Industrial Purification of 1-(2-Trifluoromethylphenyl)ethanol for High-Value Pharmaceutical Intermediates
Industrial Purification of 1-(2-Trifluoromethylphenyl)ethanol for High-Value Pharmaceutical Intermediates
In the highly regulated landscape of modern pharmaceutical manufacturing, the demand for chiral building blocks with exceptional optical purity is paramount. A significant technological breakthrough addressing this challenge is detailed in patent CN101910101A, which discloses a robust industrial purification method for optically active 1-(2-trifluoromethylphenyl)ethanol. This specific intermediate is critical for the synthesis of various bioactive molecules, yet achieving the requisite >99% enantiomeric excess (ee) has historically been fraught with inefficiencies. The patent reveals that while asymmetric reduction can produce the alcohol, it often falls short of the strict purity standards required for API intermediates without cumbersome downstream processing. The disclosed innovation lies in a surprisingly simple yet highly effective recrystallization technique using aliphatic hydrocarbon solvents, which drastically improves optical purity without the need for complex derivatization or expensive resolving agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of chiral 1-phenylethanols, particularly those with trifluoromethyl substitutions, has relied on labor-intensive strategies that hinder scalability and cost-efficiency. Conventional wisdom suggested that simple recrystallization of the free alcohol was ineffective because the racemic crystal lattice is often more stable than the pure enantiomer crystal, leading to the precipitation of racemate rather than the desired pure isomer. For analogous compounds like 1-(3,5-bistrifluoromethylphenyl)ethanol, the industry standard involved forming complexes with expensive additives such as DABCO (1,4-diazabicyclo[2.2.2]octane) to facilitate resolution. This approach introduces significant operational burdens, including the cost of the resolving agent itself and the additional unit operations required to recover the target molecule from the complex. Furthermore, alternative methods involving kinetic resolution or derivatization increase the total number of synthetic steps, inevitably lowering the overall yield and generating substantial chemical waste, which contradicts modern green chemistry principles.
The Novel Approach
The methodology presented in patent CN101910101A represents a paradigm shift by demonstrating that the ortho-trifluoromethyl substituted ethanol behaves uniquely compared to its meta- or para-substituted counterparts. The inventors discovered that by utilizing specific aliphatic hydrocarbon solvents, particularly n-heptane, the thermodynamic equilibrium during crystallization can be manipulated to favor the precipitation of the optically pure crystal directly. This eliminates the need for any chiral additives or derivatization steps, streamlining the process into a single, efficient unit operation. The process operates under mild conditions, with aging temperatures ranging from -20°C to +10°C, which significantly reduces the energy load on industrial cooling systems compared to cryogenic methods. By optimizing the solvent-to-substrate ratio between 2 mL/g and 10 mL/g, manufacturers can achieve both high optical purity upgrades and excellent recovery rates, solving the long-standing trade-off between purity and yield in chiral purification.
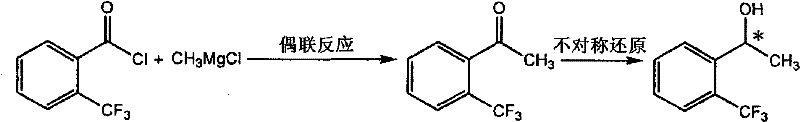
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Reduction and Crystallization
The production of the precursor alcohol typically involves a two-step sequence: a Grignard coupling followed by an asymmetric hydrogenation. The coupling of 2-(trifluoromethyl)benzoyl chloride with methylmagnesium chloride yields the corresponding acetophenone, which serves as the substrate for the critical stereoselective step. The asymmetric reduction is catalyzed by a sophisticated ruthenium complex featuring chiral ligands such as BINAP and chiral diamines (e.g., DAIPEN). This catalyst system facilitates the transfer of hydrogen to the prochiral ketone with high facial selectivity, establishing the initial chiral center. While this catalytic step is highly efficient, yielding conversion rates near 100%, the resulting optical purity often hovers around 95-97% ee, necessitating the subsequent purification step to meet pharmaceutical specifications.
