Scalable Synthesis of Potent Aryl Dihydronaphthalene Lignans for Oncology Applications
Scalable Synthesis of Potent Aryl Dihydronaphthalene Lignans for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds with high selectivity and potency for oncology treatments, particularly for resistant forms of cervical cancer. Patent CN114262270B introduces a significant advancement in this domain by disclosing a class of aryl dihydronaphthalene lignans isolated from Drynaria fortunei and, crucially, providing a viable synthetic pathway for their production. These compounds, specifically designated as Compound 1 and Compound 2, demonstrate exceptional in vitro cytotoxicity against human cervical cancer HeLa cell lines. The transition from relying solely on natural extraction, which is often plagued by low yields and supply inconsistency, to a defined chemical synthesis represents a pivotal shift for supply chain stability. This report analyzes the technical feasibility and commercial implications of this patented technology for global procurement and R&D teams.
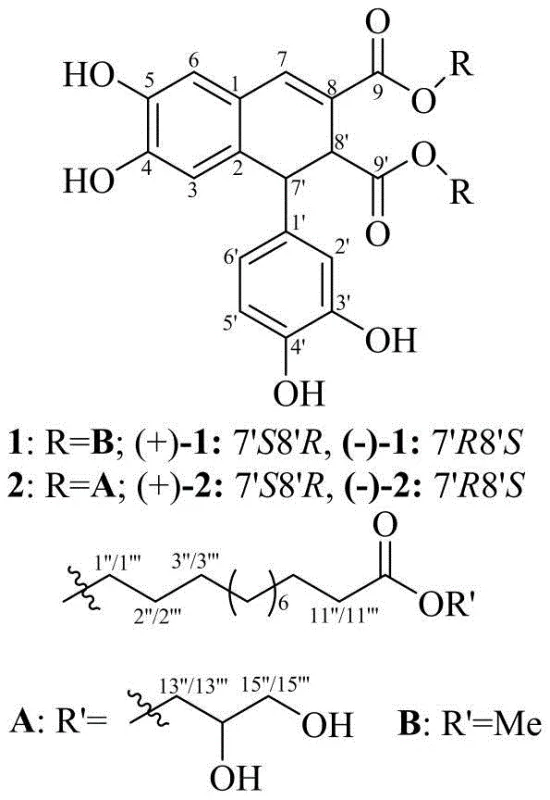
The core innovation lies in the dual approach of isolation and biomimetic synthesis. While the natural products were initially identified through extensive bio-guided fractionation of the plant's dried rhizomes, the patent elucidates a total synthesis strategy that bypasses the agricultural bottlenecks of plant sourcing. The general structure features a complex tetrahydronaphthalene core fused with caffeic acid derivatives and long-chain fatty acid esters. Understanding the precise stereochemistry is vital, as the patent highlights that the (+) enantiomers generally exhibit superior cytotoxic activity compared to their (-) counterparts. For a reliable pharmaceutical intermediate supplier, mastering the stereoselective aspects or the efficient resolution of these enantiomers is key to delivering high-value active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining high-purity lignans from Drynaria fortunei involves laborious extraction processes that are inherently inefficient for large-scale manufacturing. The conventional method requires harvesting vast quantities of dried rhizomes, followed by repeated methanol extraction and concentration. The resulting crude extract must then undergo multiple stages of silica gel column chromatography using complex gradient elution systems involving petroleum ether, ethyl acetate, and dichloromethane. This process not only consumes significant volumes of organic solvents but also results in poor overall recovery rates due to the structural similarity of co-extracted impurities. Furthermore, natural variation in plant material leads to batch-to-batch inconsistency in the content of the active lignans, posing a severe risk for cost reduction in API manufacturing where reproducibility is paramount. The reliance on seasonal agricultural inputs also introduces volatility into the supply chain, making it difficult to guarantee continuous availability for clinical or commercial needs.
