Scalable Synthesis of Anagrelide Trichloro Derivative for Global Pharmaceutical Quality Control
Scalable Synthesis of Anagrelide Trichloro Derivative for Global Pharmaceutical Quality Control
The pharmaceutical industry relies heavily on the availability of high-purity reference standards to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). A critical development in this domain is disclosed in patent CN107903269B, which details a robust preparation method for the Anagrelide trichloro derivative. This specific compound, chemically known as 6,7,8-trichloro-1,5-dihydroimidazo[2,1-b]quinazolin-2(3H)-one, serves as a pivotal impurity marker for Anagrelide hydrochloride, a drug approved by the FDA for treating essential thrombocythemia. The structural integrity of this molecule is paramount for regulatory compliance, as it is explicitly listed in major pharmacopoeias. 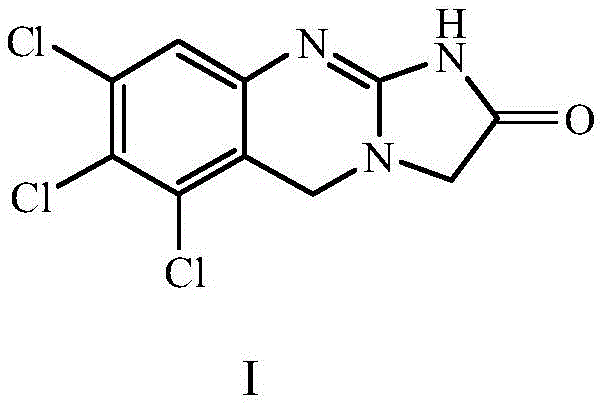
Prior to this innovation, the synthetic landscape for this specific trichloro derivative was notably sparse, with no comprehensive reports in domestic or international patents or periodicals. This lack of standardized methodology posed significant challenges for quality control laboratories and API manufacturers who required authentic samples for method validation and stability testing. The disclosed technology bridges this gap by offering a systematic, five-step synthetic route that transforms simple starting materials into the complex target molecule with high fidelity. For R&D directors and procurement specialists, understanding this pathway is essential for securing a stable supply of critical quality control materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic impurities like the Anagrelide trichloro derivative has been fraught with difficulties due to the absence of a defined, scalable protocol. In the absence of a dedicated patent, laboratories often resorted to ad-hoc modifications of the parent API synthesis, which frequently resulted in low yields and difficult purification processes. The introduction of three chlorine atoms at specific positions on the quinazoline ring creates significant steric and electronic challenges that conventional cyclization methods struggle to address efficiently. Furthermore, without a optimized route, the risk of generating unknown by-products increases, complicating the chromatographic profiling required for regulatory submissions. These inefficiencies not only drive up the cost of goods but also extend the lead time for obtaining certified reference materials, creating bottlenecks in the drug approval and release pipeline.
