring complex metal scavenging steps.
Q: How does the new Suzuki coupling protocol improve work-up efficiency?
A: The process optimizes the base usage in the Suzuki coupling step by reducing potassium phosphate equivalents from four to 1.5. This significant reduction minimizes the formation of inorganic solids and emulsions during aqueous work-up, thereby streamlining the separation of organic and aqueous phases and reducing processing time.
Q: Is the synthesis of intermediate 1495 scalable for industrial production?
A: Yes, the synthesis of intermediate 1495 from 6-bromodihydrocoumarin (1490) is highly scalable. The reaction utilizes standard Suzuki conditions with palladium acetate and achieves a robust yield of 74% with GC purity greater than 98%, making it suitable for large-scale manufacturing of the terphenyl core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable VB0004 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of complex pharmaceutical intermediates like VB0004. Our team of expert process chemists has extensively analyzed the route disclosed in CN112457280A and possesses the technical capability to implement and optimize this synthesis for your specific needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities and residual metals down to ppm levels.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, helping you maximize the economic potential of this efficient route. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us help you secure a sustainable and cost-effective supply of this vital terphenyl compound.