The Novel Approach
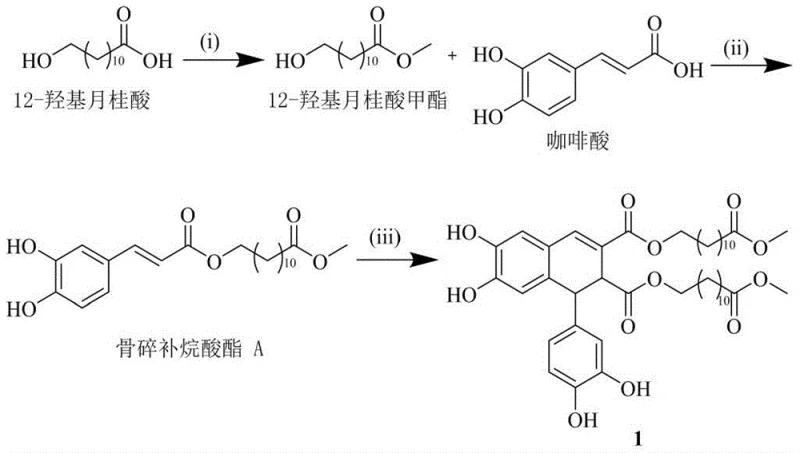
The patented synthetic route offers a transformative solution by constructing the complex lignan skeleton from simple, commercially available building blocks. Instead of extracting the molecule from nature, the process builds it up through a sequence of methylation, esterification, and oxidative coupling reactions. This approach decouples production from agricultural cycles and allows for precise control over reaction parameters such as temperature, stoichiometry, and reaction time. By utilizing 12-hydroxylauric acid and caffeic acid as starting materials, the synthesis ensures a consistent chemical identity regardless of external environmental factors. Moreover, the synthetic pathway is designed to be modular, meaning that analogues can potentially be generated by varying the fatty acid chain or the phenolic component. This flexibility supports the rapid development of structure-activity relationship (SAR) studies, accelerating the drug discovery timeline for potential new chemical entities targeting cervical cancer.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The cornerstone of this synthesis is the biomimetic oxidative coupling reaction, which constructs the central dihydronaphthalene core. This step mimics the biosynthetic pathway found in plants, where phenolic precursors are dimerized enzymatically. In the laboratory setting, this is achieved using ferric chloride hexahydrate (FeCl3·6H2O) as a single-electron oxidant in an acetone solvent system. The mechanism likely involves the generation of phenoxy radicals from the caffeic acid moieties of the precursor molecules. These radicals then undergo regioselective coupling to form the new carbon-carbon bonds that define the lignan skeleton. The use of iron salts is particularly advantageous from a green chemistry perspective, as iron is abundant, non-toxic, and inexpensive compared to precious metal catalysts like palladium or platinum often used in cross-coupling reactions. This choice of catalyst significantly lowers the barrier for commercial scale-up of complex pharmaceutical intermediates by simplifying downstream purification and reducing heavy metal residue concerns.
Following the coupling reaction, the control of stereochemistry becomes the primary challenge. The oxidative coupling typically yields a mixture of stereoisomers due to the formation of new chiral centers at the coupling sites. The patent details a rigorous purification protocol involving Sephadex LH-20 gel chromatography to remove polymeric byproducts and unreacted starting materials. Subsequently, the enantiomers are resolved using semi-preparative high-performance liquid chromatography (HPLC) equipped with a Cellulose-5 chiral column. This step is critical for R&D teams aiming to evaluate the specific biological activity of each enantiomer, as the data indicates a marked difference in potency between the (+) and (-) forms. The ability to isolate these pure enantiomers ensures that preclinical toxicology studies are conducted on well-defined substances, reducing the risk of failure in later development stages due to off-target effects from the less active isomer.
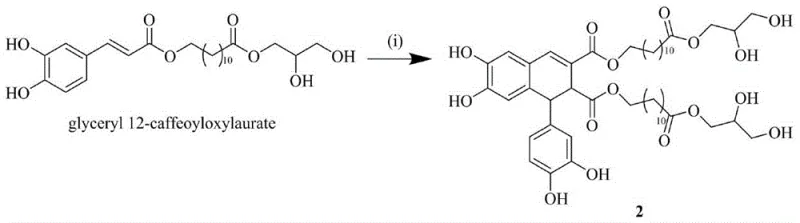
How to Synthesize Aryl Dihydronaphthalene Lignans Efficiently
The synthesis protocol described in the patent provides a clear roadmap for reproducing these high-value compounds in a laboratory or pilot plant setting. The process begins with the activation of the fatty acid chain, followed by conjugation with the phenolic acid, and concludes with the dimerization step. Each stage has been optimized to balance yield and purity, ensuring that the final product meets the stringent requirements for biological testing. The use of standard organic synthesis techniques means that this route can be readily adopted by contract development and manufacturing organizations (CDMOs) without the need for specialized exotic equipment. For detailed operational parameters, safety data, and exact stoichiometric ratios required for GMP production, please refer to the standardized synthesis guide below.
