Advanced Industrial Synthesis of Galanthamine Derivatives for Pharmaceutical Manufacturing
Introduction to Patented Galanthamine Synthesis Technology
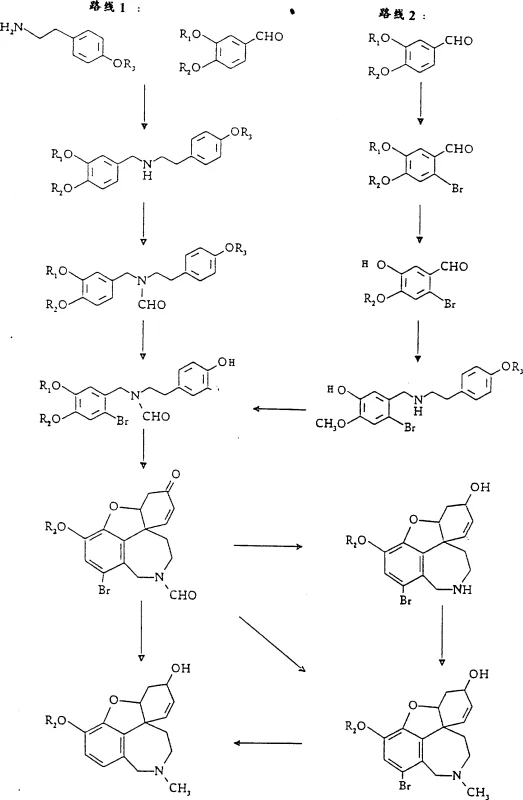
The global demand for effective treatments for Alzheimer's disease has placed significant pressure on the supply chain for key active pharmaceutical ingredients (APIs), particularly Galanthamine. Patent CN1170395A introduces a groundbreaking preparation method for 4a,5,9,10,11,12-hexahydro-6H-benzofuro[3a,3,2-ef][2]benzazepine derivatives, specifically targeting the efficient industrial production of Galanthamine. This technology addresses critical bottlenecks in previous synthetic routes by utilizing a novel bromine-mediated pathway that facilitates high-yield production via intermediates such as bromine-N-demethyl galanthamine. The core innovation lies in the strategic use of oxidative cyclization followed by a highly diastereoselective reduction using L-Selectride, which ensures superior stereochemical control without the need for complex chiral resolution steps at every stage. For procurement managers and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is essential for securing a stable supply of high-purity neurological agents.
![General formula of 4a,5,9,10,11,12-hexahydro-6H-benzofuro[3a,3,2-ef][2]benzazepine derivatives showing variable substituents](/insights/img/galanthamine-intermediate-synthesis-pharma-supplier-20260309042802-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Galanthamine has been plagued by inefficiencies that render large-scale production economically unviable. Traditional methods, such as those described by Kametani or Holton, often rely on oxidative cyclization of phenethylamine derivatives which typically suffer from extremely low yields, frequently reported below 1% of the theoretical yield. Furthermore, earlier approaches frequently necessitated the use of stoichiometric amounts of expensive and toxic heavy metal catalysts, such as palladium trifluoroacetate and thallium salts, to drive the phenolic coupling reaction. These legacy processes not only inflate the cost of goods sold (COGS) due to reagent expenses but also create significant environmental liabilities regarding heavy metal waste disposal. Additionally, the lack of stereocontrol in the reduction steps often resulted in complex mixtures of diastereomers, requiring tedious and costly chromatographic purification that is impractical for multi-kilogram manufacturing campaigns.
The Novel Approach
The methodology outlined in CN1170395A represents a paradigm shift in cost reduction in API manufacturing by introducing a robust bromine-templated strategy. Instead of relying on low-yield direct coupling, this route employs a bromine atom to direct the oxidative cyclization and subsequently serves as a handle for stereochemical control. The process achieves individual step yields of 85% to 95%, a dramatic improvement over the single-digit yields of prior art. Crucially, the method enables the isolation of intermediates via crystallization rather than chromatography, significantly simplifying the downstream processing. By replacing hazardous halogenated solvents like chloroform with industrially friendly toluene, the process enhances operator safety and aligns with modern green chemistry principles, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into L-Selectride Catalyzed Stereoselective Reduction
The heart of this synthetic breakthrough is the diastereoselective reduction of the bromo-narwedine ketone intermediate. In conventional reductions using agents like lithium aluminum hydride, the attack on the carbonyl group at the C-6 position is poorly controlled, leading to a mixture of the desired Galanthamine alcohol and the unwanted epigalanthamine epimer. The patent specifies the use of L-Selectride (lithium tri-sec-butylborohydride), a bulky hydride donor that sterically directs the hydride attack to the less hindered face of the molecule. This steric bias ensures that the hydroxyl group is installed in the beta-configuration with high fidelity. Experimental data within the patent indicates that this reduction proceeds with yields ranging from 70% to 85% theoretically, with chromatographic analysis showing no detectable quantities of the epibromine-N-demethyl galanthamine impurity. This level of purity is critical for R&D teams focused on impurity profiling and regulatory compliance, as it minimizes the burden on subsequent purification stages.