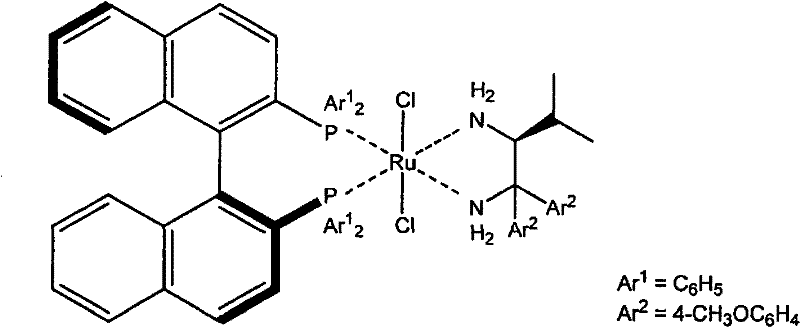
The subsequent recrystallization mechanism relies on the subtle interplay between solute-solvent interactions and crystal lattice energy. In aliphatic hydrocarbon solvents like n-heptane, the solubility of the minor enantiomer impurity is sufficiently high to keep it in the mother liquor, while the major enantiomer reaches supersaturation and nucleates. The presence of the bulky ortho-trifluoromethyl group likely introduces steric strain that destabilizes the formation of racemic co-crystals in this specific solvent environment, thereby promoting the growth of homochiral crystals. This phenomenon allows for the exclusion of impurities not just chemically, but stereochemically, resulting in a final product with >99.9% ee and high chemical purity. The process effectively acts as a final polishing step that corrects the minor stereochemical imperfections of the catalytic reduction.
How to Synthesize 1-(2-Trifluoromethylphenyl)ethanol Efficiently
To implement this purification strategy effectively, precise control over solvent quality and thermal profiles is essential. The process begins with the dissolution of the crude alcohol, which typically possesses an optical purity between 90% and 98% ee, into the designated aliphatic hydrocarbon solvent. It is crucial to heat the mixture to approximately 30-40°C to ensure a homogeneous solution before initiating the cooling ramp. The detailed standardized synthesis and purification steps, including specific agitation rates and filtration protocols, are outlined below to ensure reproducibility at scale.
- Dissolve the crude optically active alcohol (90-98% ee) in an aliphatic hydrocarbon solvent such as n-heptane at a ratio of 2-10 mL per gram of substrate.
- Heat the mixture to 30-40°C to ensure complete dissolution, then cool gradually to an aging temperature between -20°C and +10°C.
- Maintain the aging temperature with stirring for 1-5 hours to allow preferential crystallization of the pure enantiomer, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology offers tangible strategic benefits that extend beyond mere technical performance. By transitioning from additive-dependent resolution methods to a solvent-based recrystallization process, organizations can significantly simplify their supply chains and reduce exposure to volatile raw material costs. The elimination of expensive resolving agents like DABCO removes a costly line item from the bill of materials and eradicates the associated logistics of handling and recovering these specialty chemicals. Furthermore, the use of commodity solvents like n-heptane ensures consistent availability and pricing stability, mitigating the risk of supply disruptions that often plague specialized reagent markets.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the downstream processing workflow. By removing the requirement for stoichiometric chiral additives and the subsequent recovery steps, the manufacturing process consumes fewer resources and generates less waste. The ability to recycle the aliphatic hydrocarbon solvent through simple distillation further enhances the economic viability, allowing for a circular solvent usage model that minimizes disposal costs. Additionally, the high recovery rates achieved mean that less starting material is lost to mother liquors, directly improving the overall mass balance and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available aliphatic hydrocarbons rather than niche resolving agents strengthens the resilience of the supply chain. n-Heptane is a standard industrial solvent with a robust global supply network, ensuring that production schedules are not held hostage by the lead times of specialized fine chemicals. The simplified process flow also reduces the number of critical control points in manufacturing, decreasing the likelihood of batch failures due to reagent quality issues. This reliability translates into more predictable delivery timelines for downstream pharmaceutical customers, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with green chemistry initiatives. The reduction in chemical waste, specifically the absence of nitrogen-containing resolving agent byproducts, simplifies effluent treatment and lowers the environmental footprint of the facility. The mild operating temperatures (-20°C to +10°C) are easily achievable with standard industrial chillers, avoiding the need for energy-intensive cryogenic equipment. This ease of scale-up ensures that the process can be transferred from pilot plant to multi-ton commercial production with minimal engineering hurdles, supporting rapid market entry for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear understanding of the process boundaries and capabilities.
Q: Why is n-heptane preferred over aromatic solvents for this purification?
A: Patent data indicates that aromatic solvents like toluene fail to precipitate crystals efficiently or result in low optical purity enhancement. Aliphatic hydrocarbons like n-heptane uniquely favor the crystallization of the pure enantiomer over the racemate for this specific ortho-substituted compound.
Q: What is the achievable optical purity using this recrystallization method?
A: Starting from material with 95-97% ee, this method can consistently upgrade the optical purity to >99.5% ee (often reported as 100% ee in examples) while maintaining high chemical purity and recovery rates.
Q: Does this process require expensive chiral additives?
A: No. Unlike previous methods for similar compounds that required stoichiometric amounts of expensive resolving agents like DABCO, this method relies solely on solvent selection, eliminating the need for additive recovery steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-Trifluoromethylphenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of chiral pharmaceuticals depends on the availability of high-purity intermediates produced via robust and scalable routes. Our technical team has extensively analyzed the pathway described in patent CN101910101A and possesses the expertise to execute this asymmetric reduction and recrystallization sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99% ee for every batch released.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal standards before committing to production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