The Novel Approach
The methodology presented in patent CN107903269B introduces a streamlined, linear synthesis that effectively overcomes these historical barriers. The process initiates with a Sandmeyer reaction on 3,4,5-trichloroaniline, a readily available commodity chemical, to construct the trichloro-substituted isatin core. This is followed by a strategic oxidation-reduction sequence that manipulates the oxidation state of the side chain, converting a carboxylic acid intermediate into a reactive benzyl alcohol. 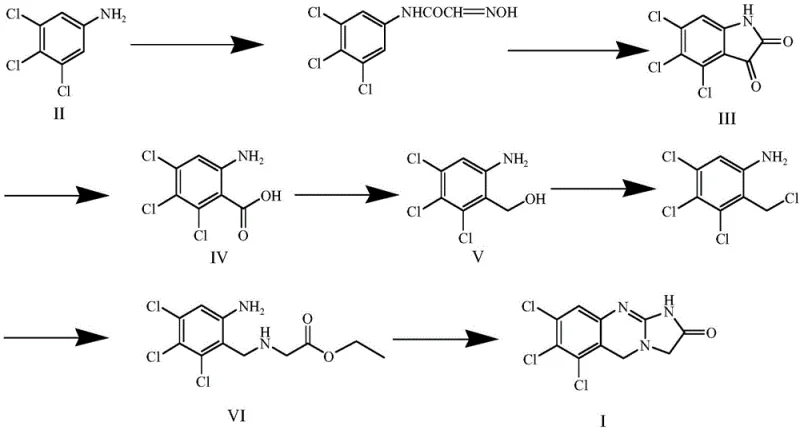
The brilliance of this approach lies in its final stages, where a condensation with glycine ethyl ester and a subsequent cyclization with cyanogen bromide cleanly close the imidazole ring. This stepwise construction allows for the isolation and purification of intermediates, ensuring that impurities do not carry through to the final product. For supply chain managers, this modularity means that if one step encounters a delay, the intermediates can potentially be sourced or stockpiled independently, enhancing overall supply resilience. The route avoids exotic catalysts or extreme conditions, relying instead on well-understood organic transformations that are easily transferable from the laboratory to pilot plant scales.
Mechanistic Insights into the Multi-Step Synthetic Pathway
A deep dive into the reaction mechanism reveals the careful optimization of conditions required to handle the highly chlorinated aromatic system. The initial Sandmeyer-type reaction involves the formation of an isonitrosoacetanilide intermediate which cyclizes under strong acidic conditions (concentrated sulfuric acid) to form the isatin ring. The presence of three electron-withdrawing chlorine atoms deactivates the aromatic ring, necessitating rigorous control of temperature (70-80°C) and acid concentration to drive the cyclization to completion without degrading the sensitive isatin moiety. The subsequent oxidation step utilizes hydrogen peroxide in an alkaline medium, a green chemistry approach that cleaves the isatin ring to reveal the amino-benzoic acid functionality. This transformation is critical as it sets up the carbon skeleton for the eventual side-chain extension.
The reduction of the carboxylic acid to the benzyl alcohol using lithium aluminum hydride is a high-energy transformation that requires strict anhydrous conditions, typically in tetrahydrofuran at 0°C. This step is mechanistically significant because it converts a planar, resonance-stabilized acid into a flexible alcohol capable of nucleophilic attack. Following chlorination with thionyl chloride, the resulting benzyl chloride acts as an electrophile in a nucleophilic substitution with glycine ethyl ester. Finally, the cyclization with cyanogen bromide activates the amine nitrogen to attack the ester carbonyl, closing the second heterocyclic ring. This final intramolecular amidation is the key structural determinant that defines the molecule as an Anagrelide analogue, locking the conformation into the bioactive scaffold required for impurity identification.
How to Synthesize Anagrelide Trichloro Derivative Efficiently
The synthesis of this critical pharmaceutical intermediate requires precise adherence to stoichiometric ratios and temperature profiles to maximize yield and purity. The patented process outlines a clear progression from raw material to finished good, emphasizing the importance of intermediate purification. For technical teams looking to implement this route, the following guide summarizes the operational workflow derived directly from the patent specifications. Detailed standard operating procedures regarding specific solvent volumes, quenching protocols, and drying parameters are essential for reproducibility and should be consulted alongside this overview.
- Perform a Sandmeyer reaction on 3,4,5-trichloroaniline using chloral hydrate and hydroxylamine hydrochloride under acidic conditions to yield 4,5,6-trichloroisatin.
- Oxidize 4,5,6-trichloroisatin using hydrogen peroxide in an alkaline sodium hydroxide solution to obtain 6-amino-2,3,4-trichlorobenzoic acid.
- Reduce the carboxylic acid group of 6-amino-2,3,4-trichlorobenzoic acid using lithium aluminum hydride in tetrahydrofuran to form 6-amino-2,3,4-trichlorobenzyl alcohol.