- Perform methylation of 12-hydroxylauric acid followed by Mitsunobu esterification with caffeic acid to generate the precursor Drynaria alkanoate A.
- Execute oxidative coupling of the precursor using ferric chloride hexahydrate (FeCl3·6H2O) in acetone solution at low temperature to form the dimeric core.
- Purify the crude coupling product via Sephadex LH-20 chromatography and separate enantiomers using semi-preparative chiral HPLC with a Cellulose-5 column.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers substantial strategic advantages for procurement managers looking to optimize the supply chain for oncology intermediates. By shifting from extraction to synthesis, companies can mitigate the risks associated with raw material scarcity and price fluctuation inherent in botanical sourcing. The starting materials, such as caffeic acid and hydroxylauric acid, are commodity chemicals with stable global supply networks, ensuring that production can be scaled up rapidly to meet demand without long lead times. This transition effectively transforms the supply model from an agriculture-dependent variable to a manufacturing-driven constant, providing greater predictability for long-term project planning and inventory management.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for processing tons of plant biomass, which drastically reduces logistics costs, waste disposal fees, and solvent consumption associated with large-scale extraction. Furthermore, the use of iron-based catalysts instead of expensive transition metals lowers the direct material cost of the reaction. The streamlined purification process, while requiring chiral HPLC, is offset by the higher overall throughput and consistency of the synthetic input, leading to a more favorable cost-of-goods-sold (COGS) profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Synthetic manufacturing is not subject to the seasonal variations, weather conditions, or geopolitical issues that can disrupt the harvest of medicinal plants. This reliability is crucial for maintaining continuous clinical trial supplies and ensuring that commercial launches are not delayed by raw material shortages. Establishing a synthetic supply chain also allows for multi-vendor sourcing strategies, as any capable fine chemical manufacturer can replicate the process, thereby reducing dependency on a single geographic region or supplier.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of acetone and mild temperatures, are amenable to large-scale reactor operations. The absence of persistent organic pollutants or heavy metal catalysts simplifies the environmental impact assessment and waste treatment protocols. This alignment with green chemistry principles not only reduces regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing partner, which is increasingly important for pharmaceutical buyers evaluating their Scope 3 emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these aryl dihydronaphthalene lignans. The answers are derived directly from the experimental data and specifications outlined in the patent documentation, ensuring accuracy for decision-makers evaluating this technology for integration into their pipelines.
Q: What is the biological potency of the synthesized aryl dihydronaphthalene lignans?
A: The compounds exhibit strong selective inhibitory activity against human cervical cancer HeLa cell lines, with IC50 values ranging from 0.02 μM to 0.16 μM, significantly outperforming their activity against other cancer cell lines like HCT116 or MCF-7.
Q: How is chirality controlled in the synthesis of these lignans?
A: While the oxidative coupling produces a racemic mixture, the patent describes a robust resolution method using semi-preparative HPLC with a Cellulose-5 chiral column, allowing for the isolation of distinct (+) and (-) enantiomers with high optical purity.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis relies on readily available starting materials such as 12-hydroxylauric acid and caffeic acid, which facilitates cost-effective commercial scale-up compared to total extraction from Drynaria fortunei rhizomes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Dihydronaphthalene Lignans Supplier
The technical complexity of synthesizing chiral lignans requires a partner with deep expertise in process chemistry and scale-up engineering. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of handling the sophisticated analytical requirements for chiral separation and purity verification, guaranteeing that every batch meets stringent purity specifications required for oncology drug development.
We invite you to discuss how we can optimize this synthetic route for your specific volume requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis to demonstrate the economic benefits of switching to this synthetic supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