Furthermore, the oxidative cyclization step utilizes potassium ferricyanide in a biphasic toluene-water system, mediated by a base such as potassium carbonate. This mechanistic pathway avoids the radical scrambling often seen with other oxidants. The presence of the bromine substituent at the ortho-position relative to the phenolic oxygen stabilizes the transition state during the phenolic coupling, ensuring the formation of the correct benzazepine ring system. The subsequent removal of the bromine atom via catalytic hydrogenation or zinc-mediated reduction is a clean transformation that restores the aromatic system without affecting the sensitive ether bridge or the newly formed stereocenter. This sequence demonstrates a sophisticated understanding of functional group tolerance, allowing for the synthesis of high-purity OLED material precursors or pharmaceutical intermediates with minimal side reactions.
How to Synthesize Galanthamine Efficiently
The synthesis of Galanthamine via this patented route involves a logical sequence of condensation, protection, cyclization, and reduction steps designed for maximum efficiency. The process begins with the condensation of isovanillin derivatives with tyramine, followed by N-formylation to protect the amine during the critical oxidative step. After bromination and oxidative cyclization to form the tricyclic ketone, the key L-Selectride reduction establishes the required stereochemistry. The detailed standardized synthesis steps for implementing this route in a GMP environment are provided in the guide below.
- Condensation of isovanillin derivatives with tyramine followed by reduction to form the benzylamine precursor.
- N-formylation and selective bromination of the aromatic ring to prepare for cyclization.
- Oxidative phenolic coupling using potassium ferricyanide to form the benzazepine core structure.
- Diastereoselective reduction of the ketone using L-Selectride to establish the correct stereochemistry at the 6-position.
- N-methylation and catalytic debromination to yield the final Galanthamine active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost savings derived from process intensification. By eliminating the need for expensive stoichiometric heavy metal catalysts like palladium and thallium, the raw material costs are drastically simplified. Moreover, the ability to isolate products through crystallization rather than silica gel chromatography reduces solvent consumption and waste generation, directly impacting the environmental compliance costs associated with chemical manufacturing. This efficiency translates into a more reliable supply chain, as the process is less susceptible to the bottlenecks associated with complex purification workflows.
- Cost Reduction in Manufacturing: The replacement of toxic halogenated solvents with toluene not only improves safety but also reduces the cost of solvent recovery and disposal. The high yields achieved in each step, particularly the 85% yield in the stereoselective reduction, mean that less starting material is required to produce the same amount of API. This efficiency eliminates the need for expensive recycling loops and minimizes the loss of valuable chiral intermediates, leading to significant overall cost optimization.
- Enhanced Supply Chain Reliability: The robustness of this method ensures consistent batch-to-batch quality, which is vital for maintaining long-term contracts with pharmaceutical partners. The use of readily available reagents like potassium ferricyanide and L-Selectride, combined with standard unit operations like filtration and crystallization, reduces the risk of supply disruptions caused by specialized reagent shortages. This stability allows for better inventory planning and reduces lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, avoiding the pitfalls of laboratory-only techniques. The avoidance of chromatographic purification makes the process inherently scalable to multi-ton production levels without a proportional increase in operational complexity. Additionally, the reduced reliance on hazardous chemicals aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory changes and enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners.
Q: How does the L-Selectride reduction improve stereochemical purity compared to traditional methods?
A: The patented process utilizes L-Selectride (lithium tri-sec-butylborohydride) to achieve diastereoselective reduction of the bromo-narwedine ketone. This specific reagent favors the formation of the desired 6-beta-hydroxy configuration with yields reaching 85%, significantly minimizing the formation of the epigalanthamine impurity which is common with non-selective reducing agents like lithium aluminum hydride.
Q: What are the environmental advantages of this synthesis route?
A: This method replaces toxic halogenated solvents such as chloroform with toluene, which is safer for industrial handling and reduces ecological impact. Furthermore, the process optimizes isolation through crystallization rather than extensive column chromatography, drastically reducing solvent waste and processing time.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly describes the method as suitable for industrial scale-up. By avoiding expensive stoichiometric heavy metal catalysts like palladium and thallium used in earlier routes, and by enabling product isolation via crystallization, the process offers the reproducibility and cost-efficiency required for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Galanthamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for neurological therapeutics. Our team of expert chemists has extensively evaluated the technology described in CN1170395A and possesses the technical capability to implement this advanced bromine-mediated pathway. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot plant to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the precise control of stereoisomers required for Galanthamine production.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your time to market.