- Chlorinate the benzyl alcohol with thionyl chloride followed by condensation with glycine ethyl ester hydrochloride to generate N-(2,3,4-trichloro-6-amino-benzyl) glycine ethyl ester.
- Execute a final cyclization reaction using cyanogen bromide and triethylamine to close the imidazoquinazoline ring, yielding the target Anagrelide trichloro derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers distinct strategic advantages beyond mere technical feasibility. The primary benefit lies in the utilization of commodity-grade starting materials. 3,4,5-Trichloroaniline is a bulk chemical produced globally, ensuring that the supply chain is not dependent on niche or single-source vendors. This broad availability significantly mitigates the risk of raw material shortages that often plague the production of specialized pharmaceutical intermediates. Furthermore, the reagents used throughout the five steps, such as thionyl chloride, hydrogen peroxide, and triethylamine, are staples of the fine chemical industry, allowing for competitive pricing and easy sourcing from multiple suppliers.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and the avoidance of precious metal catalysts. Traditional cross-coupling reactions often require expensive palladium or rhodium catalysts which add substantial cost and require complex removal steps to meet residual metal specifications. By utilizing classical organic synthesis techniques like Sandmeyer reactions and nucleophilic substitutions, this route eliminates the need for costly transition metals. Additionally, the ability to isolate stable intermediates allows for better inventory management, reducing waste associated with failed batch runs and optimizing the overall cost of goods sold for the final reference standard.
- Enhanced Supply Chain Reliability: The linear nature of the synthesis enhances supply chain visibility and control. Each of the five steps produces a discrete, isolable solid or liquid intermediate, which simplifies quality control checkpoints. If a specific batch fails quality assurance at an early stage, it can be reworked or discarded without compromising the entire production campaign. This modularity ensures a consistent output of the final Anagrelide trichloro derivative, which is crucial for maintaining the continuous validation schedules of API manufacturers. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in environmental parameters, further stabilizing delivery timelines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for straightforward scale-up. The reactions do not require ultra-high pressures or cryogenic temperatures below 0°C, making them compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The use of hydrogen peroxide as an oxidant generates water as a by-product, aligning with green chemistry principles and reducing the burden on wastewater treatment facilities. This environmental compatibility facilitates easier permitting and regulatory approval for manufacturing sites, ensuring long-term production continuity without the risk of shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the Anagrelide trichloro derivative. These insights are derived from the detailed experimental data and claims within the patent documentation, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: Why is the Anagrelide trichloro derivative critical for pharmaceutical manufacturing?
A: This compound is a specified impurity in both the United States Pharmacopeia (USP) and European Pharmacopoeia (EP) standards for Anagrelide hydrochloride. Accurate synthesis and characterization are essential for validating the purity and safety of the final API.
Q: What are the key advantages of this patented synthesis route?
A: The patented method offers a linear, high-yield pathway starting from readily available 3,4,5-trichloroaniline. It eliminates the need for complex, unreported procedures, providing a reliable and reproducible method for generating reference standards.
Q: Can this process be scaled for commercial production of reference materials?
A: Yes, the process utilizes standard industrial reagents such as thionyl chloride and lithium aluminum hydride. The reaction conditions, including temperature controls between 0°C and 80°C, are compatible with standard stainless steel reactors used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anagrelide Trichloro Derivative Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your pharmaceutical products depends on the quality of your reference standards and intermediates. Our team of expert chemists has extensively analyzed the synthetic pathways described in patent CN107903269B and possesses the technical capability to execute this complex five-step sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need gram-scale samples for method development or kilogram quantities for routine QC, we can meet your demands. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Anagrelide trichloro derivative meets the exacting standards required by the USP and EP.
We invite you to collaborate with us to secure a stable and cost-effective supply of this critical intermediate. By leveraging our process optimization expertise, we can help you achieve significant efficiencies in your quality control operations. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your Anagrelide hydrochloride production remains compliant and uninterrupted.
